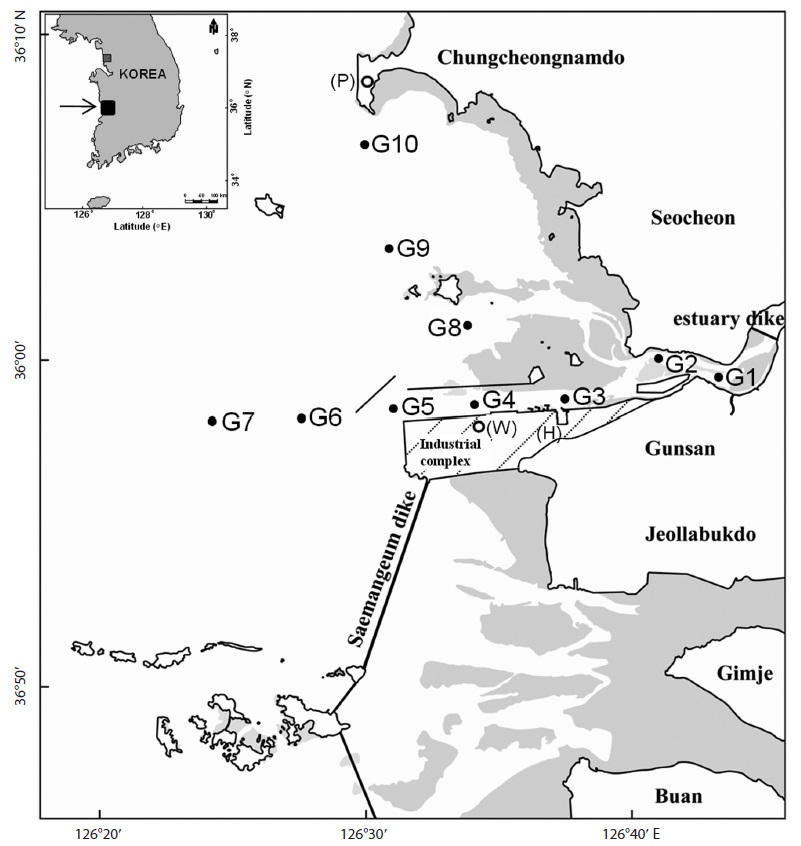
Gunsan, located in the western coast of Korea, is characterized by muddy flat with wide tidal ranges. Fresh water flows into this coast mainly from the Kum River at the eastern end of the coast. The inner part of the coast contains industrial complexes, free trade zones, and international ports. In particular, Gunsan-Gunjang industrial complex was initiated in 1989. It includes motor, ship, and construction machinery industries, and has played a central role in the economies of the Jeonbuk and Gunsan areas, with production and export amounts for 2009 worth USD 2.3 billion, and accounting for 59% of the total production and 63.2% of the total exports of Gunsan City (Korea Industrial Complex Corporation, 2011). However, the rapid industrialization of this area has resulted in substantial environmental deterioration that may lead to a variety of social and health problems. Only a few studies have been reported on environmental pollution levels in the Gunsan coast (Kwon et al., 2000; Kim and Jeong, 2003; Seo and Park, 2007), and those studies focused on eutrophication and levels of trace metals. To our knowledge, the present study is the first to investigate concentrations of anthropogenic organic chemicals and to perform sediment toxicity testing in the marine environment of Gunsan.
In this study, the target compounds included tracers of sewage contamination and toxic chemicals; fecal sterols, nonylphenol (NP), polycyclic aromatic hydrocarbons (PAHs), and butyltins (BTs). Coprostanol (COP), a fecal sterol, has been widely used as a tracer of fecal and sewage contamination in aquatic environments because of its high concentrations in human
feces (Chan et al., 1998). NP is a biodegradation product of nonionic surfactants widely used in a various industrial processes, and residential and commercial cleaning products. NP also acts as an endocrine disrupter and is frequently found in effluents from industrial complexes (Bennie et al., 1997; Li et al., 2004; Choi et al., 2011a). COP and NP have been regularly used to investigate the degree of contamination of sediments from wastewater (Noblet et al., 2004; Diez et al., 2006; Lara- Martin et al., 2008; Choi et al., 2011a, 2011b).
PAHs are ubiquitous environmental pollutants that primarily result from anthropogenic activities including incomplete fuel combustion, domestic and industrial wastewaters, and the spillage of crude oil and its refined products (Hites et al., 1997; Pettersen et al., 1997). Because PAHs are toxic, carcinogenic, and mutagenic to all organisms including humans, they have been extensively studied in various environmental and biological compartments (Yim et al., 2005; Moon et al., 2007, 2010). BTs are one of the most hazardous pollutants known to occur in aquatic ecosystems. BTs such as tributyltin (TBT) have been widely used as an antifouling paint component on fishing boats, large ships and vessels, harbor structures, and marine platforms. In Korea, TBT in antifouling paints was banned in 2000 because of its adverse biological effects. However, BTs are still detected in aquatic ecosystems and high BT concentrations have been found in estuaries and harbors (Leung et al., 2006; Berto et al., 2007; Choi et al., 2011b).
The objective of this study was to determine the status of sediment contamination from anthropogenic activities by fecal sterols, NP, PAHs, and BTs. We performed the bioluminescence toxicity test using the bacteria
Surface sediments were sampled at 10 locations (G1-G10) in the Gunsan coast in December, 2008 (Fig. 1). Sediment samples were collected systematically from the inner to the outer parts of the coast following the procedure of the National Marine Environmental Monitoring Program in Korea. Sediments samples (0-4 cm depth) were collected by a van Veen Grab sampler, to provide a representative coverage of the coast. The collected samples were individually wrapped in aluminum foil and immediately frozen in a refrigerator on the vessel. Marine sediment samples were transported to the laboratory and were stored at -20℃ and freeze dried later.
The detailed descriptions of sediment sample extraction and cleanup procedures for fecal sterols, NP, PAHs, and BTs have been reported previously (Moon et al., 2007; Choi et al., 2010b, 2011a). For NP and fecal sterols, the sediment samples of 5 g were placed in a 50 mL Teflon centrifuge tube with a Teflon cap and then digested by mechanical shaking, followed by the spiking of surrogate internal standards for NP ( 13C6-NP, CLM-4306; Cambridge Isotope Laboratories, Andover, MA, USA) and fecal sterols (1-nonadecanol; Dr. Theodor Schuchardt & Co., Hohenbrum, Germany). The solutions used to extract the NP and fecal sterols were 20 mL of 50% methylene chloride (ultra residue analysis; J. T. Baker, Phillipsburg, NJ, USA) in chloroform (ultra residue analysis; J. T. Baker). In order to purify the NP, the extracts were first derivatized using 99% bis(trimethylsilyl) trifluoroacetamide with 1% trimethylchlorosilane (BSTFA; Sigma-Aldrich, St. Louis, MO, USA) and then cleaned by passing through a florisil column with 7 mL of hexane (ultra residue analysis; J. T. Baker). The extracts to be used for the analysis of fecal sterols were cleaned by passing through a florisil column, with successive elutions with 60 mL of 40% hexane in chloroform and 40 mL of 20% methanol (ultra residue analysis; J. T. Baker) in chloroform. The second fraction was concentrated and derivatized using BSTFA, then used for the analysis of the fecal sterols.
Briefly, sediment samples of 5 g were extracted in a Soxhlet apparatus using 200 mL of 25% hexane in methylene chloride for 24 h after spiking with 7 species of internal standards (d8- naphthalene, d8-acenaphthylene, d12-fluoranthene, d10-phenanthrene, d10-pyrene, d12-benzo[a]pyrene and d12-benzo[g,h,i] perylene; ES-2044; Cambridge Isotope Laboratories). Both extracts were cleaned by passage through activated silica gel (neutral, 7734, 70-230 mesh; Merck, Darmstadt, Germany) column with successive eluants of hexane and 15% methylene chloride in hexane. The eluants were concentrated to approximately 50 μL and were dissolved with 50 μL n-nonane (pesticide analysis grade; Fluka, St. Gallen, Switzerland) for instrumental analysis.
For BTs analysis, 3 g samples were extracted twice by mechanical shaking with 0.1% tropolone (Merck, Hohenbrunn, Germany)-methylene chloride, and 50% HCl (Merck, Darmstadt)-methanol in 50 mL Teflon tubes. Tripentyltin chloride (Kanto, Tokyo, Japan) was spiked, as a surrogate standard, before extraction. The extract was hexylated with a Grignard reagent, N-hexylmagnesium bromide (Tokyo Chemical Industry, Tokyo, Japan). The remaining Grignard reagent was removed with 1 N H2SO4 (Merck, Hohenbrunn). The organic fraction was decanted, concentrated with a Turbo vap LV (Caliper Life Science Inc., Hopkinton, MA, USA), and then cleaned by passage through a florisil (60-100 mesh, reagent grade; Sigma-Aldrich, Milwaukee, WI, USA) column chromatograph. Finally, tetrabutyltin (Sigma-Aldrich) was added to the concentrated eluants as a recovery standard.
The detailed conditions of instrumental analysis for fecal sterols, NP, PAHs, and BTs are presented elsewhere (Moon et al., 2007; Choi et al., 2010b, 2011a). In this study, we selected the following 16 non-alkylated PAHs that are listed as priority pollutants by the United Stated Environmental Protection Agency (US EPA): naphthalene (NaP), acenaphthylene (AcPy), acenaphthene (AcP), fluorene (Flu), phenanthrene (PhA), anthracene (AnT), fluoranthene (FluA), pyrene (Pyr), benzo[a]anthracene (BaA), chrysene (Chr), benzo[b]fluoranthene (BbF), benzo[k]fluoranthene (BkF), benzo[a]pyrene (BaP), indeno[1,2,3-c,d] pyrene (InP), dibenzo[a,h]anthracene (DbA), and benzo[g,h,i]perylene (BghiP). The BTs including TBT, dibutyltin (DBT), and monobutyltin (MBT), and the fecal sterols including COP, cholesterol (CHOE), and cholestanol (CHOA), were analyzed. Identification and quantification for target compounds were carried out using a gas chromatograph (Agilent 6890; Agilent Technologies, Inc., Wilmington, DE, USA) coupled with a mass spectrometer (Agilent 5973N). The capillary column used was a DB-5MS (30 m length, 0.25 mm inner diameter, 0.25 μm; J&W Scientific, Folsom, CA, USA). The mass spectrometer was operated under the selected ion monitoring mode using molecular ions of PAHs, BTs, NP, and fecal sterols.
All of the surrogate standards were detected with no interferences. The recovery of surrogates for sediment samples was 90 ± 6% for fecal sterols, 84 ± 8% for NP, 85 ± 23% for PAHs, and 84 ± 2% for BTs respectively. Solvents injected before and after the injection of standards showed negligible contamination or carryover. Procedural blanks of sediments were processed in the same way as the samples. Blanks did not contain quantifiable amounts of the target compounds. The calculated detection limit (S/N = 3) was 1 ng/g dry wt for individual PAHs and NP, 0.2 ng Sn/g dry wt for BTs, and 4-5 ng/g dry wt for fecal sterols, respectively.
The total organic carbon (TOC) content in the sediments was determined using a CHN Elemental Analyzer (Model 2400; PerkinElmer, Boston, MA, USA) after the removal of calcium carbonate with 1 N HCl (Choi et al., 2007).
>
Bacterial bioluminescence test by Vibrio fischeri
Bioluminescence testing was conducted on organic solvent extracts of sediments, based on the standard method of Korea’s Ministry of Land, Transport and Maritime Affairs (MLTM, 2010) with some modifications. Detailed descriptions of sediment sample preparation procedures for the bioluminescence testing have been reported previously (Choi et al., 2010b, 2010c). Briefly, 1-2 g of sediment was placed in a 50 mL Teflon centrifuge tube with a Teflon cap. The sample was extracted twice by mechanical shaking for 1 h with 20 mL of 50% dichloromethane in chloroform, and was centrifuged and then filtrated to minimize the effects of suspended particles. Solvent exchanges to the less-toxic dimethyl sulfoxide (ACS reagent; Sigma-Aldrich) were performed. Testing was conducted with an N-TOX system (Model 200; Neoenbiz Inc., Bucheon, Korea) using the organic solvent solubilization method. The determination of toxicity was performed following 30-min contact in all tests.
Potassium dichromate and 3,5-dichlorophenol solutions were purchased from Neoenbiz Inc. for use as reference chemicals. They were run for every fresh vial of bacteria to ensure the validity of all tests. Lyophilised
Spearman correlation analyses were performed to investigate the relationships among chemical concentrations and
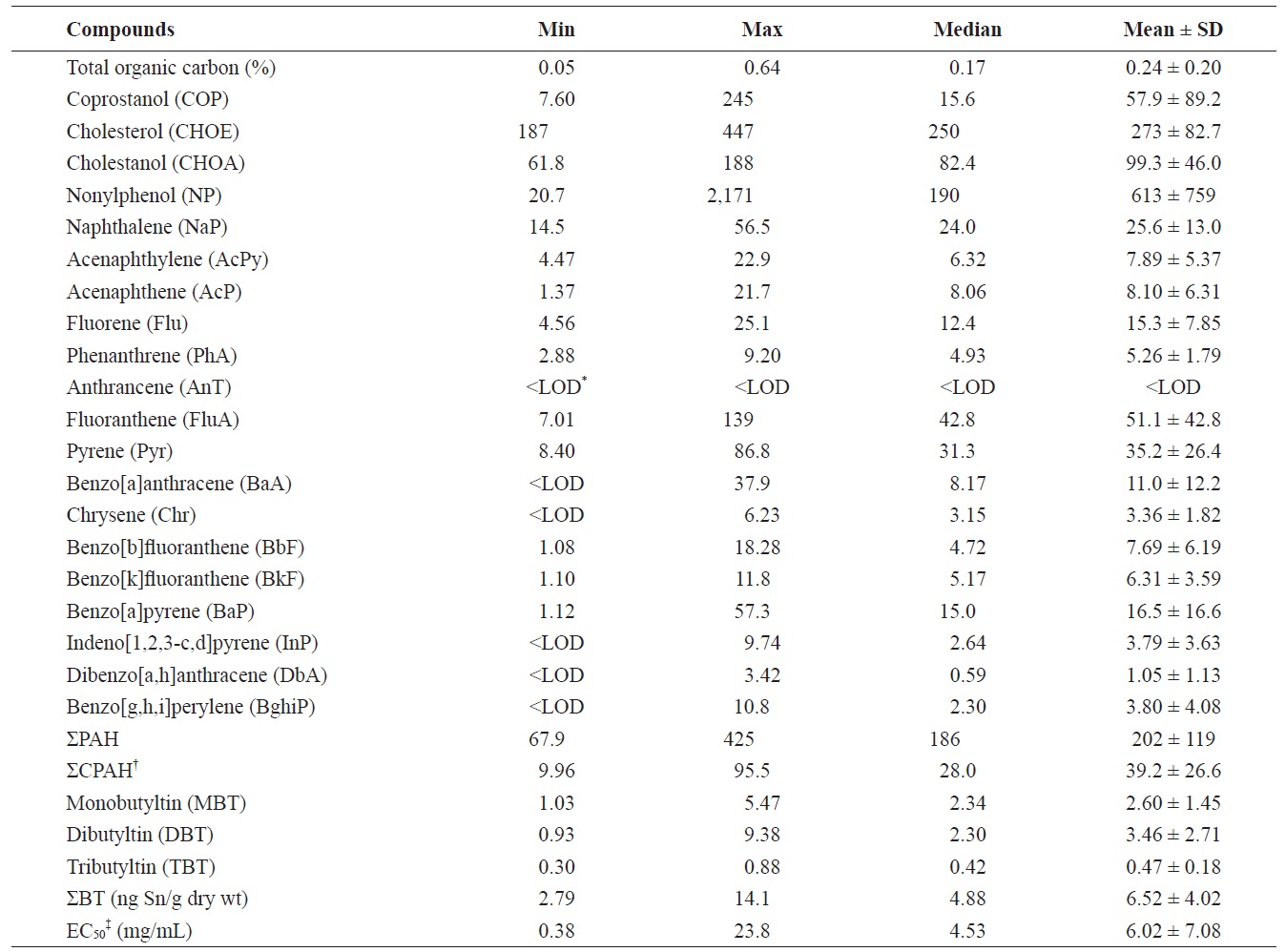
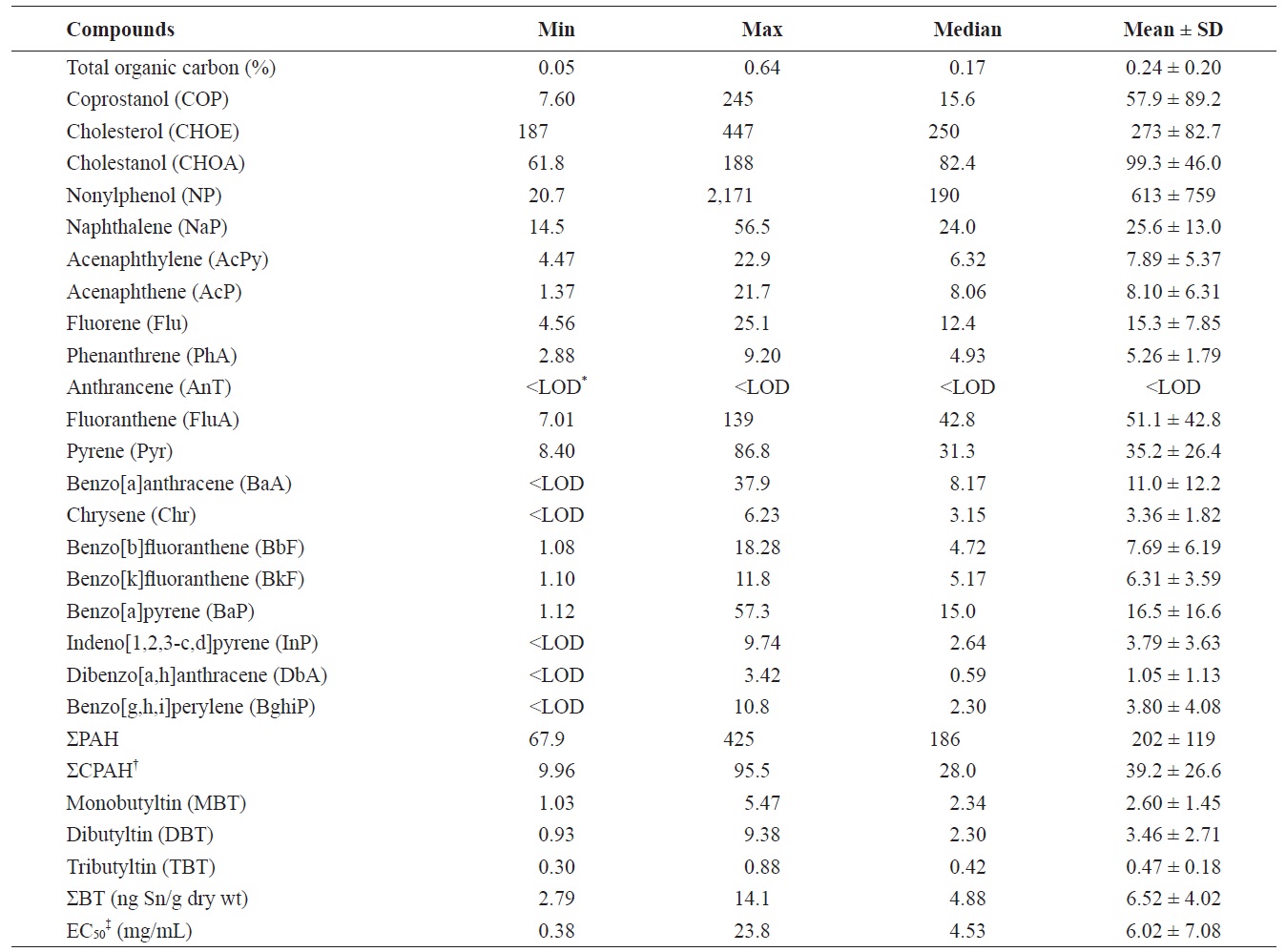
Summary of organic chemical concentrations (ng/g dry wt) and toxicity (mg/mL) in sediments from Gunsan, Korea
the toxicity using SPSS version 10.0 (SPSS Inc., Chicago, IL, USA). Non-parametric multidimensional scaling (MDS) was performed to characterize the spatial variability of chemical distributions and sediment toxicity at each of the sampling locations (Fletcher et al., 2001; Moon et al., 2008; Kim et al., 2010b). Bray-Curtis similarities were calculated from squareroot transformed data using PRIMER software for Windows (PRIMER version 5.2.9; Plymouth Marine Laboratories, Plymouth, UK).
Concentrations of TOC, fecal sterols, NP, ΣPAH (the sum of 16 PAH compounds) and ΣBT (the sum of MBT, DBT, and TBT) in surface sediment samples are summarized in Table 1. The TOC in Gunsan marine sediments ranged from 0.05
to 0.64% with a mean value of 0.24%. There are no locations with a TOC content > 3.5%, a level which can pose a high risk for impaired benthic assemblages (
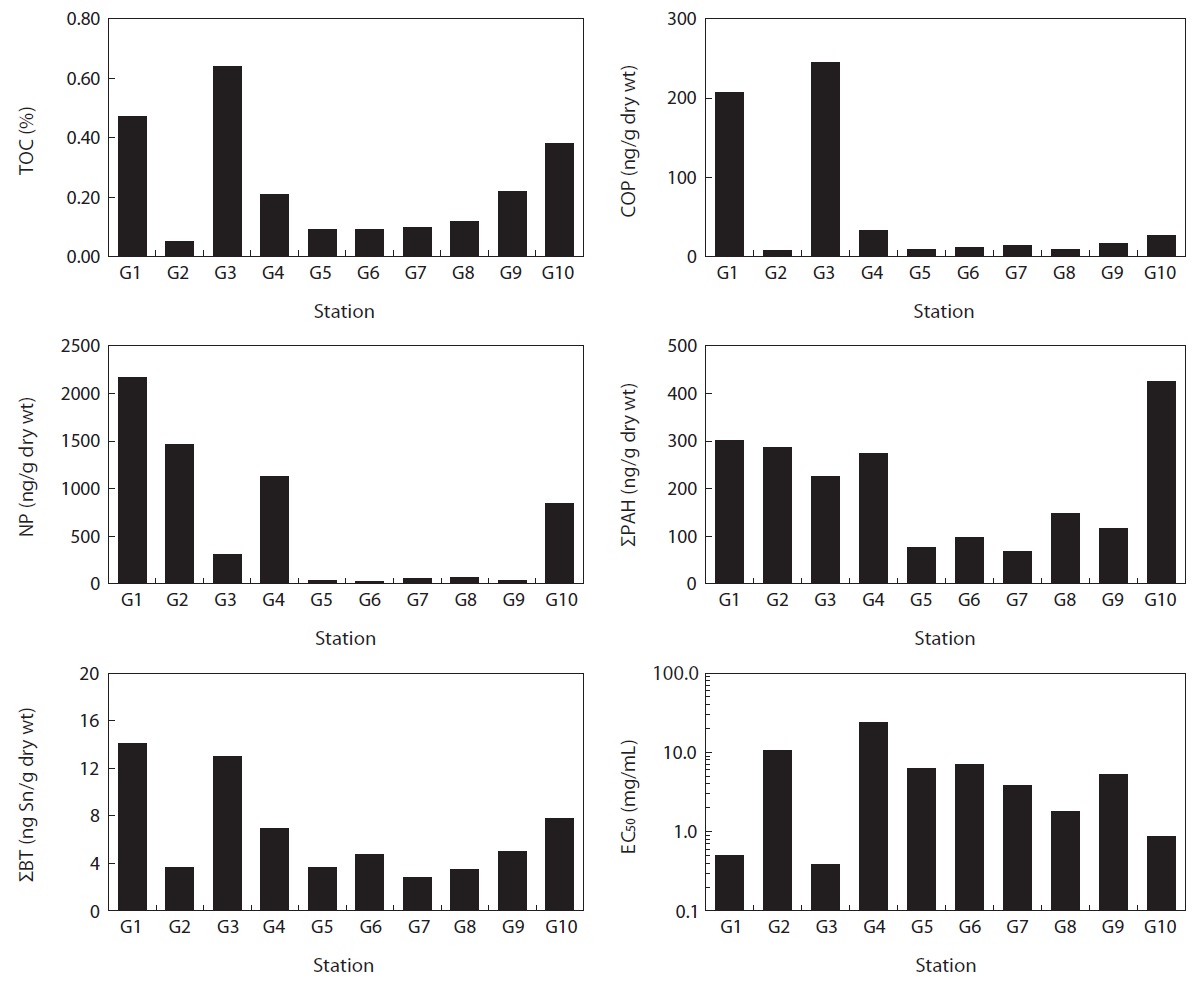
Site G3 had the highest TOC content in our study, and high contents were also found at sites G1 and G10 (Fig. 2). However, the other sites contained a lower TOC content than the mean content. The correlation of TOC contents in marine sediments was significantly high with COP, and BTs (
The concentrations of COP, CHOE, and CHOA were in the ranges of 7.60-245 ng/g dry wt (mean, 57.9 ng/g dry wt), 187- 447 ng/g dry wt (273 ng/g dry wt), and 61.8-188 ng/g dry wt (99.3 ng/g dry wt) in surface sediments, respectively. CHOE was the dominant compound in target fecal sterols, accounting for 67 ± 13% (mean ± SD). This may be because CHOE can originate from various sources including wastewater and marine organisms, from plankton to marine mammals. High CHOE concentrations were also reported in Ulsan Bay, Masan Bay, and the Mokpo coast (Choi et al., 2005a, 2005b, 2007). Seven out of the 10 samples had higher concentrations of COP than the background level of 10 ng/g dry wt (Hatcher and McGillivary, 1979), indicating an impact from fecal pollution in the Gunsan coast. The range of COP values (7.60- 245 ng/g dry wt) in the present study was lower than those previously reported for Kyeonggi Bay (3-3,800 ng/g dry wt) (Li et al., 2007), Masan Bay (130-3,964 ng/g dry wt) (Choi et al., 2005b), Ulsan Bay (141-8,257 ng/g dry wt) (Choi et al., 2005a), and the Mokpo coast, Korea (43-7,902 ng/g dry wt) (Choi et al., 2007). In addition, the concentrations of COP in the present study were much lower than those reported in Boston Harbor, USA (260-12,000 ng/g dw) (Eganhouse and Sherblom, 2001), Cootes Paradise, Ontario, Canada (150- 38,000 ng/g dw) (Mayer et al., 2007), Santa Monica Bay, USA (70-45,260 ng/g dry wt) (Venkatesan et al., 2010), and Guanabara Bay, Brazil (330-40,000 ng/g dry wt) (Carreira et al., 2004). Therefore, it can be concluded that sewage pollution in the Gunsan coast is low, compared to some other locations worldwide.
The highest concentration of COP, 245 ng/g dry wt, was found at site G3, which was located near a large port including a cruise terminal. The next highest concentration, 206 ng/g dry wt, was found at site G1 which was located near a fishing port and the mouth of Gyengpo creek, which passes through the center of Gunsan City (Fig. 2). The concentrations of COP at sites G1 and G3 were approximately four times higher than the mean concentrations. Grimalt et al. (1990) suggested that a range of 0.7-1.0 for the COP/(COP + CHOA) ratio is characteristic of sediment polluted by urban sewage and <0.3 is typical of unpolluted sediments. High COP/(COP + CHOA) ratios of approximately 0.6 were also reported in locations adjacent to a sewage outfall (Jeong and Han, 1994) and a sewage sludge disposal site (Chan et al., 1998). The ratio in the present study ranged from 0.10 to 0.57, and its distribution was similar to the COP distribution (
The concentrations of NP ranged between 20.7 and 2,171 ng/g dry wt (mean, 613 ng/g dry wt) in surface sediments. In the present study, the NP concentrations in surface sediments revealed contamination levels higher than or comparable to those previously reported for Korean coasts: Mokpo coast (4- 580 ng/g dry wt) (Choi et al., 2007), Masan Bay (92-600 ng/g dry wt) (Li et al., 2008), Kyeonggi Bay (5-1,100 ng/g dry wt) (Li et al., 2007), Lake Shihwa (10-5,100 ng/g dry wt) (Li et al., 2004; Choi et al., 2011a), and Ulsan Bay (13-5,772 ng/g dry wt) (Choi et al., 2011b). When considering the screening values and ecotoxicological potential of NP, 4 of the 10 samples from the present study exceeded the maximum permissible concentration in The Netherlands (105 ng/g dry wt) (Jonkers et al., 2005). The NP concentrations in these four samples also exceeded the Canadian sediment-quality guideline for NP (1.0 μg NP toxic equivalency/g dry wt [1% TOC]) (Environment Canada, 2002). This indicates that the Gunsan coast is heavily contaminated by industrial wastewaters including NP.
The high NP concentration recorded in this study may indicate the presence of hot spots directly receiving wastewater treatment plant (WWTP) effluents and/or untreated river waters. The highest concentration of NP, 2,171 ng/g dry wt, was found at site G1, where Gyengpo creek is discharged (Fig. 2). NP concentrations higher than 1,000 ng/g dry wt were found at sites G2 and G4, located in the proximity of Janghang City and several industrial complexes. In particular, the outfall of WWTP that handles the industrial wastewater was located close to site G4. This suggests that riverine discharges and WWTP effluents are strong potential sources of industrial wastewater in the Gunsan coast. Similar results were reported for Lake Shihwa and Ulsan Bay (Choi et al., 2011a, 2011b). Interestingly, a NP concentration exceeding the screening values, 841 ng/g dry wt, was detected at site G10 close to the power plant. In Korea, NP is used primarily as a surfactant (60%), followed by uses as additives for paints and epoxy resins (12%), copper laminates (9%), ink binder (5%), agricultural pesticides (2%), and other minor uses. The South Korean government designated it as a priority chemical, prohibiting its use as a component of household cleaners in 2002 and all domestic applications in 2007 (Ministry of Environment, 2007). There are no reports regarding the use of NP in power plants, although NP emissions are associated with industrial activities. More intensive monitoring will be needed to identify the sources of NP around this area. The spatial distribution of NP in sediments from the Gunsan coast differed from that for COP (
>
Polycyclic aromatic hydrocarbons
ΣPAH in sediments ranged from 67.9 to 425 ng/g dry wt (mean, 202 ng/g dry wt). The sum of the six potential carcinogenic PAHs (the sum of BaA, BbF, BkF, BaF, InP, and DbA) (International Agency for Research on Cancer, 1984), ranged from 9.96 to 95.5 ng/g dry wt (mean, 39.2 ng/g dry wt), accounting for 19% of ΣPAH in sediments. The predominant compounds identified were Pyr, FluA, NaP, BaF, and Flu, collectively accounting for 68% of ΣPAH.
The spatial distribution of ΣPAH in Gunsan sediments showed a clear decreasing trend from the inner to outer locations (Fig. 2). ΣPAH concentrations exhibited significant positive relationships with carcinogenic PAHs (
The highest concentration of ΣPAH, 425 ng/g dry wt, was found at site G10 close to an anthracite-fired power plant. The next highest concentration of ΣPAH was found at the inner locations (sites G1, G2, G3, and G4) near Gunsan and Janghang City, where an international cruise terminal and an industrial complex are located. Concentration profiles of PAHs can provide useful information regarding contamination sources in marine sediments (Mille et al., 1982). The ratio of FluA to Pyr > 1 implies a pyrolytic origin, and the ratio of FluA to Pyr < 1 implies a petrogenic origin of PAHs (Colombo et al., 1989). The ratio of FluA to Pyr ranged from 0.6 to 3.3. Seven of 10 locations exhibited values of FluA to Pyr > 1, indicating that fossil fuel combustion was the major source of PAHs in the region. In particular, the highest value was found at site G10 close to the power plant. However, some locations such as sites G1, G6, and G7 showed values of FluA to Pyr < 1. This result indicates both pyrolytic and petrogenic inputs, although inputs of pyrolytic origin predominate at these sites.
BT was found in all sediment samples and ΣBT concentrations ranged from 2.79 to 14.1 ng Sn/g dry wt. Concentrations of TBT, DBT, and MBT were in the ranges of 0.30-0.88 ng Sn/g dry wt, 0.93-9.38 ng Sn/g dry wt, and 1.03-5.47 ng Sn/g dry wt, respectively. DBT was the main BT compound identified and its proportions accounted for 50 ± 12% (mean ± SD) of the total, suggesting that TBT has no ongoing source in the Gunsan coast. High concentrations of ΣBT were observed at sites G1 and G3. However, TBT concentrations in the Gunsan coast are much lower than previously reported in Busan Harbor (4-1,917 ng Sn/g dry wt) (Choi et al., 2009b), Ulsan Harbor ( Spearman rank correlation coefficients of sediment toxicity (EC50) with chemical concentrations screening and ecotoxicological values: the Australian low and high trigger values (5 and 70 ng Sn/g dry wt) (Burton et al., 2005), chronic effects on amphipods and polychaetes (42 ng Sn/g dry wt) (Matthiessen and Thain, 1989), and chronic effects on clam (307 ng Sn/g dry wt) (Langston and Burt, 1991). Therefore, the TBT level in Gunsan marine sediments is considered to be low and safe for aquatic organisms.

>
Sediment toxicity using Vibrio fischeri
The EC50 values of sediments ranged from 0.38 to 23.8 mg/ mL. The EC50 values of sediments in this study were much higher than those reported in the following Korean coastal regions associated with intensive marine and industrial activities: Masan Bay (0.014-0.089 mg/mL), Lake Shihwa (0.018- 0.061 mg/mL), Ulsan Bay (0.066-1.126 mg/mL) (Choi et al., 2010a), and the outfall area of the Masan WWTP (0.014-0.148 mg/mL) (Choi et al., 2010c). However, Southern Puget Sound, USA (0.31-175.30 mg/mL) (Long et al., 2002), the Po River, Italy (0.46-36.80 mg/mL) (Vigano et al., 2003), Waukegan Harbor, USA (0.42-14.50 mg/mL) (Kemble et al., 2000), and the Ebro River, Spain (0.04-12.96 mg/mL) (Ocampo-Duque et al., 2008) were higher or comparable to our results. Therefore, sediment toxicity in the present study is considered within the middle range of toxicities reported worldwide.
Long et al. (2002) suggested critical values of less than 80% and 90% of the negative control response, EC50 < 0.51 mg/mL and <0.06 mg/mL, respectively, to identify toxic samples. Critical values were calculated on the basis of the frequency distribution of Microtox data from US nationwide surveys by the National Oceanic and Atmospheric Administration (NOAA) to denote the 80% and 90% lower prediction limits (LPL) of the NOAA Microtox database (
Spearman rank correlation coefficients between sediment toxicity and chemical levels were estimated (Table 2). The sediment toxicity (EC50) was negatively correlated with all contaminant components, suggesting that environmental disturbance by chemical contaminants results in changes in sediment toxicity. The significant negative relationships of sediment toxicity with TOC (
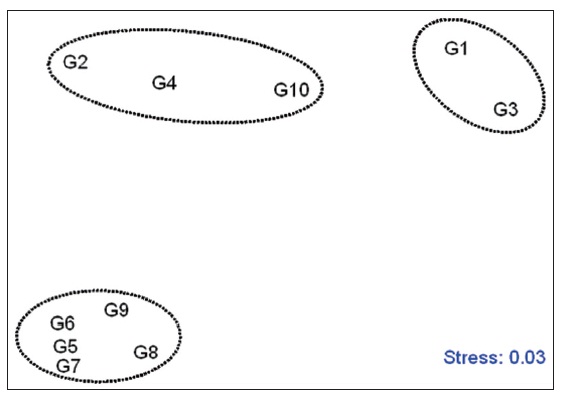
Non-parametric MDS ordination using the sediment toxicity and chemical data for the sampling locations was performed
to characterize spatial variations (Fig. 3). Non-parametric MDS suggested three cluster groups among the sampling sites. The stress of the plots was 0.03; less than 0.1 represents a good ordination with little chance of misinterpretation (Clark and Warwick, 1994). The first group comprised the sites G1 and G3, which were characterized by severe pollution from organic contaminants (TOC, COP, and BTs), and sediment toxicity. The second group represented sites G2, G4, and G10, which were characterized by relatively high concentrations of contaminants related to industrial activity (NP and PAHs) compared to those in the first group. The other group was the remaining sites G5, G6, G7, G8, and G9, which were characterized by relatively low contaminant concentrations owing to the long distances from inland sources and the influence of stronger currents. Therefore, anthropogenic organic pollution in the Gunsan marine sediments is considered to be mainly affected by point sources including the WWTP and the power plant, and ports with high shipping traffic for both cruises and fishing boats.