Defects in the cholesterol metabolism leading to hypercholesteremia are very important and recognized risk factors for atherosclerosis and cardiovascular diseases [1-3]. Abnormalities in lipid profiles associated with obesity are characterized by elevated triglycerides and low-density lipoproteins (LDLs) with abnormal compositions and structures [4]. Epidemiological studies have shown that high-density lipoprotein (HDL)-cholesterol levels could potentially contribute to anti-atherogenesis, including inhibition of LDL oxidation to protect the endothelial cells from the cytotoxic effects of oxidized LDL [5, 6]. Besides medication, concerted efforts to make changes in life styles and dietary regulations are being advised to control the epidemic of cardiovascular ailments affecting the populations across the world.
Cereals and millets are important components of the human diet throughout the world, particularly in tropical and subtropical regions [7]. Cereals constitute a major source of dietary carbohydrates, proteins, vitamins and minerals, especially for the vegetarians worldwide [8]. The present study deals with a formulated diet comprising of millets and cereals -
Adult albino female rats (
At the culmination of experiment, animals were fasted overnight and sacrificed under mild ether anesthesia. Blood was collected by retro-orbital puncture into ethylenediaminetetraacetic acid (EDTA)-coated tubes, and plasma was separated by centrifugation. Livers and kidneys were excised, and both plasma and tissues were kept frozen until analyzed. Fecal samples were also collected for biochemical analyses.
Plasma total lipid (TL) content was estimated by using the sulpho-phospho-vanillin method [10]. Plasma total cholesterol (TC), HDL cholesterol (HDL-C) and triglycerides (TG) were measured by using standard kits (Eve’s Inn Diagnostics, Vadodara, India). The low-density lipoprotein cholesterol (LDL-C = TC − HDL-C − TG/5) and very low density lipoprotein cholesterol (VLDL-C = TG/5) levels and the atherogenic index (AI = TC/HDL-C) were calculated [11]. The hepatic TL was extracted in a chloroform and methanol (2 : 1) mixture [12] and was estimated by using a gravimetric analysis. The same extract was used to estimate the TC and the TG contents by using the respective kits (Eve’s Inn Diagnostics, Vadodara, India).
Hepatic HMG-CoA (3-hydroxy-3-methyl-glutaryl-CoA) reductase (EC 1.1.1.34) activity was measured in terms of the ratio of HMG-CoA to mevalonate [13]. Colorimetric assays were performed for both HMG-CoA and mevalonate by using hydroxylamine reagent at alkaline pH and acidic pH, respectively, and the ratio of HMG-CoA to mevalonate was determined, which is inversely proportional to the enzyme activity. The alkaline ethanolic extract of liver tissue was acidified, and the total bile acid content was estimated using vanillin-phosphoric acid reagent [14].
The fecal cholesterol was extracted using a chloroform and methanol (2 : 1) mixture [12], and the total cholesterol content was estimated by using standard kits (Eve’s Inn Diagnostics, Vadodara, India). From the same extract, TL was estimated by using a gravimetric analysis. The alkaline ethanolic extract of fecal matter cholesterol was acidified and was used with vanillin-phosphoric acid reagent to estimate the total bile acid content [14].
The hepatic and renal lipid peroxidation (malondialdehyde concentration) was determined by using a thiobarbituric acid (TBA) assay [15]. Total ascorbic acid was estimated using 2,4-dinitrophenyl hydrazine reagent [16]. Superoxide dismutase (SOD; EC 1.15.1.1; nitroblue tetrazolium method) and catalase (EC 1.11.1.6; decomposition of H2O2) activities were assayed following the methods of Kakkar
Data are presented as means ± SEMs (Standard Error of the Mean). A one-way analysis of variance (ANOVA) with Tukey’s significant difference post-hoc test was used to compare differences among groups. Data were statistically handled by Graph Pad Prism Version 3.0 statistical software.
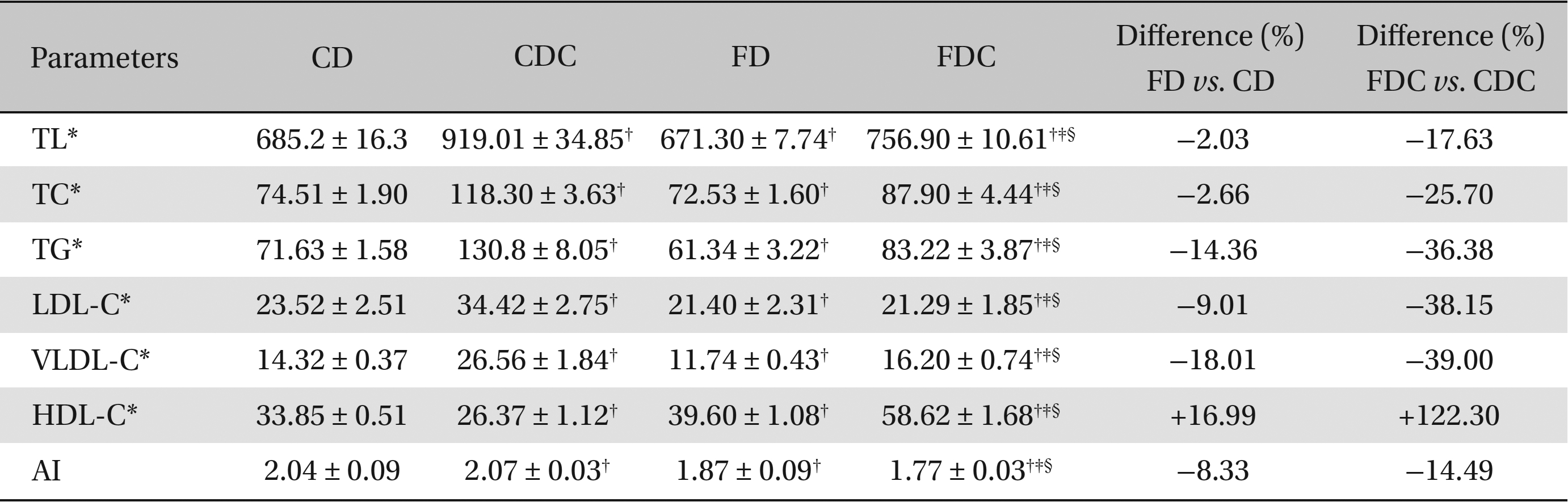
When the animals were fed commercial and formulated diets, the plasma lipid profiles (except HDL-C) in the FD group were significantly lower than those in the CD group. While the HDL-C content in the FD group was significantly elevated, the atherogenic index was substantially lower. With the addition of cholesterol to the diet of the CDC group, significant increases in plasma lipid profiles occurred, but the HDL-C content declined. On the other hand, although the FDC group exhibited elevated TL, TC, TG, and VLDL-C levels, the LDL-C level and the AI did not show any significant increases. Additionally, the HDL-C content in the FDC group was increased by 122%. The differences between the FD and the CD groups and between the FDC and the CDC groups clearly indicate the influence of a formulated (multigrain) diet, with or without cholesterol added, on lipid metabolism (Table 1).
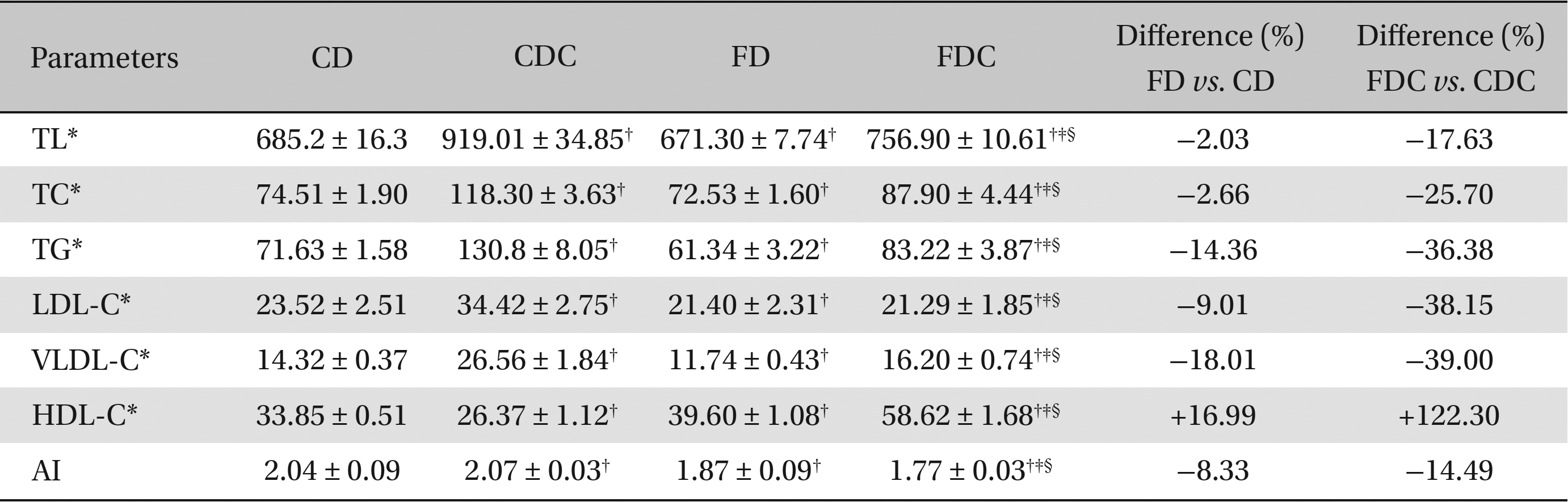
Effects of a multigrain-formulated diet on the plasma lipid profiles of the experimental animals
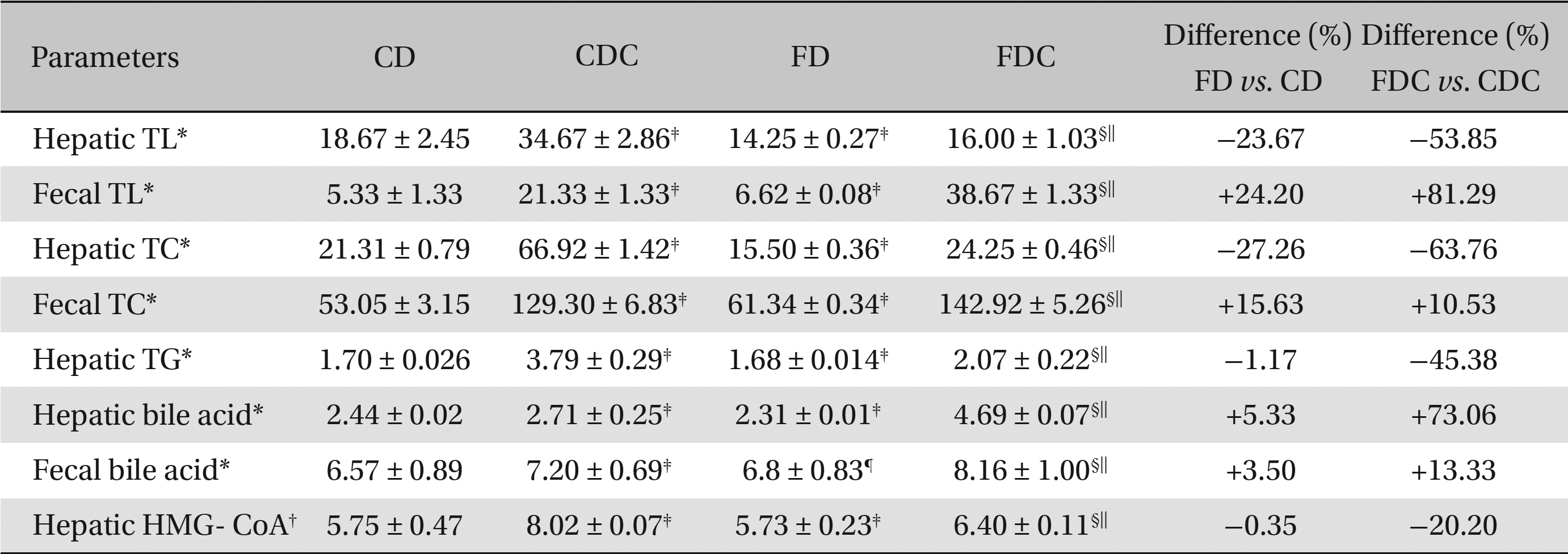
The hepatic and the fecal lipid profiles of the CD, FD, CDC, and FDC groups essentially reflect the trends of the lipid profiles observed in the plasmas of these groups. When compared to the CD group, the FD group exhibited declined hepatic TL, TC and TG levels with an increase in hepatic and fecal bile contents and fecal TL and TC.
However, lipid synthesis in the hepatic tissues of the CD and the FD groups did not vary significantly. Between the CDC and the FDC groups, the FDC group showed significant improvements in hepatic, as well as fecal, lipid profiles over the CDC group. The hepatic HMG-CoA: mevalonate ratio in the FDC group was found to be 20% lower than that in the CDC, indicating an increase in hepatic HMG-CoA activity (Table 2).
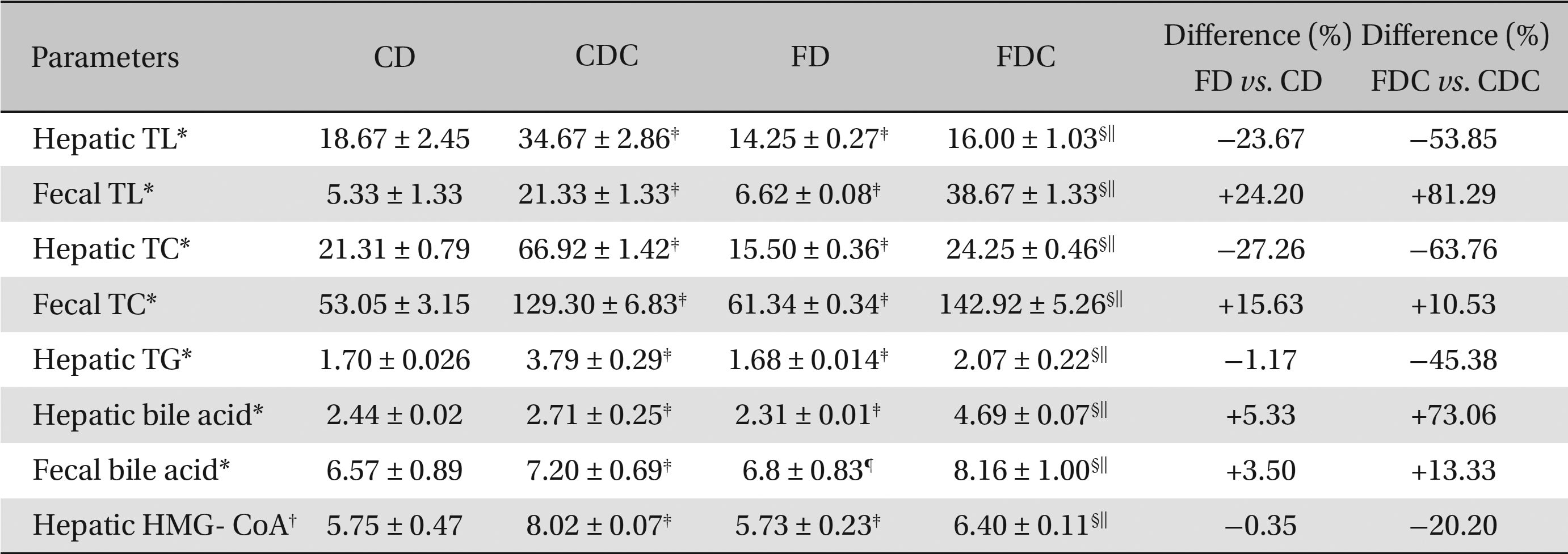
Effects of a multigrain-formulated diet on the hepatic and the fecal lipid profiles and on the HMG-CoA reductase activity of the experimental animals
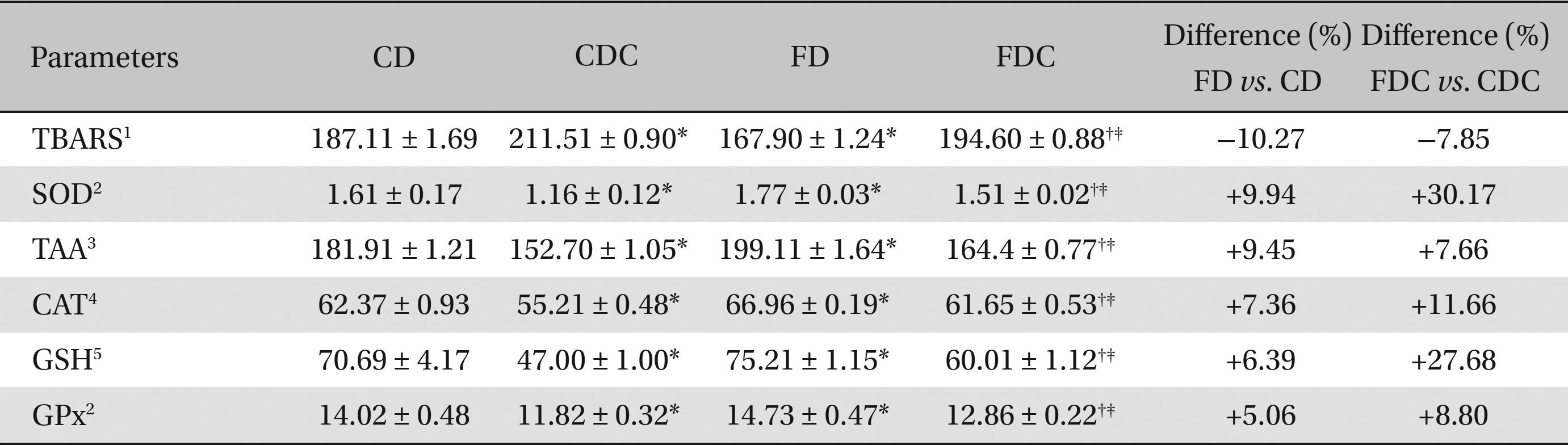
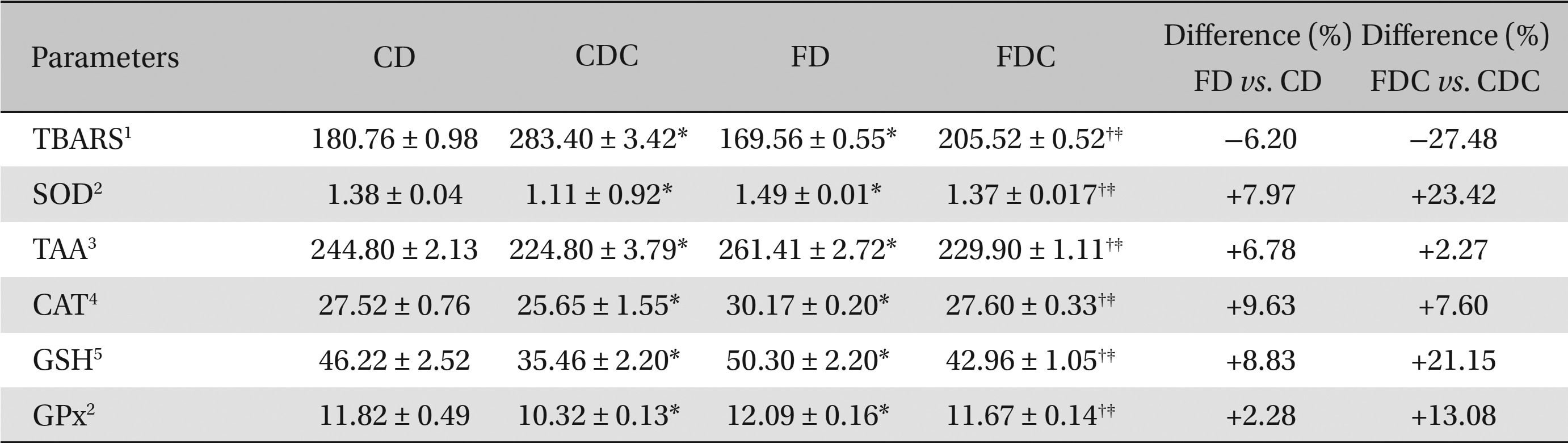
Between the CD and the FD groups, the FD group registered lowered hepatic and renal lipid peroxidation and increased antioxidant profiles, i.e., increases in SOD, catalase CAT and GPx activities; the total ascorbic acid TAA and glutathione content (in both hepatic and renal tissues) also increased in the FD group. A similar improvement was found in the FDC group, as compared to the CDC group, with reference to lipid peroxidation and to the non-enzymatic and the enzymatic antioxidant parameters (Tables 3,4).
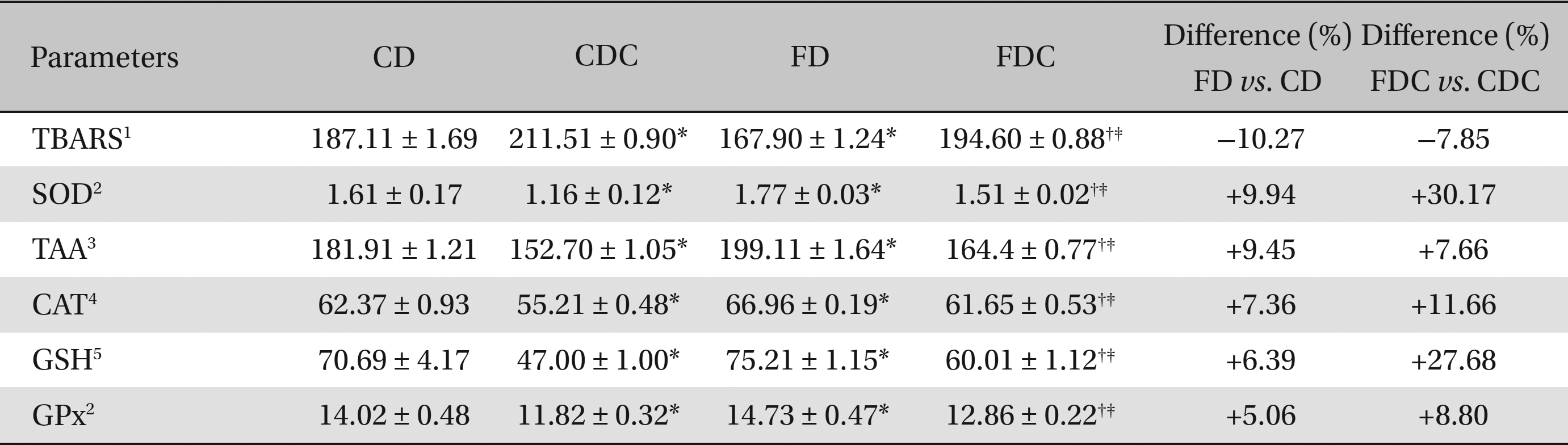
Effects of a multigrain-formulated diet on the hepatic lipid peroxidation and antioxidant status of the experimental animals
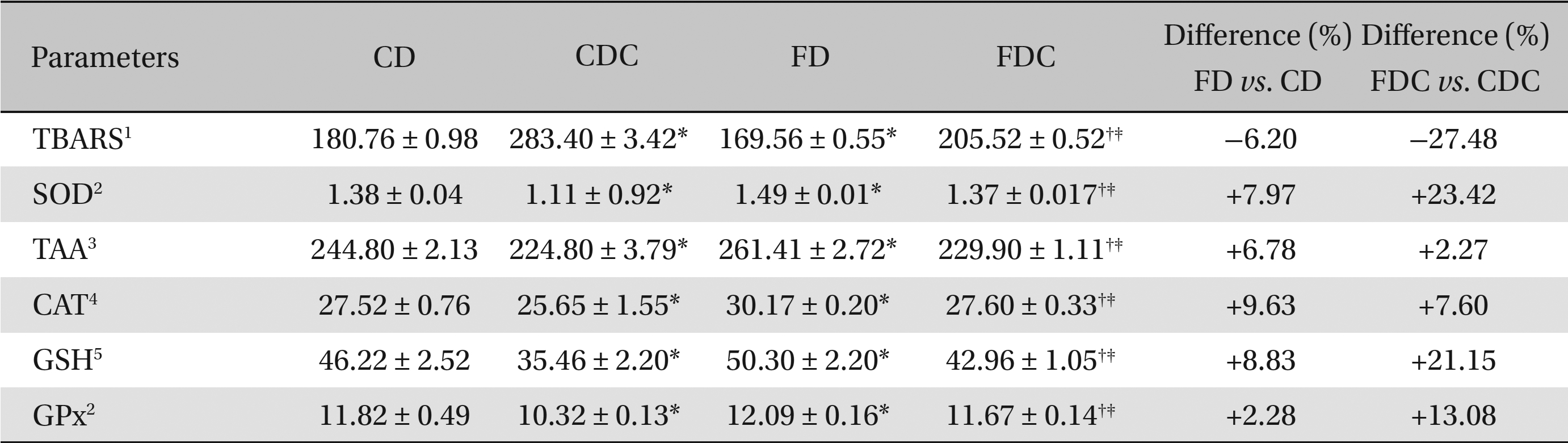
Effects of a multigrain-formulated diet on the renal lipid peroxidation and antioxidant status of the experimental animals
High plasma cholesterol represents a major risk factor for ischemic heart diseases. Defects in the cholesterol metabolism are major causes of cardiovascular disorders [21]. Chronic consumption of a diet with high fat content is well known to lead to an increase in plasma cholesterol, resulting in premature atherosclerosis [22]. The incidence of atherosclerosis and cardiovascular disease is related to high levels of plasma lipids, especially low density lipoprotein cholesterol (LDL-C), which is a predisposing factor for cardiovascular disorders [23, 24]. On the other hand, high density lipoprotein cholesterol (HDL-C) has a protective effect [25].
In the present study, we have examined the role of a multigrain diet in the regulation of body lipid and antioxidant metabolism. Since hyperlipidemia is often associated with high fat diet consumption, the animals (albino rats) were administered cholesterol along with two different diets: a commercial (basal) diet and a formulated diet. When the formulated diet alone was administered in the FD group, the formulated diet brought about a significant decline (2% - 18%) in the basal lipid profiles, an increase in HDL-C content (by 17%), and a decline in the atherogenic index (by 8%) when compared to the effects of a commercial diet in the CD group. With the addition of cholesterol to both the diets, the plasma TL, TC, TG and VLDL-C levels in both the CDC and the FDC groups exhibited significant increases compared to the levels in the CD and the FD groups. However, when the plasma lipid profiles of the FDC group were compared with those of the CDC group, the former registered significantly lower TL, TC, TG, LDL-C, and VLDL-C levels and atherogenic index. Further, the HDL-C content in the FDC group was increased by about 122% (Table 1). The hypolipidemic effect of the formulated diet was also seen in both the FD and the FDC groups, where the hepatic TL, TC and TG levels declined appreciably with improvements in fecal TL and TC contents. Concomitant increases in hepatic and fecal bile acid contents in the FDC group also confirmed the lipid regulatory role of the diet. Owing to an increased fecal excretion of lipids in the group fed the formulated diet, the HMG-CoA: mevalonate ratio in hepatic tissue decreased (Table 2), indicating an increased lipid synthesis, perhaps as a result of a feedback mechanism in the cholesterol biosynthesis. Comparisons between the FD and the CD groups and between the FDC and the CDC groups with regard to hepatic and fecal lipid profiles clearly indicate that while hepatic TL, TC, and TG declined sharply in the FD and the FDC groups, the fecal excretion of TL and TC in these groups significantly increased compared to the CD and the CDC groups (Table 2).
As compared to the commercial diet, the formulated diet appeared to lower the lipid peroxidative processes and elevate the antioxidant status significantly in hepatic and renal tissues: particularly, the enzymatic activities of SOD, CAT, and GPx and the contents of TAA and GSH were increased significantly in the FDC group when compared to the CDC group. When the FD and the FDC groups were compared with their counterparts (the CD and the CDC groups), clear increases in the hepatic and renal enzymatic (7% - 30%) and non-enzymatic (5% - 27%) antioxidant contents were observed in the FD and the FDC groups (Tables 3,4) Concomitant increases in hepatic bile acid contents in the FD and the FDC groups (5.33% and 73%) over the CD and the CDC groups, along with elevated fecal bile acid contents in these groups (3.5% - 13%) over the CD and the CDC groups, also indicate the hypolipidemic activity of the formulated diet (Table 2).
The antioxidant status was elevated significantly in the FD and the FDC groups when compared to that in the CD and the CDC groups as can be seen from the differences found between the FD and the CD and between the FDC and the CDC groups. Among the enzymatic activities of the antioxidant enzymes in hepatic and renal tissues, the SOD activity in both the FD and the FDC groups was highest (9.94% and 30% higher than in the CD and the CDC groups) as compared to CAT and GPx activities. While the increases in non-enzymatic antioxidants, TAA and GSH, contents in the hepatic and renal tissues of the FD and the FDC groups were of the order of 2.27% to 11.66% (TAA) and 6.39% to 27.68% (GSH), the lipid peroxidation (formation of thiobarbituric acid reactive substances) in both the liver and kidneys of the FD and the FDC groups was found to be significantly lower (ranging from 6% to 27%) than it was in the CD and the CDC groups (Tables 3,4)
The results of the present study clearly demonstrate the physiological role of a multigrain diet in metabolic regulations of lipid and antioxidant profiles in hypercholesteremic rats. The presently observed hypolipidemic and antioxidant effects of a multigrain diet could be attributable to the dietary components and to the phytochemical/ nutrient composition of the formulated diet. While the commercial diet contained wheat and soy as the principal components, the formulated diet was made up of
In conclusion, the formulated multigrain diet used in the present investigation is found to be superior to a basal (commercial) diet in controlling both the lipid and the antioxidant profiles of the animals exposed to a cholesterol- containing diet. Therefore, mixed diets (using different grains) could be useful as they can improve and maintain health while obviating the necessity for drug intake over long periods.