



Aquatic oligochaeta is one of the most abundant and ecologically important groups in freshwater benthic environments (Jung, 2011). Most species in this group are < 1mm to a few centimeters in length. This group contains several families. Among them, naidid worms are small benthic and/or epibenthic oligochaetes with a worldwide distribution. About 180 species of naidid oligochaetes have been described(Erséus, 2005) and 24 genera are currently recognized(Envall et al., 2006).
The species diversity of this family has been the subject of considerable investigation in China, Japan, and Korea. Fiftyeight and 30 species have been reported in China and Japan, respectively(Timm, 1999; Wang and Cui, 2007). In recent years, 15 naidid species in 11 genera have been added to the Korean aquatic oligochaete fauna (Jung, 2011, 2012; Park, 2013). Of these, one species in the genus
The specimens were collected with a plankton hand net (mesh size 100μm) from the roots of waterweeds at the edge of streams covered with sand or organic matter. The samples werekept cooland were sorted in the laboratory using a stereomicroscope while the worms were alive. Then, the samples were preserved in 70% ethanol solution. The specimens were stained with Rose Bengal dye for observations and measurements and were temporarily mounted in glycerin. Pictures were taken with a BX41 optical microscope(Olympus,Tokyo, Japan) and an EOS 650D camera(Canon, Tokyo, Japan). The figures are drawn using a microscope equipped with a drawing tube. Measurements were taken using a micrometer on the microscope. These mounted collections are being kept in the lab of Ecology Genetic(LEG), Department of Science Education, Ewha Womans University. Other material, preserved in 70% ethanol solution, was submitted to the National Institute of Biological Resources(NIBR) of the Republic of Korea.
Order Haplotaxida Family Naididae Ehrenberg, 1828 Genus Dero Oken, 1815
Xantho decaopada Dutrochet, 1819: 155. Nais decapoda(Dutrochet). Blainville, 1825: 131. Uronais decapoda(Dutrochet). Gervais, 1838: 18. Dero decapoda(Dutrochet). Vaillant, 1890: 386. Dero darsale Ferronière, 1899: 255. Dero tubicola Pointner, 1911: 274, Pl. XXVIII, figs. 4, 5; Schuster, 1915: 18, figs. 10-14; Malevich, 1929: 47; Ude, 1929: 36, fig. 41. Dero austrina Stephenson, 1925: 882, Pl. I, fig. 1; Aiyer, 1929: 34, figs. 10, 11; Stephenson, 1931: 269; Michaelsen, 1933: 334; Chen, 1940: 57, figs. 18, 19. Dero dorsalis Ferronière, Michaelsen, 1933: 334; Sperber, 1948: 162; 1950: 70, figs. 22, 23a; Cekanovskaya, 1962: 171, fig. 90; Naidu, 1962: 529, fig. 12a-h.
Material examined. All specimens were collected from Geumchoncheon stream, Geumchon-dong, Paju-si, Gyeonggi-do, Korea, 37˚ 76′64.69′′N, 126˚ 77′42.50′′E, 16 Jun, 2013 (collector: Jung J). KN 1306161, KM1306162, KM1306163, and KM1306164: mature, mounted on slides, deposited at LEG. KOSPIV0000181419, KOSPGR0000258786, and KO SPIV0000181431: mature, preserved in 70% ethanol solution, mature specimen, deposited at NIBR.
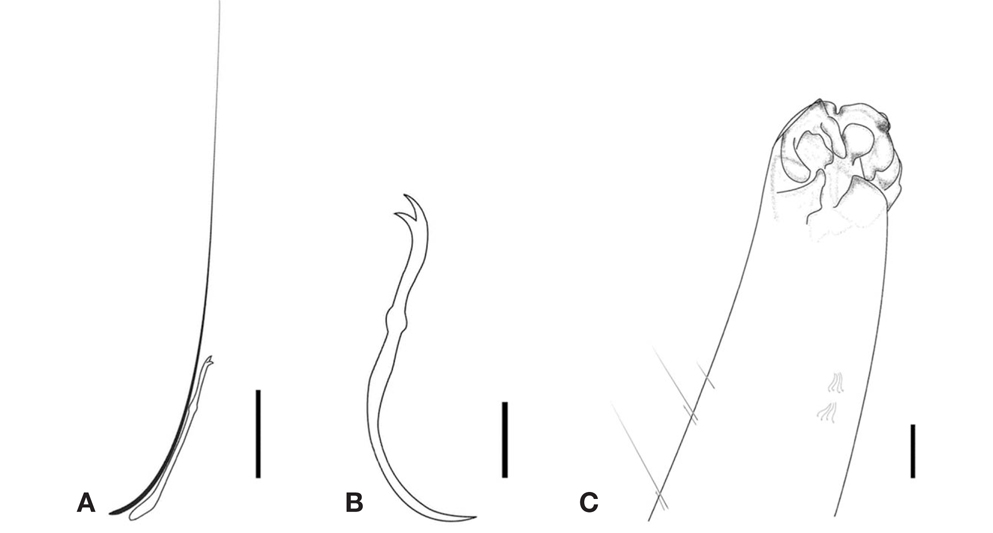
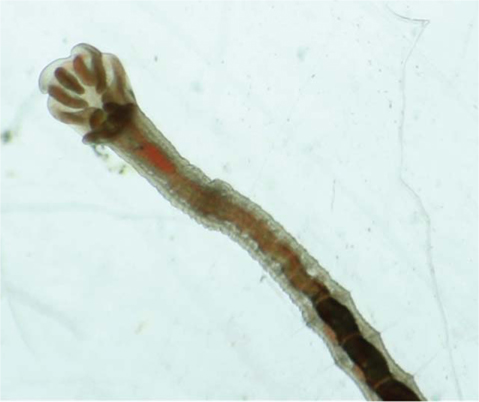
Diagnosis. Length 1.04-2.76mm, width at XI 0.11-0.25mm, number of segments 16-85. Small worms red in color. No eye present. Dorsal chaetae starting from IV, one hair and one needle; needle chaeta with split end; length of hair chaeta about three times longer than needle chaeta (Fig. 1A). Ventral chaetae of anterior segments, bifid, arranged in two rows of 4-5 chaetae per bundle, 35.68-36.37 μm long, 0.72-1.47 μm wide, with distal nodulus and longer upper tooth (Fig.1B). Ventral chaetae of posterior segments with proximal bend. Branchial fossa with two diverging processes from the postero-lateral border(Figs. 1C, 2). Five pairs of gills; two postero-ventral foliate, one lateral foliate, and 2 antero-dorsal foliates(Fig. 1C).
Distribution. Europe, South Asia, and East Asia.
Remarks.
>
Nais pectinata Stephenson, 1910: 236, Pl. XI, fig. 1a-f; 1920: 198; 1923: 63, fig. 19a-f; 1932: 229, fig. 1a, b; Aiyer, 1929: 19, fig. 2a-d. (non) Nais pectinata Stephenson. Stephenson, 1931: 302. Nais denticulata Chen, 1940: 39, fig. 8A-E. Allonais pectinata(Stephenson). Sperber, 1948: 206; Naidu, 1962: 915; Brinkhurst, 1971: 125, fig. 4J; 1986: 85; Ohtaka, 2003: 194, 195, fig. 3c.
Material examined. All specimens collected from a shallow ditch covered with duckweed in Mangchi-ri, Irun-myeon, Geoje-si, Gyeongsangnam-do, Korea, 34˚48′52.38′′N, 128˚ 40′31.15′′E, 24 Apr 2013(collector Lee J). GN1304241 and GN1304242: mature, mounted on slides; deposited at LEG. KOSPIV0000193673, KOSPGR0000257078, and KOSPIV 0000193707: mature, preserved in 70% ethanol solution; deposited at NIBR.
Diagnosis. Length 0.7-1.54mm, width at VI 0.19-0.43mm, number of segments 31-55. No eyes. Prostomium rounded. Dorsal chaetae starting from III. Dorsal chaetae of III with no hair, two needles with pectinate tips(Fig. 3A). Dorsal chaetae bundles of remaining segments, consisting of one hair and one needle with pectinate tip(Fig. 3C). Dorsal chaetal bundle rarely consisting of one needle and two hairs. Pectinate needles consisting of marginal teeth of almost equal length and 3-5 intermediate teeth. Ventral chaetae beginning from II, four per bundle, sometimes one or two per bundle; the upper tooth slightly longer than the lower tooth; upper tooth thinner than lower tooth(Fig. 3D).
Distribution. North America, Asia, Australia, and Africa.
Remarks. This is the first record of
![Allonais pectinata (Stephenson, 1910). A, Dorsal pectinate chaetae of III (length 2.52 μm [left], 2.62 μm [right]); B, Dorsal chaeta of IV (length 3.30 μm); C, Dorsal chaetae of VI (length 3.91 μm [left], 3.37 μm [middle]); D, Ventral chaeta of III (length 5.93μm). Scale bars: A-D=1μm.](http://oak.go.kr/repository/journal/13258/DMBRBT_2014_v30n2_119_f003.jpg)