Ciliates are globally distributed in many diverse habitats, ranging from oceans to terrestrial environments, and are associated with high tropic level ecosystems (Sherr and Sherr, 1988). Recently, Foissner et al. (2008) reported that ciliate diversity may have expanded by an order of magnitude, with an estimated diversity of 27,000-40,000 free-living organisms, because of the diverse habitats that they live in and their broad ecological distribution. However, despite previous studies (Shin, 1994, 2012; Kwon, 2009), approximately 200 free-living ciliates have been reported in South Korea. The genus Anteholosticha of the Urostylidae family includes Anteholosticha petzi and approximately 40 other species (Berger, 2006; Chen et al., 2010; Shao et al., 2011) and was established by Berger (2003). Anteholosticha species are characterized by the following characteristics: the adoral zone of membranelles (AZM) is continuous; rearmost membranelles are not wider than the remaining membranelles of the proximal portion; 3 enlarged frontal cirri (FC); buccal cirrus/cirri (BC) near the right of the paroral membrane (PM); frontoterminal cirri (FTC) are present; a midventral complex composed of midventral pairs (MP) only; pretransverse ventral cirri (PTC) are present or absent; the number of transverse cirri (TC) is usually distinctly lower than the number of MP; one left (LMR) and one right marginal row (RMR); and caudal cirri are lacking (Berger, 2006).
The genus Ponturostyla of the Oxytrichidae family was transferred from Paraurostyla Borror, 1972 by Jankowski (1989), and currently only contains 1 species, P. enigmatica (tor review, see Berger, 2008). Ponturostyla enigmatica has the following characteristics: 8 FC; 5 ventral cirri VC; 5 TC; 5-9 marginal rows (MR) of cirri on each side; cortical granules in lines on the ventral side and irregularly scattered on the dorsal side; a single contractile vacuole (CV) in the midbody on the left; and 4 complete and 3-5 incomplete dorsal kineties (DK) (Song, 2001).
In the present study, we identified the 2 marine hypotrichous ciliates A. petzi and P. enigmatica from the Yellow Sea and the Korea Strait, respectively, marking the first report of the species in South Korea, and described them based on the observations of live and protargol-impregnated specimens. Furthermore, we examined the nuclear small subunit ribosomal RNA (SSU rRNA) gene sequences of the 2 species and compared them to previously annotated sequences retrieved from the GenBank.
Anteholosticha petzi was collected in January 2011 from Incheon Harbor (salinity, 29‰; temperature, 0.7℃; 37°26′N, 126°35′E) in the Yellow Sea, South Korea. Ponturostyla enigmatica was collected in June 2011 from Dongbaek Island (salinity, 34.1‰; temperature, 14.7℃; 35°09′N, 129°09′E) in the Korea Strait, South Korea. Both a sieve (1 mm) and a filter (200 μm) were used to separate the aqueous mixture of marine periphyton from floating seawater matter. The filtered water was transferred onto Petri dishes containing rice grains for the enrichment of bacterial cultures. Protargol-impregnated (Foissner, 1991) and live specimens were observed using a light microscope (DM2500; Leica, Wetzlar, Germany) at magnifications of 50 to 1,000×. Classification and terminology were performend according to Berger (1999), Song (2001), Berger (2006), and Lynn (2008).
Phylum Ciliophora Doflein, 1901
Class Spirotrichea Butschli, 1889
Subclass Stichotrichia Small and Lynn, 1985
Order Urostylida Jankowski, 1979
Family Urostylidae Butschli, 1889
Genus Anteholosticha Berger, 2003
Anteholosticha petzi Shao et al., 2011 (Table 1, Fig. 1)
Anteholosticha petzi Shao et al., 2011: 255, figs. 1-23.
Material examined. One population was collected from
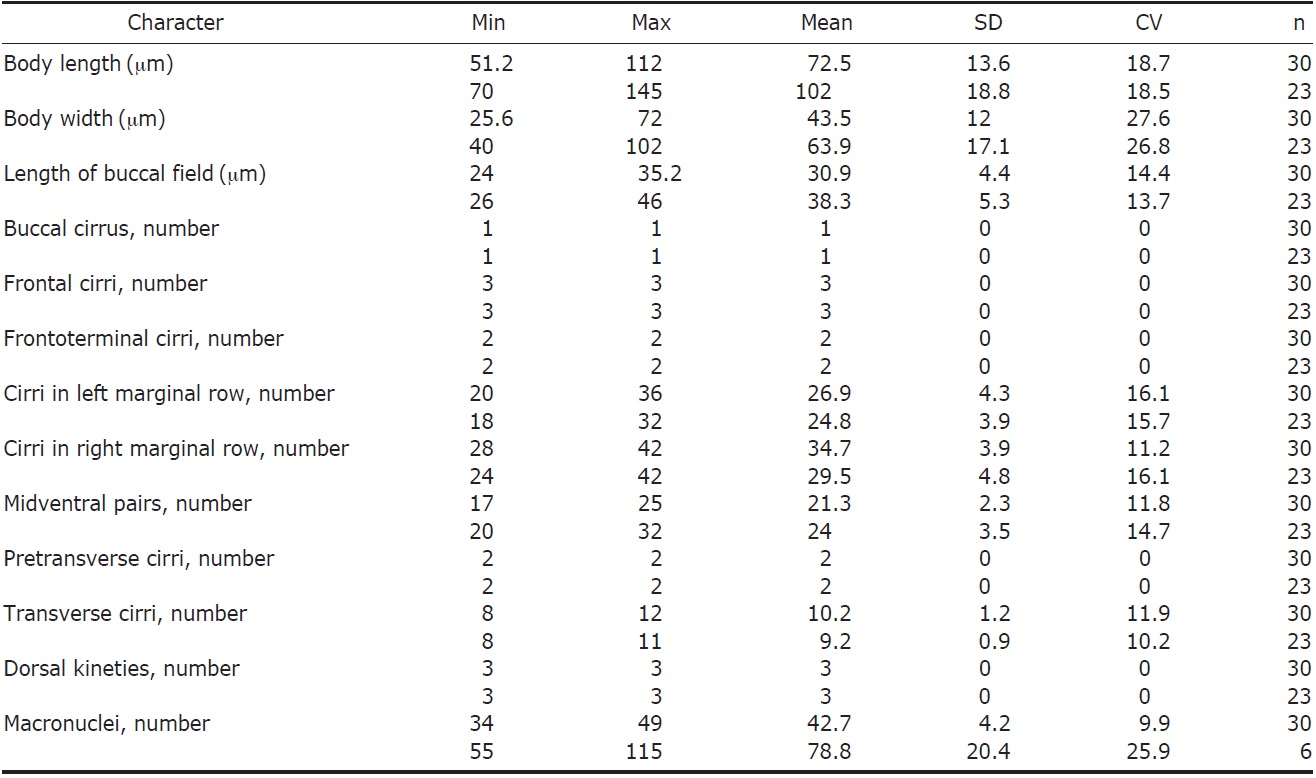
[Fig. 1.] Morphology of Anteholosticha petzi from live (A-F, K, N-P) and protargol-impregnated (G-J, L, M) specimens. A, E, F, Ventral views of live specimens; B, D, Dorsal views: D, Distribution of type I (arrow), type II (arrowhead), type III (double arrowhead) cortical granules on the dorsal side; E, Type II cortical granules around marginal row (arrowhead) and midventral row (double arrowhead); F, Type II cortical granules around transverse cirri (arrow); C, Variable outline of the body; G-J, Views of protargol-impregnated specimens: G, H, Infraciliature of the ventral (G) and dorsal (H) side; I, FC including III/2 cirrus (double arrowheads), FTC (arrowheads), and BC (arrow); J, An additional basal body (arrowhead) with 3 bipolar dorsal kineties on the dorsal side. K-P, Drawing view of A. petzi: L, M, Infraciliature of the ventral (L) and dorsal (M) side; N, Variable body shape; O, arrangement of type II cortical granules and cirri on the ventral side; P, Distribution of type I (arrow), type II (arrowhead), type III (double arrowhead) cortical granules on the dorsal side. AZM, adoral zone of membranelles; BC, buccal cirrus; DK, dorsal kineties; EM, endoral membrane; FC, frontal cirri; FTC, frontoterminal cirri; LMR, left marginal row; Ma, macronuclei; Mi, micronuclei; MP, midventral pairs; PM, paroral membrane; PTC, pretransverse cirri; RMR, right marginal row; TC, transverse cirri. Scale bars: A, B, G, H, K, L, N=50 μm.
Incheon Harbor in South Korea on January 20, 2011.
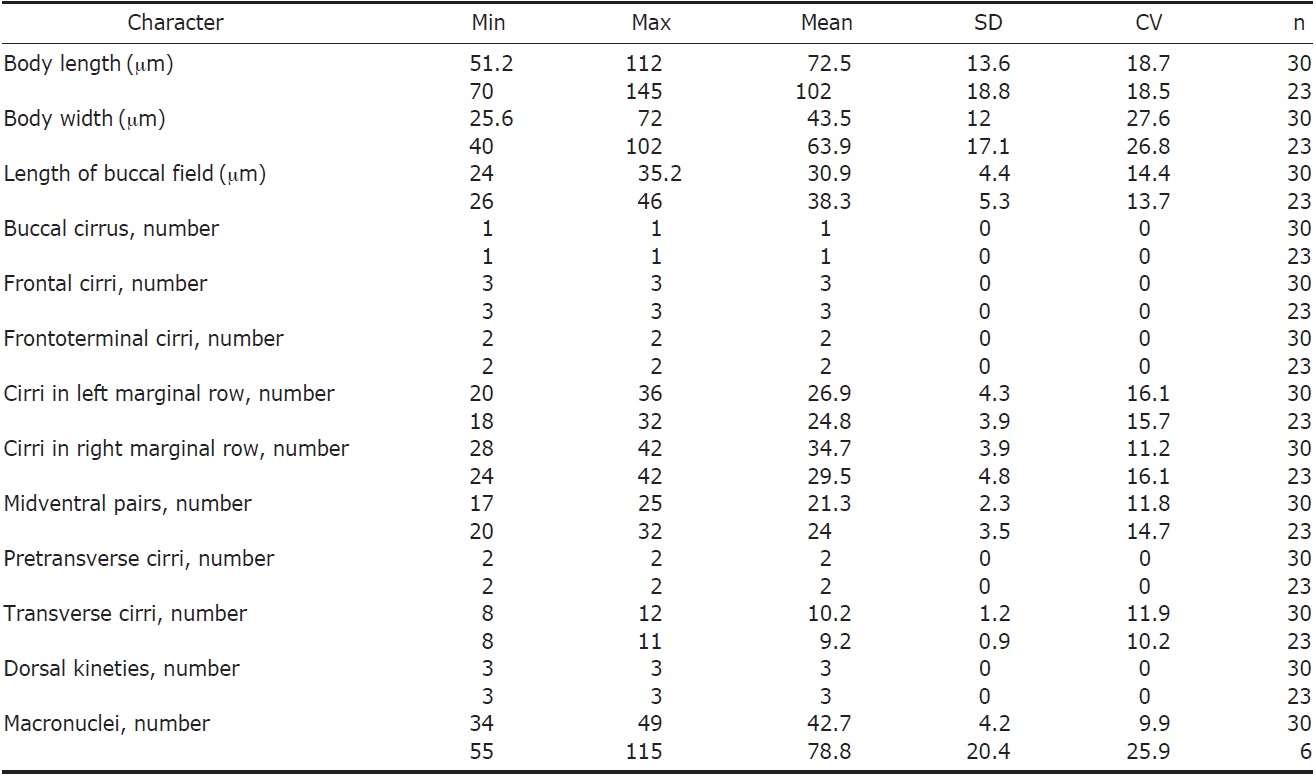
Description. Specimens have highly contractile, flexible and a variable body outline, although usually it is oval and shieldlike in shape. Cell size is typically measured around 75×43 μm in vivo (Fig. 1A, B, K-M), and CV is absent. Approximately 34-49 macronuclei (Ma) and 1-2 micronuclei (Mi) are identified (Table 1, Fig. 1H, M).
Three types of cortical granules exists: mitochondrial-like type I granules are the largest at about 4×2 μm in size and colorless on cortex of cells (Fig. 1D, P, arrow); type II granules are measured approximately 1 μm in diameter, partially gathered and weak gray in color (Fig. 1D, P, arrowhead); type III granules are measured approximately 0.5 μm in diameter, dotted on membrane of cell, and gray in color (Fig. 1D, P, double arrowhead). Further, type II cortical granules are arranged in rows along cirri on cell membrane (Fig. 1E, F, O). Cytoplasm is colorless or slightly gray (Fig. 1D).
AZM occupy about 25-40% of body length (Fig. 1A, G, K, L). Both paroral membrane (PM) and endoral membrane (EM) are either close to straight or slightly curved (Fig. 1L). There are 3 enlarged FC (Fig. 1I, double arrowheads, L), 2 FTC are close to distal end of adoral zone (Fig. 1I, arrowheads), and 1 BC is near PM (Fig. 1I, arrow, L). “Zig-zag” pattern of midventral complex is composed of 8-12 pairs (Fig. 1G, L). 2 pretransverse cirri are positioned ahead of 8- 12 TC (Fig. 1L) and 2 MR are distinctly separated at posterior end of cell body (Fig. 1K).
Three bipolar DK arrange in row on dorsal side of cell (Fig. 1H, M, arrowhead), and further, one basal body ahead of RMR(Fig. 1J, arrowhead).
Distribution. China and Korea (this study).
Remarks. Anteholosticha petzi was first reported by Shao et al. (2011) in the Jiaozhou Bay of Qingdao, China. Cell body outline and distributional pattern of cirri in A. petzi are very similar to those of A. scutellum (Berger, 2003). Anteholosticha petzi, however, has more cirri than A. scutellum as follows: MP (17-25 vs. 12-14), RMR (28-42 vs. 10-18), and LMR (20-36 vs. 11-17) (Table 1, Fig. 1L) (Chen et al., 2010). Anteholosticha warreni (Song and Wilbert, 1997) also shows similar morphology to A. petzi, however, it only contains 1 type of cortical granule and has fewer marginal cirri compared to A. petzi (22-27 vs. 20-36 in LMR; 21-26 vs. 28-42 in RMR) (Hu et al., 2000; Berger, 2006).
Korean population of A. petzi, therefore, is similar to Chinese population described by Shao et al. (2011). Korean population, however, differs from Chinese in color of cortical granules, which are not red-brown to brick-red as previously described, smaller cell size (72×43 μm vs. 102×64 μm, on average), and lower number of Ma (42 vs. 115) (Table 1).
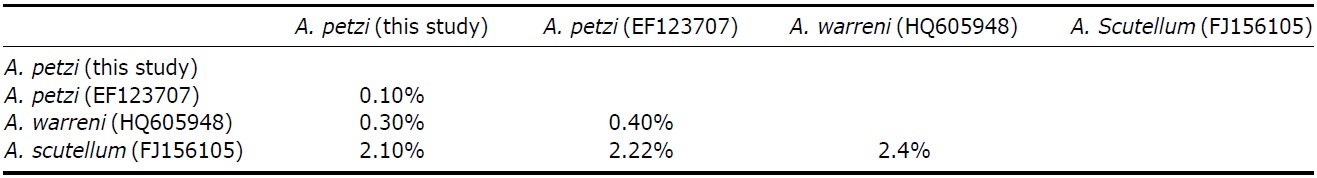
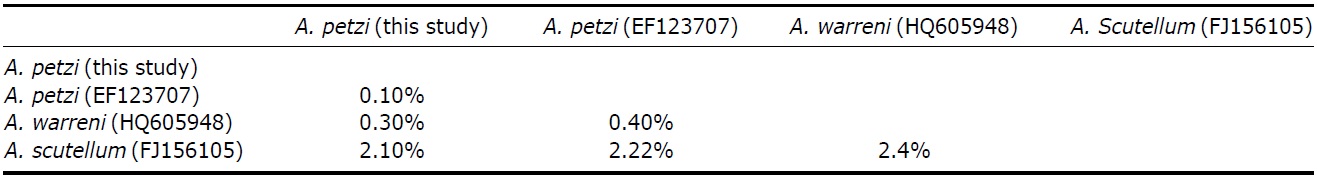
SSU rRNA gene was 1,586 base pairs in length (GenBank accession no: KC896648). Pairwise distances among related 3 Anteholosticha species were as follows: 0.1% between Korean A. petzi and Chinese A. petzi, 0.3% between Korean A. petzi and A. warreni, and 2.1% between Korean A. petzi and A. scutellum (Table 2). The pairwise distance value between Chinese A. petzi (EF123707) and Chinese A. warreni (HQ605948) (Shao et al., 2011) was also very low, 0.39%, which similar to those between Koreans. While Anteholosticha petzi and A. warreni showed clear differences in numbers of cirri and cortical granules which are important characters in the morphological taxonomy. Therefore we thought that A. petzi and A. warreni are different species like Shao (Shao et al., 2011).
Phylum Ciliophora Doflein, 1901
Class Spirotrichea Butschli, 1889
Subclass Stichotrichia Small and Lynn, 1985
Order Sporadotrichida Faure-Fremiet, 1961
Family Oxytrichidae Ehrenberg, 1830
Genus Ponturostyla Jankowski, 1989
Ponturostyla enigmatica (Dragesco & Dragesco-Kerneis 1986) Jankowski, 1989 (Table 3, Fig. 2)
Ponturostyla enigmatica Dragesco and Dragesco-Kerneis, 1986: 432.
Ponturostyla enigmatica Dragesco and Dragesco-Kerneis, 1986; Jankowski, 1989, figs. 1-10, 26-30, 33; Song, 2001: 184, figs. 1-10, 26-35.
Material examined. One population was collected from Dongbaek Island on 2 Jun 2011.
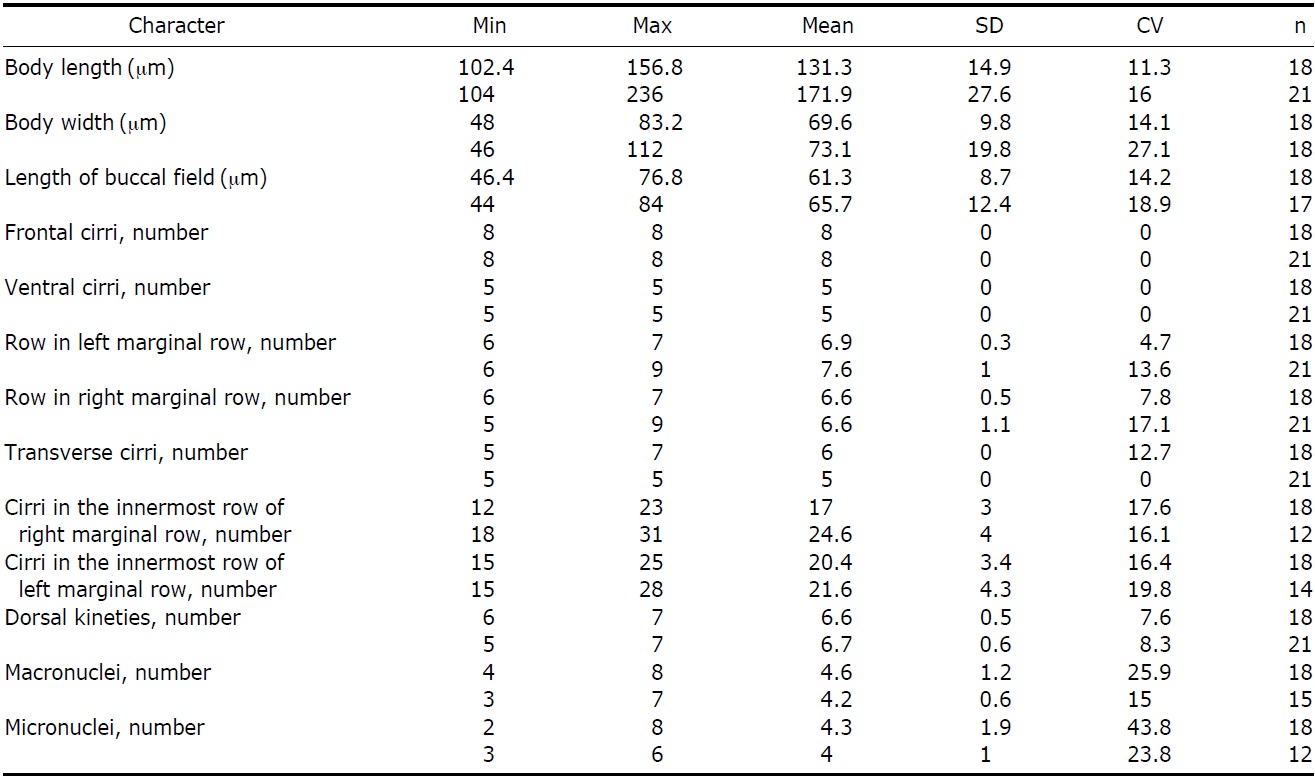
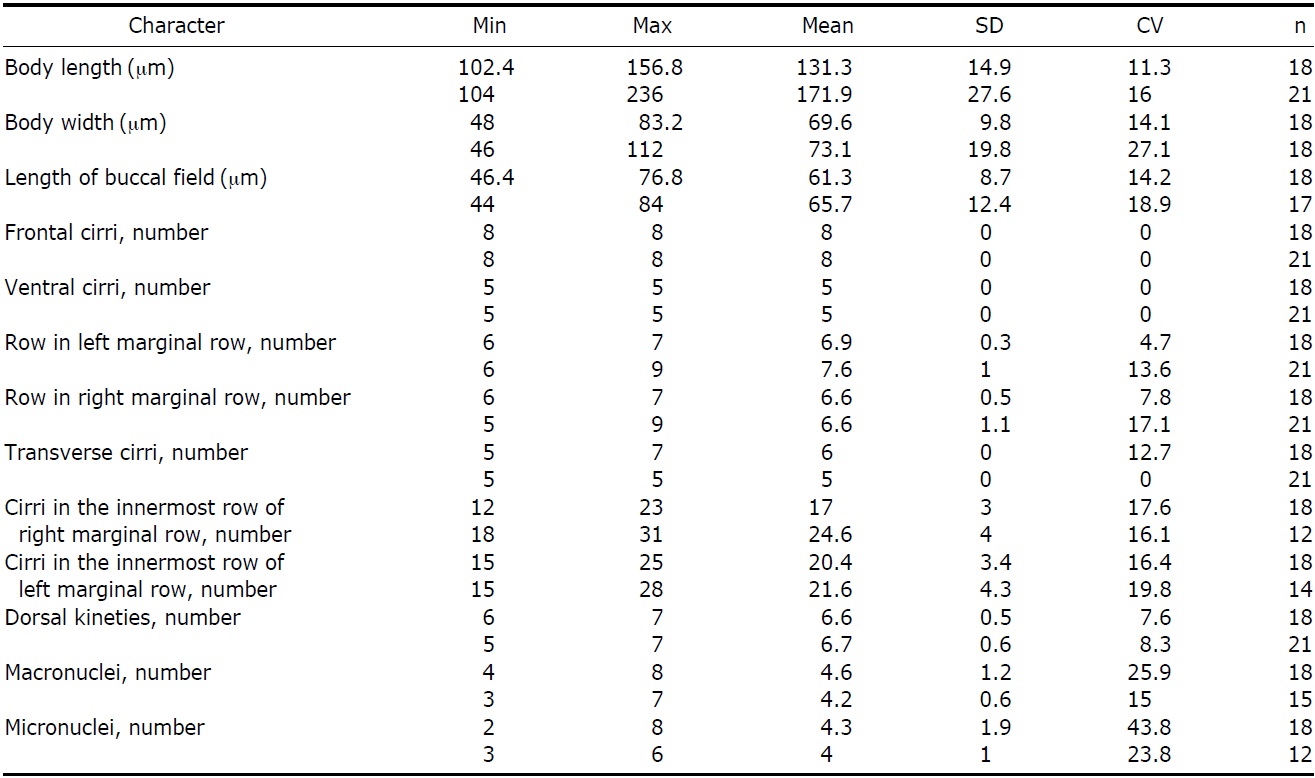
Description. Cell body; 102-156×50-75 μm in size in vivo, highly flexible, slightly contractile, and generally ellipsoid
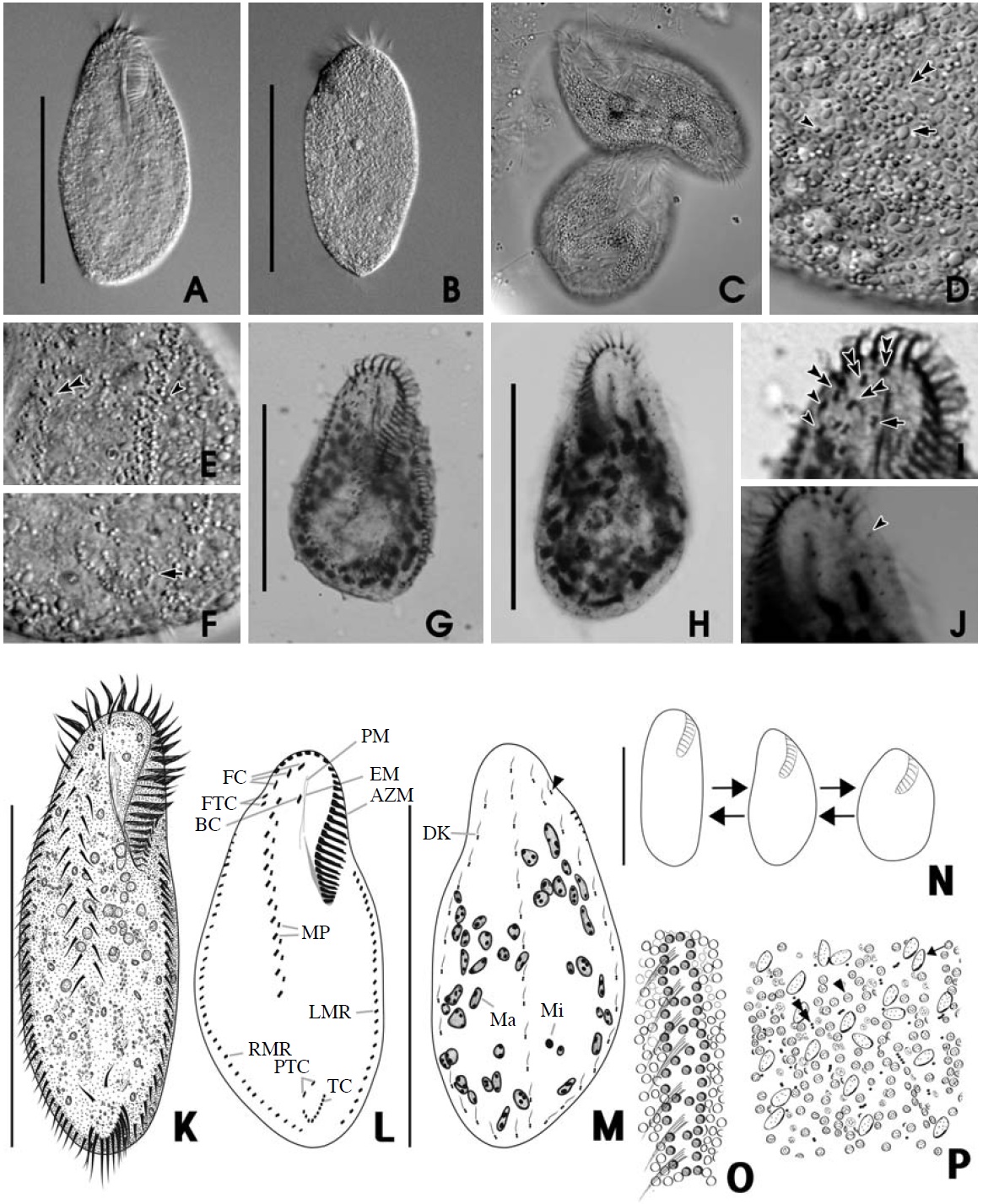
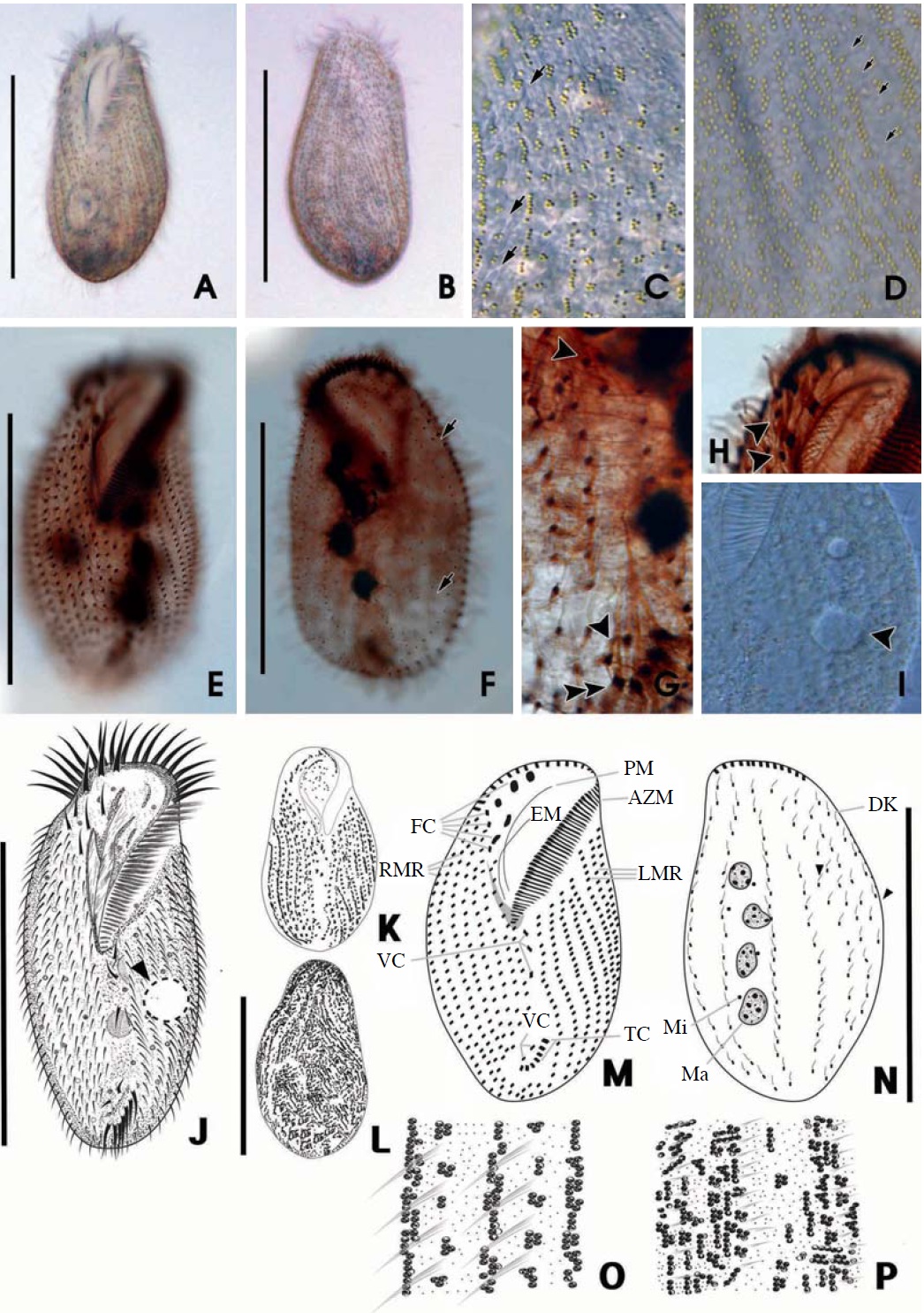
[Fig. 2.] Morphology of Ponturostyla enigmatica from live (A-D, I-L, O, P) and protargol impregnated (E-H, M, N) specimens. A, C, I, Ventral views of live specimens: C, Greenish cortical granules arranged in lines between cirri (arrows); I, Arrowhead marking the contractile vacuole. B, Dorsal view of live specimen; D, Irregular distribution of greenish cortical granules on the dorsal side; E, F, Infraciliature of the ventral (E) and dorsal (F) side: F, Arrow marking shortened dorsal kineties; G, 5 (3+2) ventral cirri (arrowheads) and 5 transverse cirri (double arrowhead); H, 8 frontal cirri (arrowhead). J-P, Drawing view of P. enigmatica: J-L, The ventral (J, K) and dorsal (L) view of live specimen; J, Arrowhead indicating the contractile vacuole; M, N, Infraciliature of the ventral (M) and dorsal (N) side; O, P, Arrangement of cirri and cortical granules of the ventral (O) and dorsal (P) side. AZM, adoral zone of membranelles; DK, dorsal kineties; EM, endoral membrane; FC, frontal cirri; LMR, left marginal row; Ma, macronuclei; Mi, micronuclei; PM, paroral membrane; RMR, right marginal row; TC, transverse cirri; VC, ventral cirri. Scale bars: A, B, E, F, J, L, N=100 μm.
shaped with a tendency to slightly narrow towards end. Both ends look like wide and round without partiality (Fig. 2A, B). CV occupy left middle of body (Fig. 2I, J, arrowhead), and 4-8 Ma 5-7×8-10 μm in size (Fig. 2E, F, N) with 2- 8 Mi approximately 2 μm in diameter (Fig. 2F, N).
Circular cortical granules are tinged with green, measured approximately 0.6 μm in diameter, regularly arranged in columns on ventral side, and irregularly distributed on dorsal side (Fig. 2A-D). MR are aligned between cortical granules on ventral side (Fig. 2C, arrows, O).
AZM was approximately 40% of body length and continuous, and relatively large of buccal lip (Fig. 2A, B, J). Both PM and EM have a long and highly curved appearance (Fig. 2M). There are 8 FC: 5 in a line along anterior of buccal field and 3 near PM (Fig. 2H, arrowheads). 5 VC are separated into anterior and posterior groups. 2 groups of VC occupy center of body and upper TC, respectively (Fig. 2G, arrowheads). LMR and RMR are distributed 6-7 each (Fig. 2J, M) and 5-7 TC are positioned near posterior end of cell body (Fig. 2G, J, M).
There are 6-7 DK composed of 4 complete kineties and 2-3 shortened kineties on dorsal side (Fig. 2F, arrows, N, arrowhead).
Distribution. Benin, China and Korea.
Remarks. Ponturostyla enigmatica was first identified in Cotonou, Benin, by Dragesco and Dragesco-Kerneis (1986). Ponturostyla enigmatica shares morphological characteristics with 2 genera, Paraurostyla and Oxytricha. The genus Paraurostyla is characterized as follows: a flexible body; a question mark-like AZM; 3-7 frontoventral rows; 6-9 TC; 1 LMR and one RMR; 5-7 DK; presence of caudal cirri (Berger, 1999). The genus Oxytricha is characterized as follows: AZM formed like a question mark; 1 LMR and 1 RMR; 4 DK; presence of caudal cirri (Berger, 1999). Ponturostyla enigmatica, however, has features that distinguishes it from genera Paraurostyla and Oxytricha which include lack of caudal cirri, 6-7 MR on each side, and 6-7 DK (Table 3, Fig. 2M, N).
Korean population of P. enigmatica conforms to the description of Chinese population provided by Song (2001). Korean population, however, differs from Chinese as follows: a smaller cell size (131×70 μm vs. 172×73 μm, on average), a lower maximum number of MR (7 vs. 9), and a higher maximum number of TC (7 vs. 5) (Table 3).
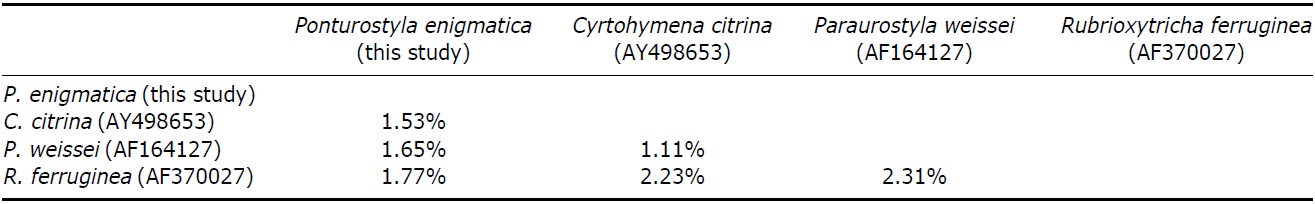
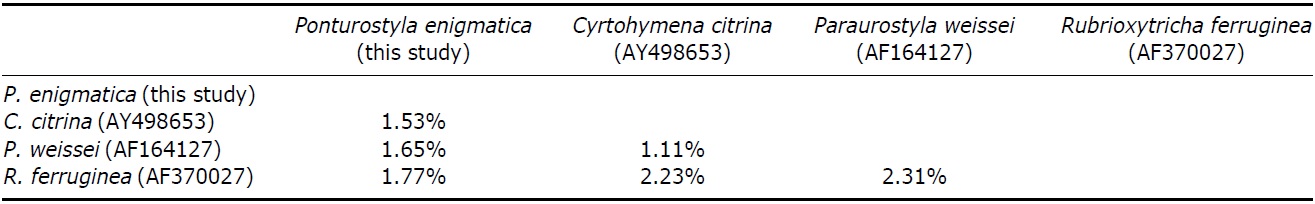
Pairwise distances between Korean P. enigmatica and similar species of family Oxytrichidae were as follows: 1.5% from Cyrtohymena citrina, 1.8% from Rubrioxytricha ferruginea, and 1.6% from Paraurostyla weissei (Table 4). This study represents first description of SSU rRNA gene sequence of P. enigmatica (GenBank accession no: KC896649), which show a close relationship with Cyrtohymena undulating membranes species (see Berger, 1999). Afrokeronopsis aurea of Neokeronopsidae, a newly established family, is closely related to Cyrtohymena undulating group (Foissner and Stoeck, 2008). To investigate their phylogenetic positions and relationship between Cyrtohymena in Oxytrichidae and Neokeronopsidae, further genetic materials should be acquired.