Aerial algae are algae that live in rock faces, earthen rocks, stone walls, and plants but not in water. Edaphophytes can also be included as aerial algae (Chung 1993). Studies on algae have mostly concentrated on aquatic algae, and studies conducted on aerial algae have mostly focused on biological pollution and preventive stone cultural heritage research (National Research Institute of Cultural Heritage 2008). Some taxa in
In research on aerial algae habitat distribution, it was determined that cyanophytes, such as
According to Hyvert (1972), 39 aerial algae species have been identified, and studies on the genus
Collections of aerial algae were sampled at Gyeonggido, Jeolla-do, Gyeongsang-do, and Chungcheong-do on 24 stoneworks from March 2009 to October 2010 (Table 1, Fig. 1). The aerial algae samples were collected from more than one part and in every aspect of the stoneworks using a soft brush and sterilized depressor. A 5 × 5 cm2 quadrat was used for quantitative analysis. Each sample was sealed and refrigerated in a light-tight container with sterilized distilled water and transferred to the laboratory (Crispim et al. 2004). Some of the samples were stored fixed in 1% formalin. Among them, the unrecorded Korean species were cultured indoors, and maintained in the algal culture collection of Kyonggi University (ACKU).
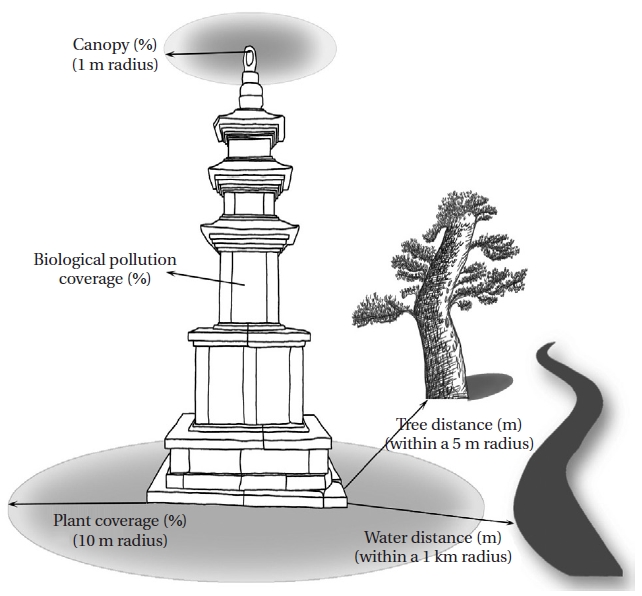
An analysis of physico-chemical environmental factors such as surface temperature of the stonework, light intensity, and humidity were conducted for all sampling dates. Temperature was measured with a stem thermometer, and surface temperature was measured with a Testo 830- T1 (Testo, Lenzkirch, Germany), humidity was measured with a Testo 625 (Testo), and light intensity was assessed with a LX-1108 instrument (Lutron, Taipei, Taiwan). Temperature, humidity, and light intensity have tremendous effects on algae distribution; however, these variables largely depend on the season and time of collection. Therefore, we measured plant coverage, canopy, distance to trees, and distance to water to identify relatively stable environmental factors that could replace temperature, humidity, and light intensity (Fig. 2).

Information on the 24 stoneworks from which algae were collected from March 2009 to October 2010
Plant coverage was considered the distribution of woods within a 10 m radius of the stoneworks as a percentage. The number of trees screening light was measured within a 1 m radius of the stoneworks as a percentage to assess the canopy. Tree distance was considered the closest distance to a tree within a 5 m radius. The water distance was the closest distance to a river or lake within a 1,000 m radius. Biological pollution coverage was calculated by measuring the bryophyte area using AutoCAD 2008 (Autodesk Inc., San Rafael, CA, USA). The taxonomic classification system was based on Hirose et al. (1977), and aerial algae were identified referring to Prescott (1973), Prescott et al. (1972, 1977, 1981, 1982), Chung (1993), John et al. (2002), and Wehr and Sheath (2003). Dominant species were analyzed by relative abundance. The collected samples were examined under
The physico-chemical and ecological environmental factors at 24 stoneworks from March 2009 to October 2010
a light microscope (400-1,000×) to determine the total number and the dominant species. Light microscopy was carried out using an Olympus BX41 microscope (Olympus, Tokyo, Japan) equipped with Nomarski differential interference optics. Species newly recorded in Korea were illustrated using a drawing attachment together with light microscope photographs. SPSS version 12.0 (SPSS Inc., Chicago, IL, USA) was used to analyze the relationships between aerial algae and the environmental factors.
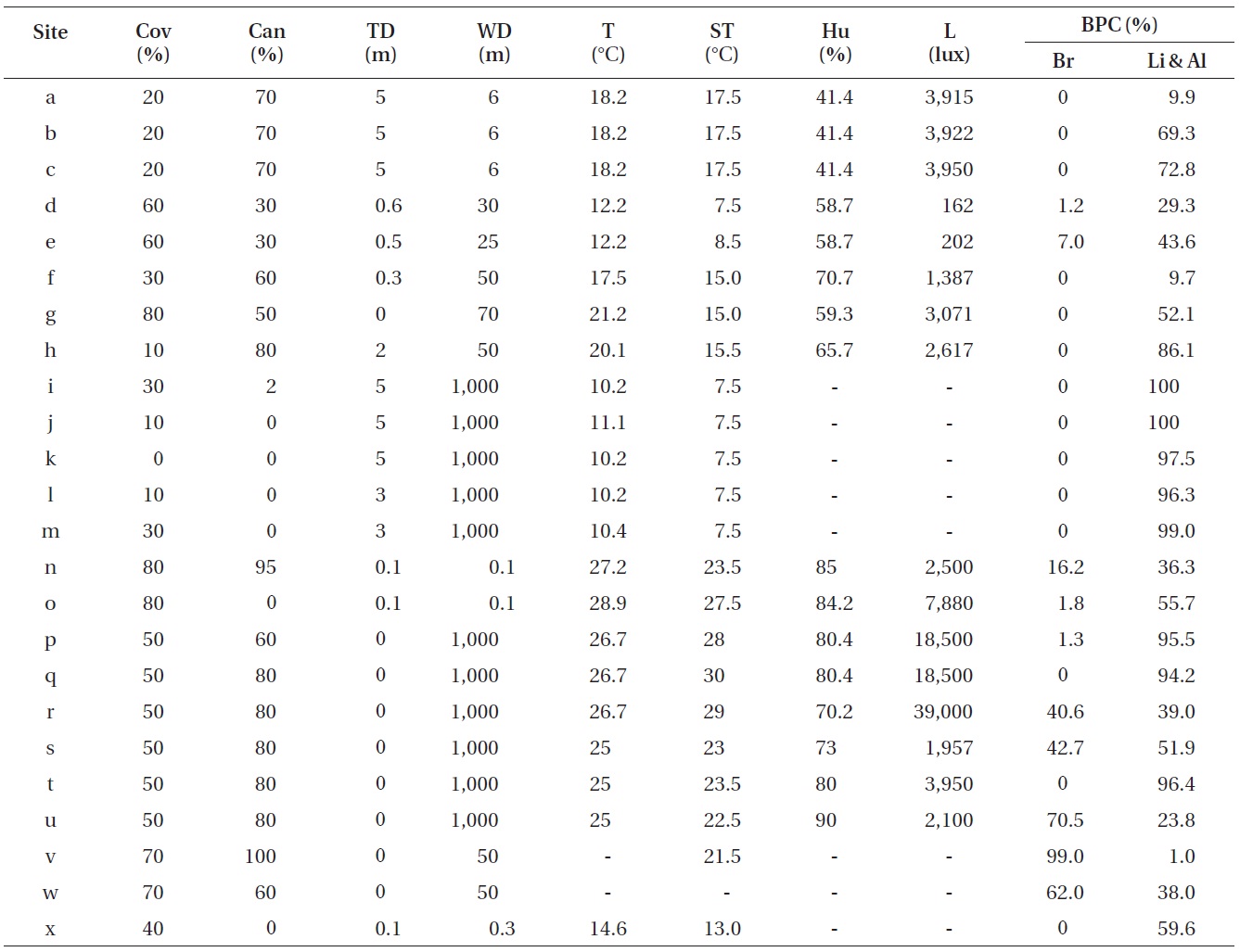
Table 2 shows the results of the physico-chemical, and ecological environmental factors of the stoneworks where the aerial algae was found. The air temperature was 10.2-29.9℃ and surface temperature was 7.5-30.0℃. Humidity was 41.4-90.0%. The light intensity values were the highest at the Yeondae small temple A-3 (39,000 lux), and the lowest were recorded at Gwang-gyo mountain 2-1 (162 lux). Due to strong wind, measuring the humidity, and light intensity at the Unjusa Temple, the stone wall at Jucksung-myeon, Danyang-gun, and the stone wall at Pyeongchang-gun was difficult. Plant coverage at the three sites including Gwang-gyo Mountain 3-2 had the highest value at 80%, whereas the Unjusa Temple A5 was not affected by plant coverage with a value of 0%. Stone wall 1 at Jucksung-myeon, Danyang-gun, which was a site where light was screened by trees, had a canopy value of 100%. Five sites, including Unjusa Temple A4, had a 0% canopy value. Tree distance ranged from 0 to 5 m, indicating that most sites were affected by trees. Water distance values were 0.1-1,000 m.
Four phyla, 4 classes, 8 orders, 4 suborders, 13 families, 23 genera, 46 species, 3 varieties, and 1 forma were collected and identified as aerial algae (Table 3). Cyanophytes were the most dominant taxa, with 3 orders, 2 suborders, 5 families, 10 genera, 26 species, 3 varieties, and 1 forma. Only one species,
Stone wall 1 at Jucksung-myeon and Danyang-gun had the most taxa among the stoneworks with 21 taxa including
The habitats of all taxa were evaluated after reviewing other studies (Table 3). Five taxa, including
Phylum Cyanophyta
Class Cyanophyceae
Order Chroococcales
Family Chroococcaceae
Synechococcus aeruginosus Nageli
Cell long and cylindrically shaped, with a width of 6-16 μm and length of 13-30 μm. Light blue-green color, with many granules; lives in freshwater and hot springs (Hirose et al. 1977).
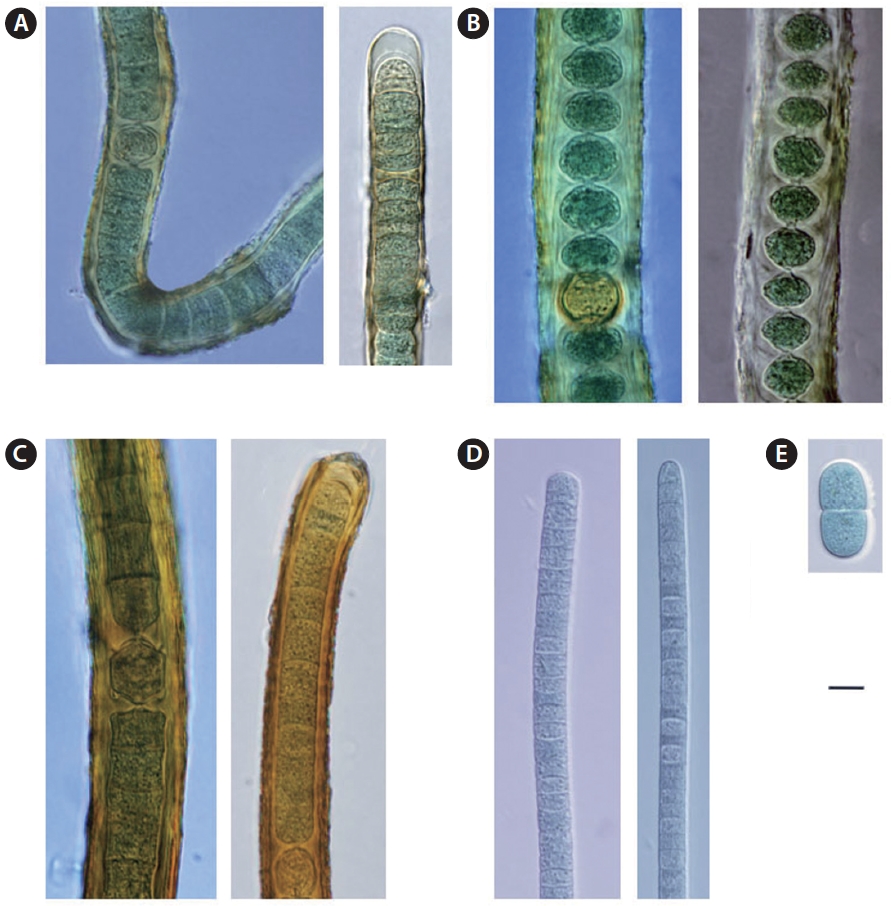
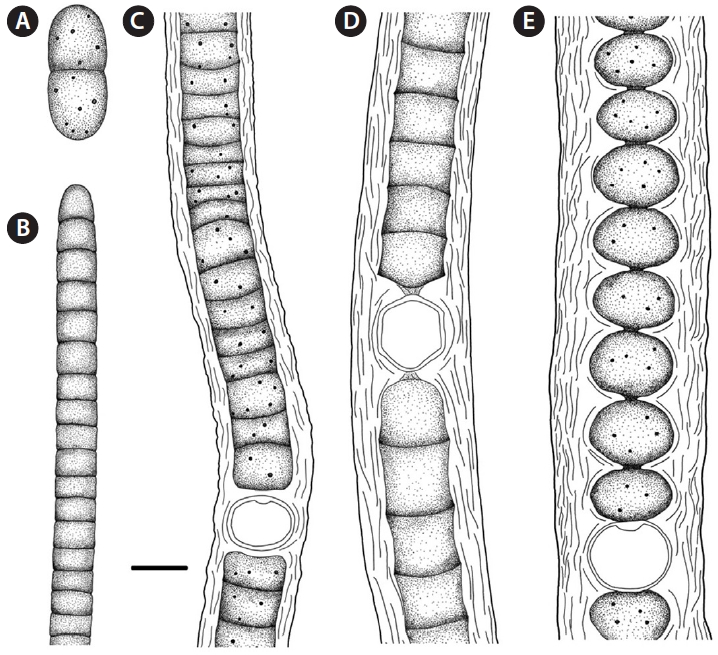
Sites of collection. t, u, v (Figs 3E & 4A).
Order Nostocales
Suborder Nostochineae
Family Scytonemataceae
Scytonema coactile Montagne var. thermalis Geitler
Cell olive or purple-green in color. Main axis pseudobranched and filamentous. Sheaths have no layered structure or color. Cell is (11)-15-18 μm in diameter and becomes narrower at the tip. Trichome is cylindrical with width of 9-11 μm and length of 7-20 μm. Heterocysts are cylindrical, and it inhabits hot springs (Hirose et al. 1977).
Sites of collection. k, p, r, s, u (Figs 3A & 4C).
Order Nostocales
Suborder Nostochineae
Family Scytonemataceae
Scytonema coactile Montagne var. minor Wille
Cell is thick and 1 mm in width. Pseudo-branching mostly originates from main axis. It has a yellow-brown colored sheath without a layered structure, and a diameter of 14-16 μm. Trichomes are cylindrical with no joints; width is 8-10 μm. Heterocysts are cylindrical or square shaped, and it inhabits hot springs (Hirose et al. 1977).

A list of aerial algae and their habitats collected at 24 stoneworks from March 2009 to October 2010

Continued

Continued
Sites of collection. a, b, e, i, k, l, m, o, v, w (Figs 3C & 4D).
Order Stigonematales
Family Stigonemataceae
Stigonema ocellatum (Dillwyne) Thuret f. ocellatum
Cell shaped like grass or a thread; 25-50 μm wide with irregular branching. Usually exists as a single filament, but occasionally can be found as double filaments. Branches are slightly slimmer than the main axis. Sheath is relatively thick, and may have yellow, yellow-brown, or no color. Trichomes are elliptical or square-shaped, and it inhabits humid dirt, rocks, and grass (Hirose et al. 1977).
Sites of collection. c, e, g, h, I, j, l, m, r, s, t, u, v, w (Figs 3B & 4E).
Order Nostocales
Suborder Oscillatoriineae
Family Oscillatoriaceae
Oscillatoria boryana Kutzing
Cell dark colored; trichomes are flexible in the shape of a wave. The cell continuously folds, and length is less than or equal to half the width. Width of the cell is 6-8 μm, and length is 4-6 μm. The apical cell is circular or a sharp circular cone, and has no calyptra (Hirose et al. 1977).
Site of collection. r (Figs 3D & 4B).
Cyanophytes were dominant at 22 sites among the aerial algae (Table 4). In particular, cyanophytes dominated by >50% at 15 sites, including Gwang-gyo mountain 1-1 (Kim et al. 2010).
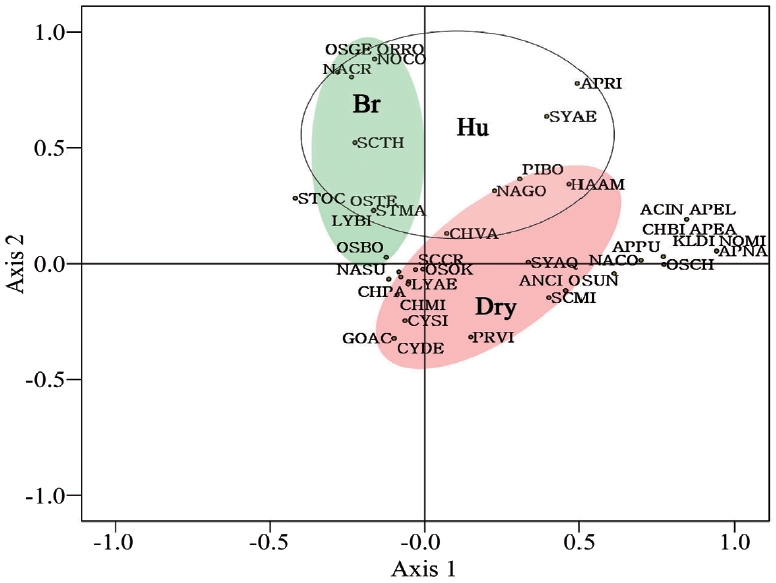
A principal component analysis was conducted to identify the relationships between the aerial algae and ecological factors. The varimax method (Kim et al. 2007) was used to obtain the results (Fig. 5).
All species were divided into ecological groups according to the correlation coefficients. The identified species were divided into biological pollution coverage of bryophyte (Br), high humidity (Hu), and dry (Dry) groups with
[Table 4.] Dominant species at 24 stoneworks in Korea during March 2009 to October 2010

Dominant species at 24 stoneworks in Korea during March 2009 to October 2010
a negative correlation with humidity and canopy (Fig. 5). Species in the Br group were cyanophytes and bacillariophytes such as
Physico-chemical environmental factors vary greatly due to seasonal differences and the weather conditions on the date of collection. This seems to affect changes in aerial algae groups inhabiting stoneworks (Lim and Lee 2008a). Korean aerial algae now include total 108 taxa by adding 5 taxa from the present study. We found the following ten species which are aquatic species found in aerial conditions:
Analysis of the correlations between environmental factors in the 24 stoneworks in Korea collected during March 2009 to October 2010
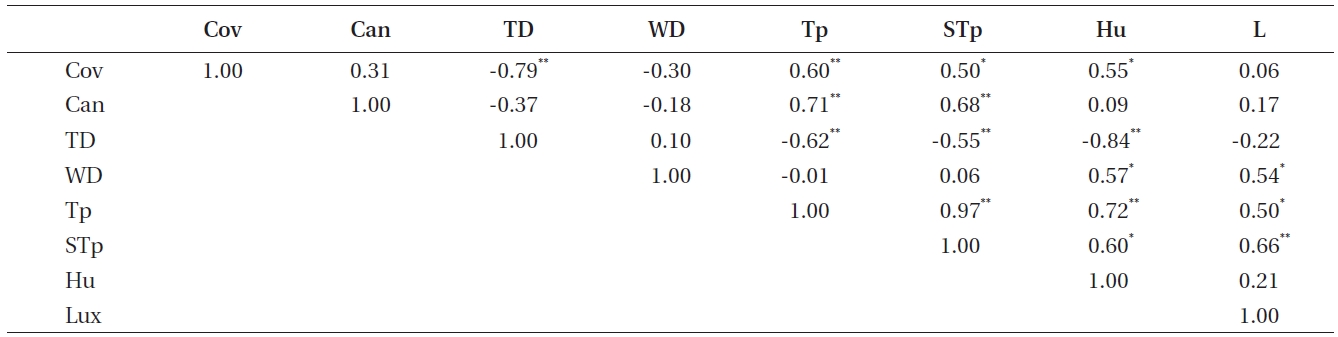
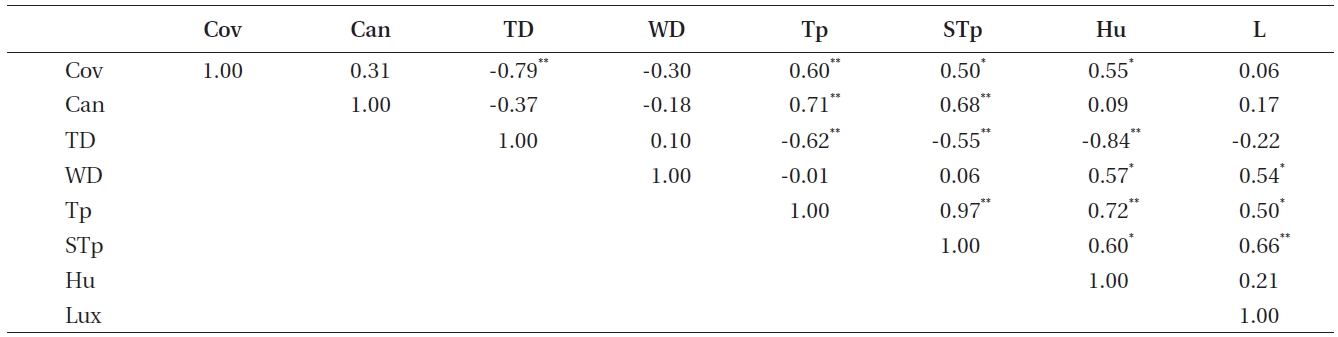
Correlations among other environmental factors were tested to identify other environmental factors that could substitute for humidity and light intensity (Table 5). As a result, tree distance, water distance, and plant coverage were replaceable environmental factors.