Molluscan shellfish are an important food resource, but contamination with natural toxins is a worldwide problem. Shellfish toxins are natural toxins that have been detected in molluscan shellfish, such as mussels, oysters, scallops, and clams. Contaminated shellfish pose a risk to human health if consumed. Shellfish toxins that contaminate bivalves include saxitoxin (STX) and its derivatives, which cause paralytic shellfish poisoning (PSP); okadaic acid (OA) group toxins, which cause diarrhetic shellfish poisoning (DSP); domoic acid (DA), which causes amnesic shellfish poisoning (ASP); and azaspiracid (AZA) group toxins, which cause azaspiracid shellfish poisoning (AZP) (Noguchi, 2003; Toyofuku, 2006). The toxins are produced mainly by algae or phytoplankton, and can accumulate in the digestive gland (hepatopancreas) of filter-feeding molluscan shellfish. They cause diseases with both gastrointestinal and neurologic symptoms in humans (Edebo et al., 1988; Silvert and Subba Rao, 1992; Indrasena and Gill, 2000; Toyofuku, 2006).
PSP toxins are produced mainly by dinoflagellates belonging to the
In our previous study, the accumulation and occurrence of PSP toxins in various bivalves in Jinhae Bay was assessed by mouse bioassay (MBA) (Mok et al., 2012), and a comparison study of MBA and pre-column high-performance liquid chromatography (HPLC) oxidation methods was conducted in mussels (Mok et al., 2013). Since aquaculture is the main source of mussels and oysters in Korea, the monitoring of PSP toxins in shellfish farming areas is important for both the industry and the consumer. In the present study, seasonal variation in PSP toxins in bivalves collected from Jinhae Bay in Korea was surveyed by pre-column HPLC oxidation. We also confirmed the profiles in the major bivalves, such as oysters
All solvents and reagents for sample preparation and HPLC analysis were of analytical or HPLC grade. Hydrochloric acid, sodium phosphate, hydrogen peroxide, acetic acid, methanol, acetonitrile, and acetic acid were obtained from Merck (Darmstadt, Germany). Sodium hydroxide and periodic acid were purchased from Sigma (St. Louis, MO, USA). Ammonium formate was obtained from Fluka (Buchs, Germany). The oxidant solution for periodate oxidation was prepared by mixing 5 mL each of 0.03 M periodic acid, 0.3 M ammonium formate, and 0.3 M sodium phosphate (Na2HPO4), and adjusting the pH to 8.2 with 0.2 M sodium hydroxide. De-ionized water was passed through a Milli-Q water purification system (Millipore, Bedford, MA, USA) and used for the preparation of HPLC mobile phases.
Certified reference materials (STX, decarbamonyl STX [dc STX], neoSTX [NEO], gonyautoxin [GTX] 1&4, GTX 2&3, decarbamonyl GTX [dcGTX] 2&3, GTX 5, and N-sulfocarbamoylgonyautoxin C [C] 1&2) were purchased from the Institute for Marine Biosciences of the National Research Council (Halifax, Nova Scotia, Canada) in glass ampoules as solutions in 0.1 M acetic acid. Stock standard solutions were prepared by diluting primary toxin standard solutions in water eight times. Working standard solutions were prepared by dilution of stock solutions in 0.1 mM acetic acid to produce instrument calibration standards for use in analysis of PSP toxins by pre-column HPLC oxidation.
Samples for analysis of PSP toxins were collected at fixed monitoring stations in Jinhae Bay in Korea in 2009 (Fig. 1). Bivalve samples such as cultured mussel
The outside of each shellfish was thoroughly cleaned with
fresh water. The shellfish samples were shucked, and drained onto a No. 10 sieve for 5 min. A 10-g portion of homogenized shellfish tissue was extracted by boiling for 5 min in 10 mL of 0.1 M HCl according to the extraction method of Lawrence and Niedzwiadek (2001). The crude extracts were centrifuged at 5,000
>
SPE C18 clean up and oxidation
A 1-mL sample of the crude extract was passed through a SPE C18 cartridge (3 mL; Supelco, Bellefonte, PA, USA) previously conditioned with 6 mL of methanol followed by 6 mL of water. The effluent was collected, and the cartridge was washed with 2 mL of water, which was combined with the effluent. For the oxidation reactions, the volume was made up to 4 mL with water. For HPLC analysis, the sample extracts were oxidized using both periodate and peroxide, as described by Lawrence and Niedzwiadek (2001).
Analysis of the PSP toxins was carried out by the pre-column HPLC oxidation method. The HPLC unit consisted of a Surveyor MS Pump Plus pump and a Surveyor AS Plus needle assembly (Thermo Electron, San Jose, CA, USA). A fluorescence detector (Finnigan Surveyor FL Plus Detector; Thermo Electron) was used for detection of toxins. Fluorescence excitation was set to 340 nm and emission to 395 nm. The column used was a Supelcosil C-18 column (15 cm × 4.6 mm id, 5 μm; Supelco). Mobile phase A was 0.1 M ammonium formate and mobile phase B was 0.1 M ammonium formate with 5% acetonitrile. The analytical conditions were as described previously (Mok et al., 2013).
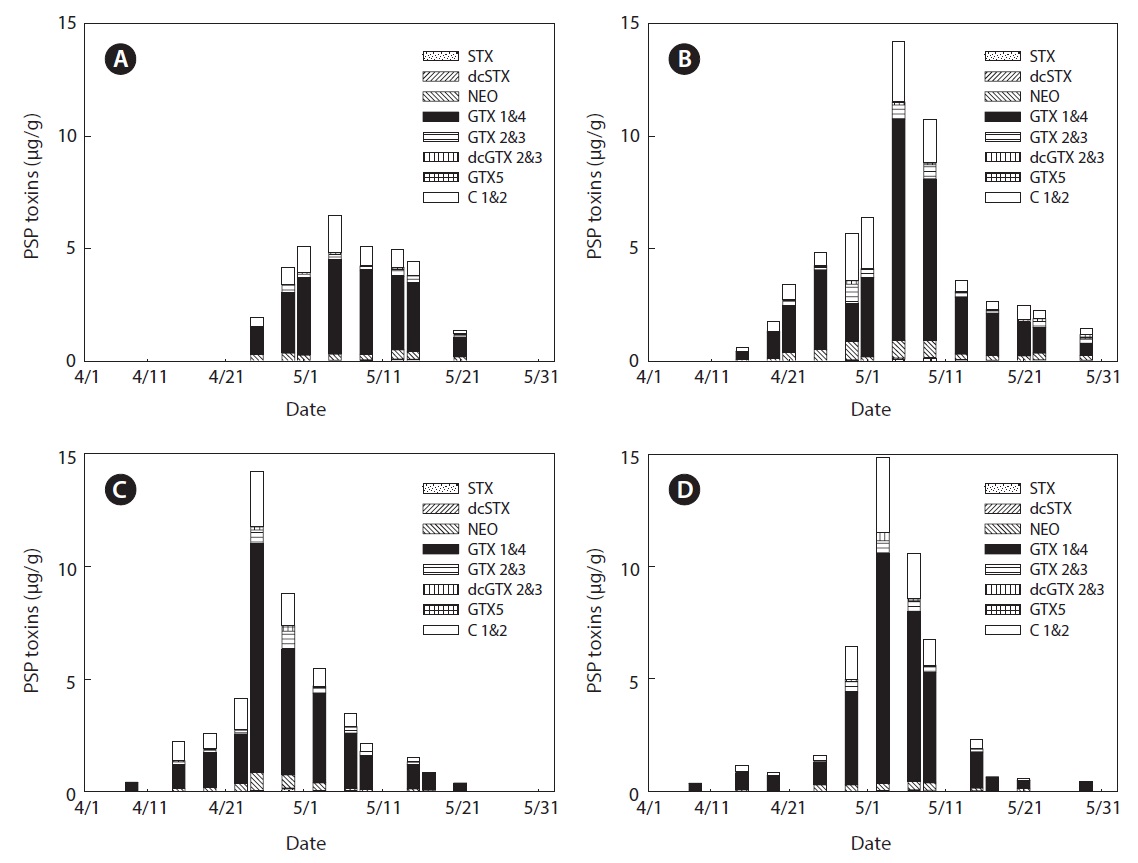
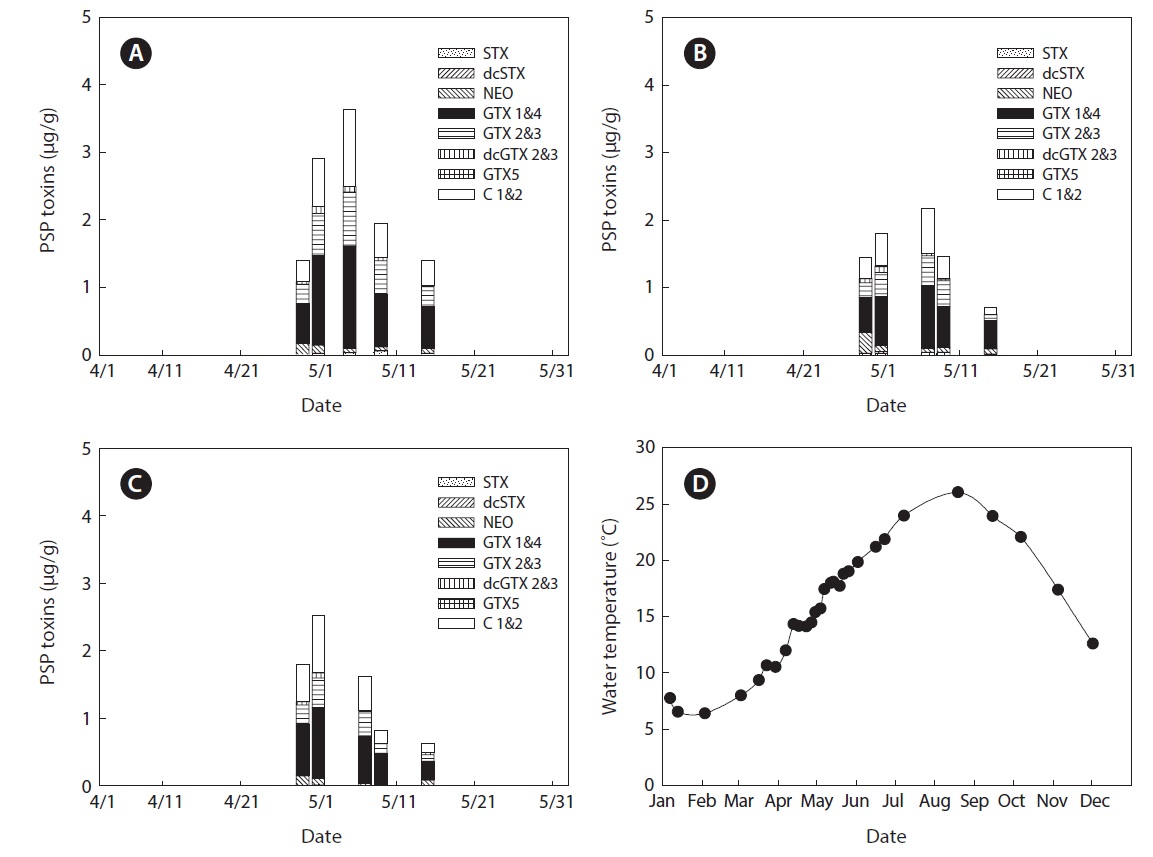
The variability of PSP toxin levels in the bivalves at each sampling station in Jinhae Bay in 2009 is shown in Figs. 2 and 3. PSP toxin levels in the bivalves, except in April and May 2009, are not shown in the figures, because no toxins were detected. During the survey period, PSP toxins in mussels were detected in 45 of 88 samples collected at four stations (Fig. 2), while toxins in oysters were detected in 15 of 95 samples collected at three stations (Fig. 3). The PSP toxin
levels in the bivalves showed remarkable seasonal variation. The overall seasonal trend showed that high toxicities were a problem during spring. The highest total toxin levels in 2009 at all stations were recorded in May (Figs. 2 and 3). We previously reported that high amounts of PSP toxins were detected in bivalves in Jinhae Bay in spring 2005 and 2006 by MBA (Mok et al., 2012). Many authors have reported high levels of PSP toxins during spring in Jinhae Bay (Chang et al., 1989; Park et al., 2000; Shon et al., 2009).
Variation in mean surface water temperature in Jinhae Bay in 2009 is shown in Fig. 3D. During the survey period, the water temperature ranged from 6.4℃ in February to 26.1℃ in August. For the period in which PSP toxins were detected in bivalves, the water temperature ranged from 12.0℃ in April to 19.0℃ in May. The variation in PSP toxin levels was closely related to water temperature, because the toxins are produced mainly by dinoflagellates (Noguchi, 2003; Toyofuku, 2006).
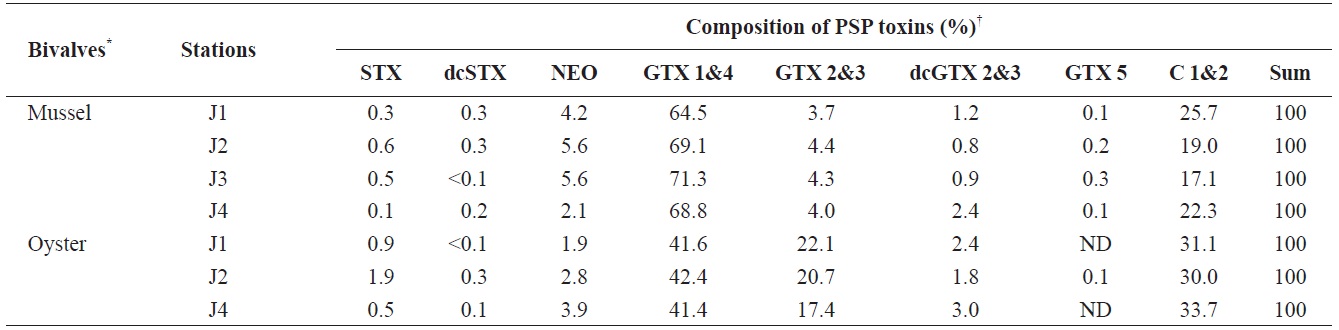
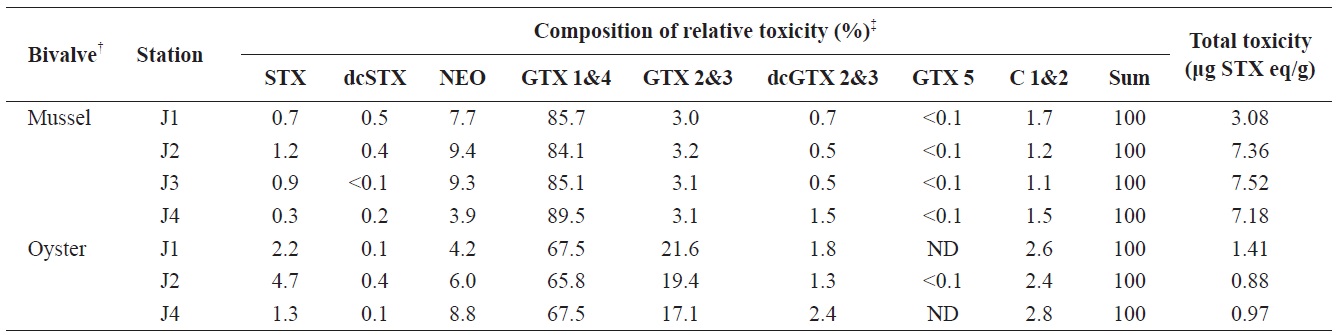
A specific difference in the profiles and levels of PSP toxins was observed between mussels and oysters collected at the same station during the survey period. The profiles of PSP toxins in bivalves containing the highest PSP toxin levels at each station during the survey period are shown in Table 1. The major toxins in bivalves were GTX 1&4 and C 1&2, but GTX 2&3 was also detected as a major component in oysters. Some toxins, such as STX, dcSTX, dcGTX 2&3, and GTX 5, were detected at levels of below 5% in bivalves. In mussels, GTX 1&4 accounted for the highest proportion (64.5-71.3%) of toxin components, followed (in order) by C 1&2 (17.1-25.7%), NEO (2.1-5.6%), GTX 2&3 (3.7-4.4%), dcGTX 2&3 (0.8-2.4%), STX (1.0-0.6%), dcSTX (<0.1-0.3), and GTX 5 (0.1-0.3%). In oysters, GTX 1&4 showed the highest proportion (41.4-42.4%), followed (in order) by C 1&2 (30.0-33.7%), GTX 2&3 (17.4-22.1%), NEO (1.9-3.9%), dcGTX 2&3 (1.8-3.0%), STX (0.5-1.9%), dcSTX (<0.1-0.3), and GTX 5 (ND-0.1%). It was also confirmed that the greatest toxicity (in μg STX eq/g) was derived from GTX 1&4 (Table 2). Park et al. (2000) also profiled PSP toxins in various bivalves from Jinhae Bay in 1997. They reported that GTX 1&4 accounted for 48.4% of toxin components in mussels, and identified them as a dominant ingredient. Shon et al. (2009) reported that the most abundant toxin components identified in mussels from Jinhae Bay were GTX 1 and GTX 4, followed by C 1, C 2, GTX 3, and neoSTX. A similar trend was observed in this study.
The contents of PSP toxins in the bivalves containing the highest PSP toxin levels at each station during the survey period are shown in Table 2. At station J4, a total toxicity of 7.18 μg STX eq/g was detected in mussels on April 30; in comparison, a total toxicity of 0.97 μg STX eq/g was measured in oysters collected on the same dayons J1, J2, J4, the highest total toxicity in mussels was approximately 4-9-fold the regulatory limit of 80 μg STX eq/100 g, whereas that in oysters from the same station was 1.1-1.8-fold the regulatory limit. Therefore, mussels accumulated more PSP toxins than did oysters. We previously compared the accumulation of PSP toxins in bivalve species from Jinhae Bay in 2005 and 2006. Our data showed that the maximum toxin levels in mussels were 1.6-2.0-, 4.0-5.9-, and 5.1-6.0-fold higher than those in bay scallops, oysters, and short-neck clams, respectively (Mok et al., 2012). Park et al. (2000) also conducted an analysis of PSP toxins in various bivalves from Jinhae Bay in 1997. They found that the toxin levels in mussels were 2.5-, 6.0-, and 7.0-fold higher than those in short-neck clams, oysters, and ark shells, respectively. The same trend was observed in this study. Therefore, mussels could be useful as a bioindicator
[Table 1.] Profiles of PSP toxins (μg/g) in the bivalves collected from Jinhae Bay of Korea in 2009
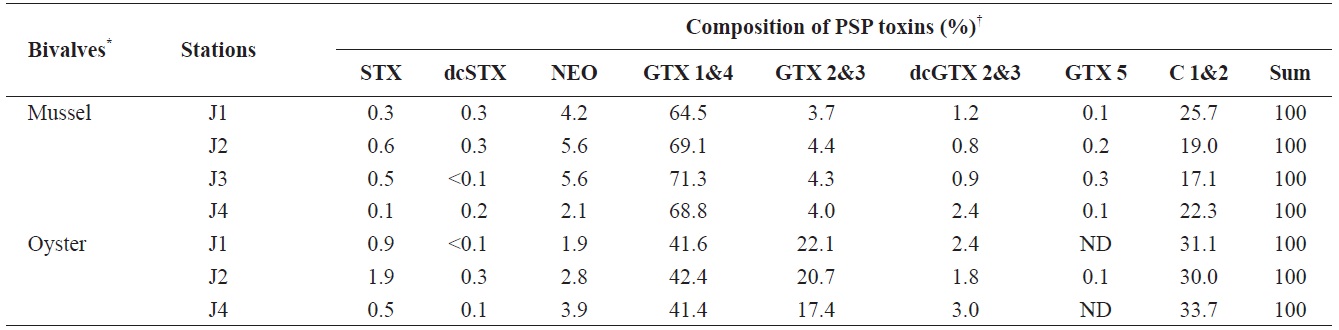
Profiles of PSP toxins (μg/g) in the bivalves collected from Jinhae Bay of Korea in 2009
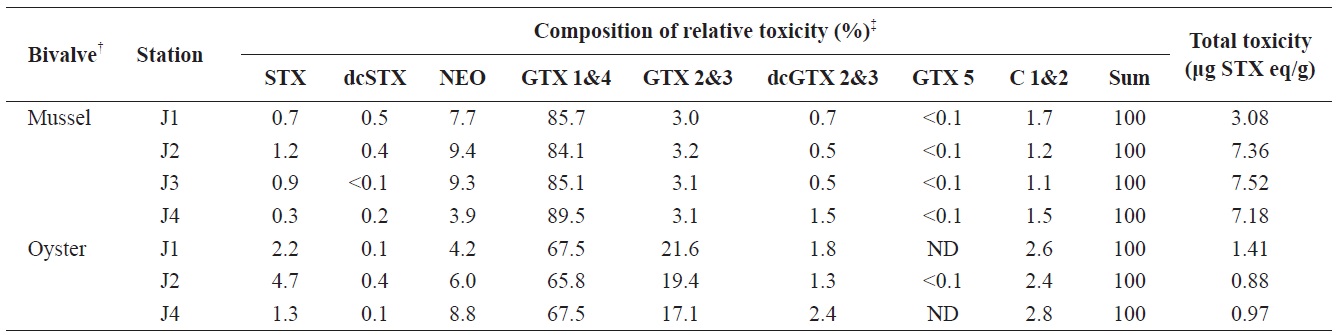
Profiles of relative toxicity (μg STX eq/g)* of PSP toxins in the bivalves collected from Jinhae Bay of Korea in 2009
for PSP toxin monitoring in Korea. Mizuta et al. (1999) analyzed the PSP toxins in mussels and oysters collected from the same area periodically, and found that toxin levels were threefold higher in mussels than in oysters.
The levels of PSP toxins in the bivalves differed significantly according to sample collection station. The highest total toxin level in mussels was measured at station J4 (Table 2, Fig. 2), while that in oysters was found at station J1 (Table 2, Fig. 3). However, the profiles of toxins in the bivalves during the survey period did not differ significantly according to sample collection station.
Korea is the fourth largest producer of shellfish, accounting for almost 2.8% of the global harvest (Lee et al., 2011). Korean shellfish products are exported to mainly the USA, China, Japan, and the EU (Pawiro, 2010; Rees et al., 2010). Jinhae Bay is an important shellfish growing area in Korea, because it produces various shellfish, such as mussels, oysters, ark shells, short-neck clams, butter clams, soft-shell clams, and comb pen shells. This bay is a typical semi-enclosed coastal area surrounded by populated cities and is one of the most eutrophic coastal bays in Korea. The bay is known for recurring red-tide blooms, including
Contamination of shellfish by PSP toxins and cases of PSP have been reported in various countries (Chang et al., 1989; Determan, 2003; Noguchi, 2003; Toyofuku, 2006). Thus, PSP is a serious global public health problem. Our data suggest that the PSP toxin levels in some samples from Jinhae Bay were above the regulatory limit during a specific period in spring. The health of consumers who eat bivalves containing such levels of PSP toxins could be at risk. Therefore, the Korean government operates a monitoring program and an early warning system to ensure the safety of shellfish for consumption.