The recovery and utilization of silk sericin (SS) have attracted much interest among scientists in the field of sericulture, since they can provide an additional benefit to the sericulture industry. SS can be recovered from degumming waste using membrane techniques (Capar
Heavy metal pollution is a serious problem that has a severe impact on the environment and human health. Lead, cadmium, mercury and chromium are the major toxic heavy metals, and their levels in any human commodities are strictly regulated by the government. Among these toxic metal ions, we focused on the removal of chromium using SS. Chromium and its compounds are widely used in various industrial processes such as tanning, electroplating, and synthesis of dyes and pigments. Chromium can exist in eleven valence states, but Cr(III) and Cr(VI) have the largest impact on the environment because of their stability (Kota?
In this study, we have prepared SS macrobeads using a previously reported method (Oh
Silkworm cocoons were kindly provided by Heung Jin Co. Ltd. All chemicals were purchased from Sigma-Aldrich (USA).
SS was extracted by boiling 25 g of Bombyx mori silkworm cocoons with 1 L of distilled water using an autoclave for 1 h at 120℃. The extracted solution was filtered with a nonwoven filter in order to remove the remaining cocoons. The SS solution was frozen at -70℃ for 4 h and lyophilized.
>
Preparation of silk sericin beads
The lyophilized SS was dissolved in 1M LiCl/ DMSO solution for 2 h at 50℃ to prepare a dope solution of 25% (w/v). The dope solution was dropped into alcohol coagulants through a 26G syringe using a syringe pump (KD scientific, USA). Methanol and ethanol were used as coagulants, and the obtained beads are designated as SS-M and SS-E, respectively. The SS beads were left in the coagulant bath for another 1 h. They were then filtered with a nonwoven filter and washed with the same coagulant to remove the residual LiCl and DMSO. To enhance the water stability and mechanical strength of the SS beads, the beads were immersed in a crosslinking reagent. The crosslinking was performed with 2% (v/v) glutaraldehyde (GA) in the same coagulant. The reaction was carried out for 1 h at room temperature. Finally, the SS beads were washed with the same coagulant followed by distilled water to remove the excess GA.
>
Batch adsorption and desorption studies
The stock solution of Cr(VI) (1000 mg/L) was prepared by dissolving 2.827 g of potassium dichromate, K2Cr2O7, in 1 L of distilled water. Different concentrations of Cr(VI) solution were prepared by diluting the stock solution with distilled water. Each adsorption experiment was conducted in a 250 mL Erlenmeyer flask containing 100 mL of Cr(VI) solution. The mixtures were stirred continuously on a multi-stirrer (JEIO TECH, Korea) at 180 rpm for 24 h at 25℃. In order to find the optimum pH, batch adsorption experiments were conducted in a pH range from 1.0 to 5.0 for the Cr(VI) solution (100 mg/L). The adsorption isotherm was studied by varying the initial Cr(VI) concentrations from 25 to 300 mg/L. The concentration of Cr(VI) in the solution was determined using a standard colorimetric method (Snell
where
The desorption of Cr(VI) was performed by using 0.1 M NaOH solution. After the adsorption experiment, the SS beads were recovered from the Cr(VI) solution and washed gently with distilled water three times to remove the residual Cr(VI) on the surface. The beads were immersed into 100 mL of 0.1 M NaOH and stirred overnight. The desorption efficiency was calculated as
>
Characterization of sericin beads
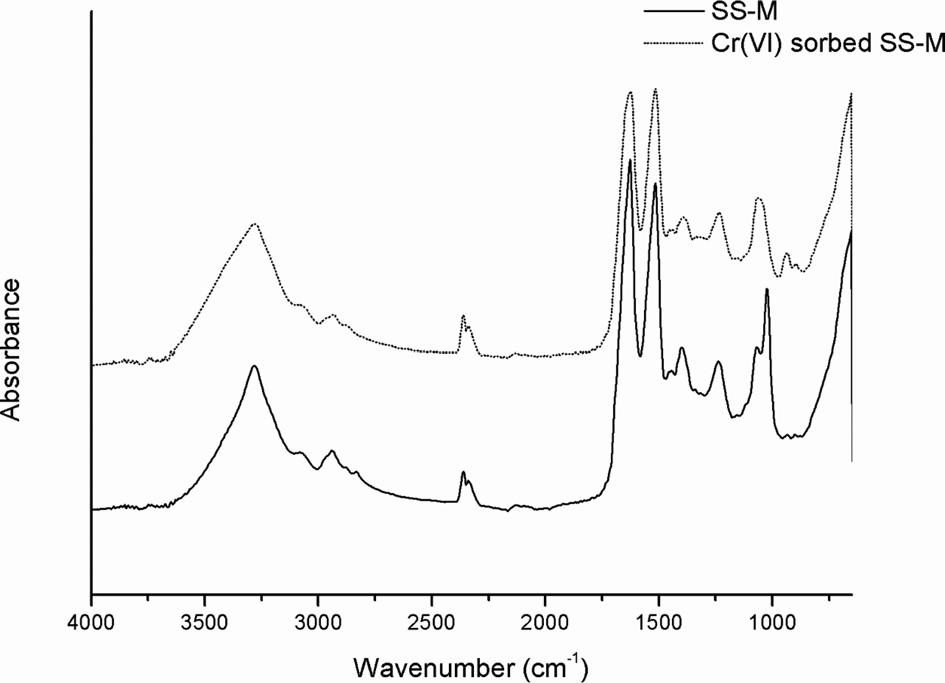
ATR-FTIR spectroscopy (Thermo Scientific, USA) was used to identify the Cr(VI) adsorbed onto the SS beads. The samples were examined within the wavenumber range 700?4000 cm-1, and 32 scans with 8 cm-1 resolution were used to obtain the spectra. The morphology and EDS spectra of the SS beads were evaluated using a field-emission scanning electron microscope (FE-SEM) (SUPRA 55VP, Carl Zeiss, Germany).
>
Evidence of Cr(VI) adsorption onto SS beads
We prepared SS beads following the same procedure reported previously by our group (Oh
also demonstrates that Cr(VI) is adsorbed on the SS bead. There have been some reports that metal ions
can induce structural deformation of proteins (Zhou
>
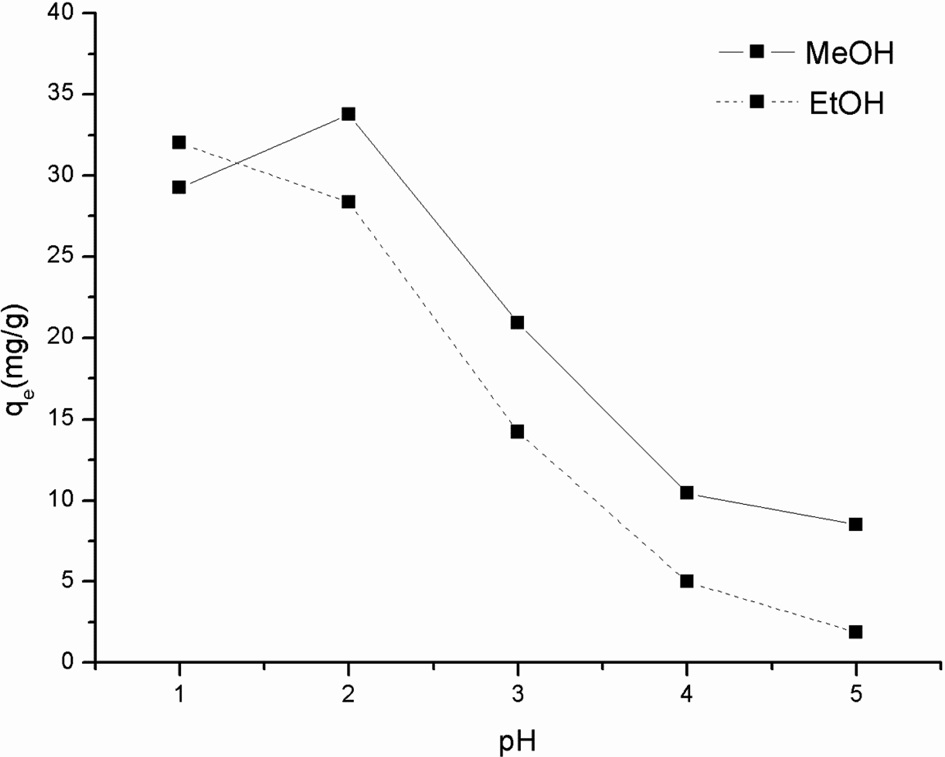
Effect of coagulant and pH on Cr(VI) adsorption
The SS beads were prepared by adding the dope solution dropwise into a coagulant. We have previously shown that both methanol and ethanol can be used as coagulants (Oh
>
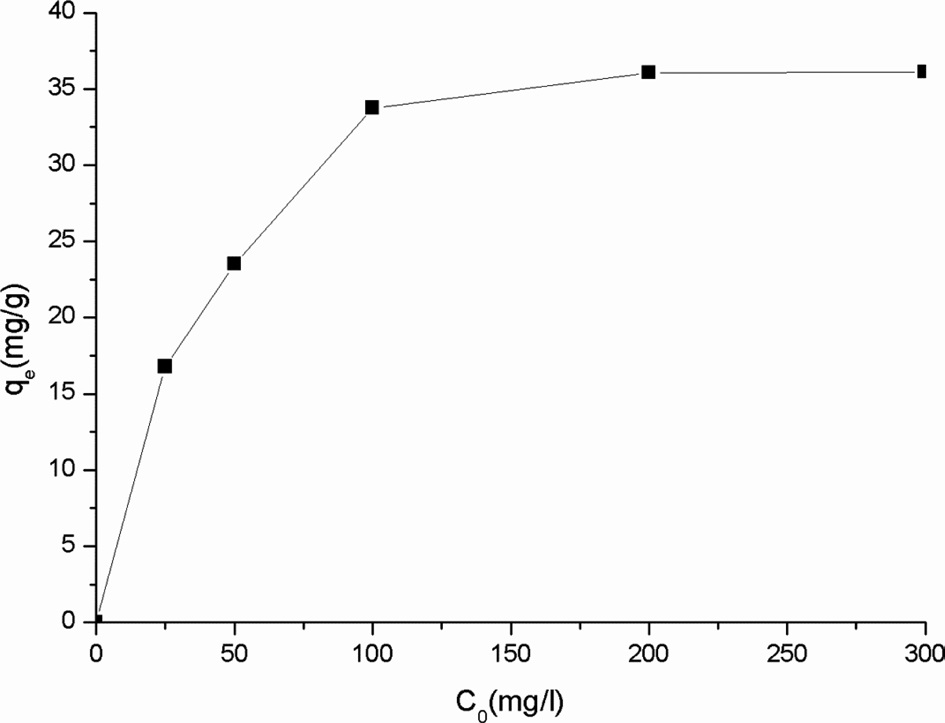
Effect of initial concentration of Cr(VI)
The initial concentration of Cr(VI),
almost linearly. The
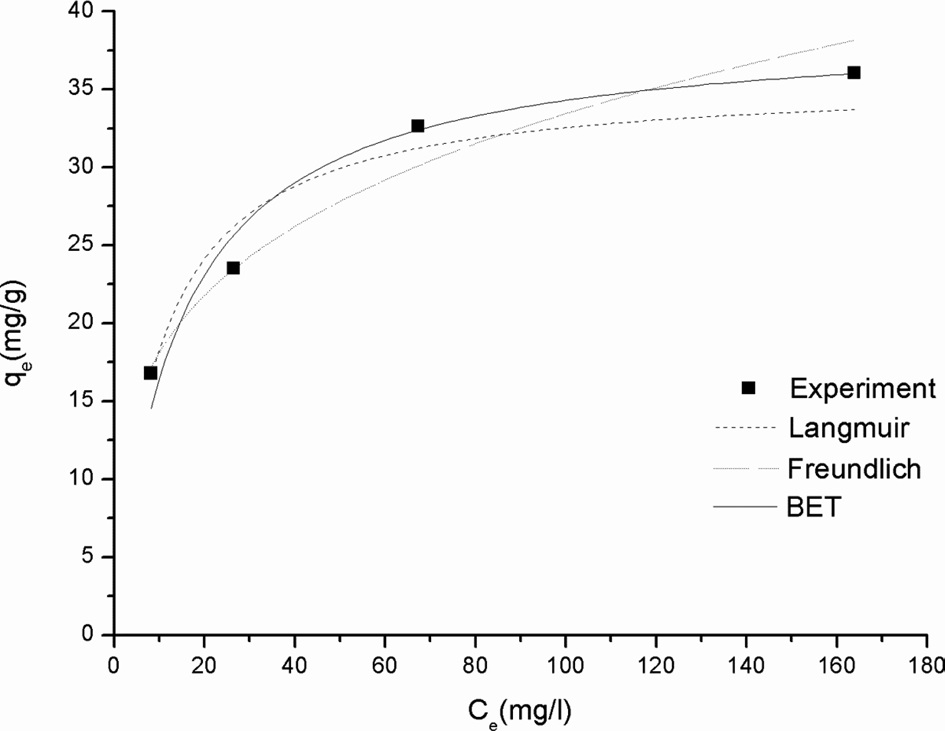
The adsorption isotherm provides useful information about what kind of interaction occurs between
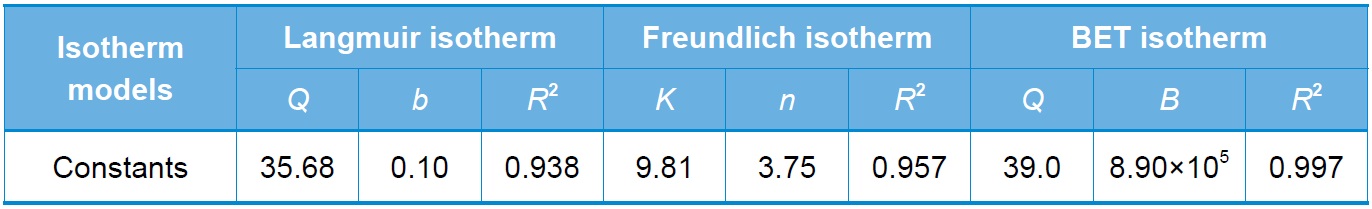
[Table 1.] Isotherm model constants
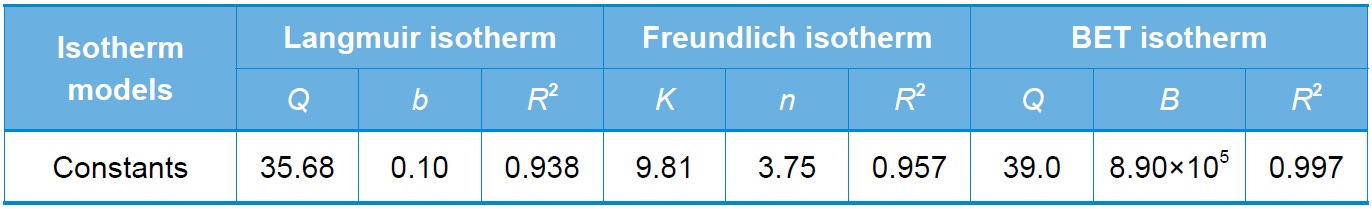
Isotherm model constants
the metal ion and the adsorbent. Fig. 5 shows the adsorption isotherm for Cr(VI) adsorption onto SSM, and Table 1 Langmuir, Freundlich, and BET model constants. It was found that the BET model fits better than the Langmuir or Freundlich model. The differences between these models are that the Langmuir model assumes monolayer adsorption while the Freundlich and BET models assume multilayer adsorption. As shown in Table 1, the Langmuir model has the least
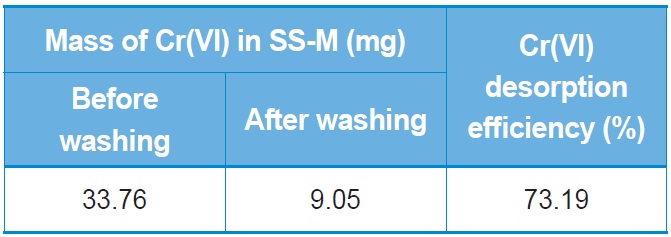
[Table 2.] Desorption efficiency for Cr(VI) from SS beads
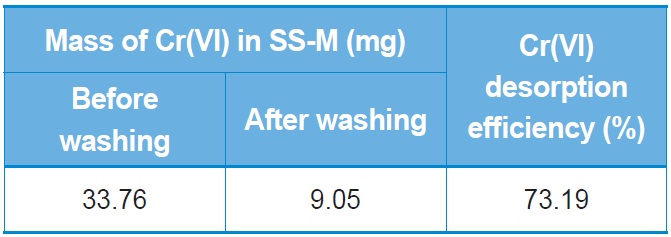
Desorption efficiency for Cr(VI) from SS beads
For reuse of the SS beads, the adsorbed Cr(VI) should be easily detached. There are many mechanisms for adsorption of Cr(VI) onto biosorbents. However, the fundamental adsorption mechanism is that the biosorbent should have positive charges in order to hold the negative chromate ions. As mentioned above, SS is a protein that has positive net charge at pH 2. If we increase the pH of the solution then the SS would have a negative net charge, resulting in desorption of the chromate ions. Therefore, we washed the Cr(VI)-adsorbed SS beads with NaOH solution. Table 2 shows the desorption results after NaOH washing. About 73.19% of the adsorbed Cr(VI) can be desorbed from the SS beads, which indicates that the SS beads can be continuously reused for Cr(VI) adsorption.