Soil animals in the forest ecosystem play vital roles in the decomposition of organic matter and nutrient cycling while influencing the soil structure and physic-chemical properties (Wallwork 1983, Lussenhop 1992). The major groups of meso- and macro-fauna in forest soil ecosystem include acari, collembola, araneae, nematodes, some dipteral larvae and other arthropods (Schaefer 1991) with dominance of acari and collembola (Kim and Jung 2008, Jung et al. 2010). Among acari, predatory gamasid mites are the top predators of saprophagous oribatid mites, prostigmatic mites and collembolans. Oribatid mites are most abundant and super species-rich arthropod group in forest soils (Petersen and Luxton 1982, Park et al. 1996, Bae and Lee 1997, Zaitsev and van Straalen 2001, Erdmann et al. 2006, Kim and Jung 2008, Kim et al. 2010, 2011). Feeding habits of oribatid mites are mostly saprophagous utilizing parenchymatous tissue of dead leaves (phyllophagy) or woody structural tissue of dead plant (xylophagy), and mycophagous feeding on fungal hyphae or spores. But few species are even bacteriophaous or predatory (Behan-Pelletier 1999). Thus oribatid mites directly regulate the decomposition of plant debris and indirectly influence the microfloral activity (Clement and Haq 1984, Norton 1985, Afifi et al. 1989) occupying the critical niche in soil ecosystem (Wallwork 1983, Jung et al. 1998, 2002, Jandi et al. 2003, Kang et al. 2007).
Even though many factors influence the stability of soil ecosystem, it is often considered that soil ecosystem is relatively resistant to the small scale disturbance due to the soil buffering effect (Scheinost et al. 1997). How-ever, disturbance is ubiquitous in most ecosystems (Clements 1936). In temperate forest, there would be
The frequency of fire disturbance is increasing recently (Ministry of Environment 2002). There are several reports on fire impacts and post-fire recovery of the vegetations (Choung et al. 2004), soil animals (Choi 1996, Son and Choi 2000, Kim and Jung 2008, Jung et al. 2010), oribatid mite communities (Choi 1996, Fukuyama et al. 1999, Dress and Boemer 2004, Malmstorm 2008). However no effort has been made to understand the recovery process after the fire in relation to the resilience after disturbance.
The aim of this study was to compare the response of soil oribatid mites to the wild fire in the sites with different fire severity and to test the model explaining the impact and recovery patterns relative to the resilience after the disturbance. Especially we examined whether the impacts and recovery were significantly different among the different fire severity sites, and if the reorganization of communities show any sign of intermediate disturbance hypothesis (Connell 1978, Huston 1979), in that case diversity would be higher in intermediately disturbed sites. Finally we asked if the pattern could be explained in terms of resilience after the disturbance.
The study sites were located at Imwon-ri and Yang-ri, Samcheok, Gangwon province, Korea. The geographic position is 37°02′ - 37°28′N and 128°57′ - 129°21′E. This area was burned during the East coast mountain fire in 2000 (Ministry of Environment 2002). The natural site (N) was selected in Yang-ri where the forest structures dominated by ap. 30-year-old pine tree (
>
Soil sampling and identification of oribatid mites
Soil samples were collected from fire damaged sites at 5, 6, 7 years after the fire in summer season. One sample consists of five soil cores (5 cm diameter and 5 cm depth) with 8 replicates in each site. Fresh leaf litter was discarded during sampling. Only hummus layer and soil layer were included in the core sample. Oribatid mites were extracted using a modified Berlese-Tullgren funnel for 72 hours with 30 watt bulb (Kim and Jung 2008, Jung et al. 2010, Kim et al. 2010a, 2011b). The extracted mites were stored at 70% ethanol solution and mounted on the slide glass using PVA mounting medium (Bioquip, Rancho Dominguez, CA, USA) (Downs 1943). Oribatid mite species were identified according to Ehara (1980), Balogh and Mahunka (1983), Choi (1984), Aoki (1999), Balogh and Balogh (2002), and Weigmann (2006) under an optical microscope (× 400) (Olympus, PA, USA).
Abundance data were transformed into square meter scale for easy comparing with published data. The abundance, species richness and diversity index (
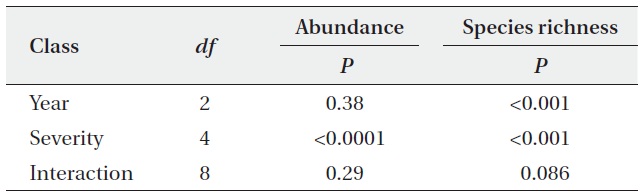
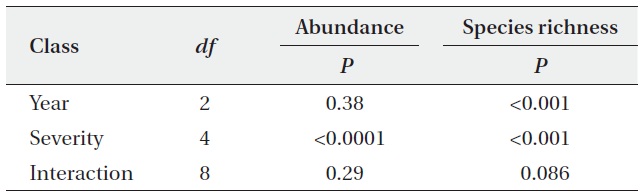
Statistical summary of analysis of variance of abundance and species richness of oribatid mite from the different degree of fire severity
ver 9.2 (SAS Institute Inc., Cary, NC, USA) (SAS Institute 2008).
The abundance and species richness of oribatid mites were significantly altered from the fire disturbance. Abundances were different among the site with different fire severity, but not among the year (Table 1). The species richness was significantly different among sites as well as over the years (Table 1).
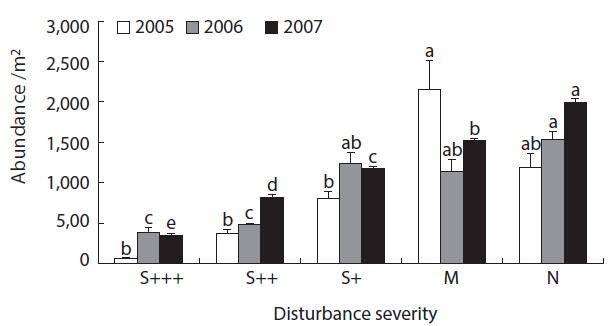
The abundances of soil oribatid mites were as low as 100 - 500 individuals per square meter area in severely damaged sites (S+++ or S++) while more than 1000 indi-viduals
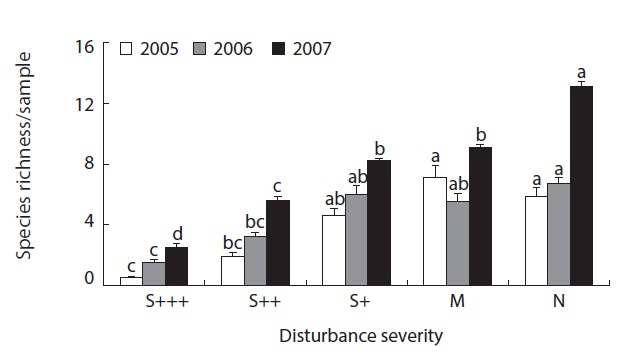
were recovered from less damaged or non-damaged natural sites (S+, M or N) (Fig. 1). There was clear pattern of increasing abundance as the disturbance severity weekened. Also even with no statistical significance, abundance was in increasing pattern as the time after disturbance increase. The same patterns were obvious in species richness data (Fig. 2). From the severely disturbed sites, 0 - 4 species were collected from each sample, but more than double of species were collected from less disturbed sites even after 5 years of the fire disturbance.
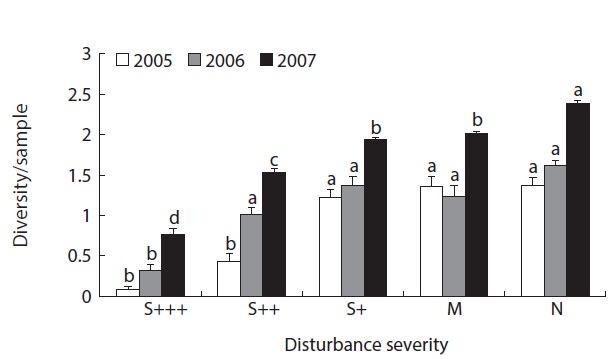
Local diversity index (
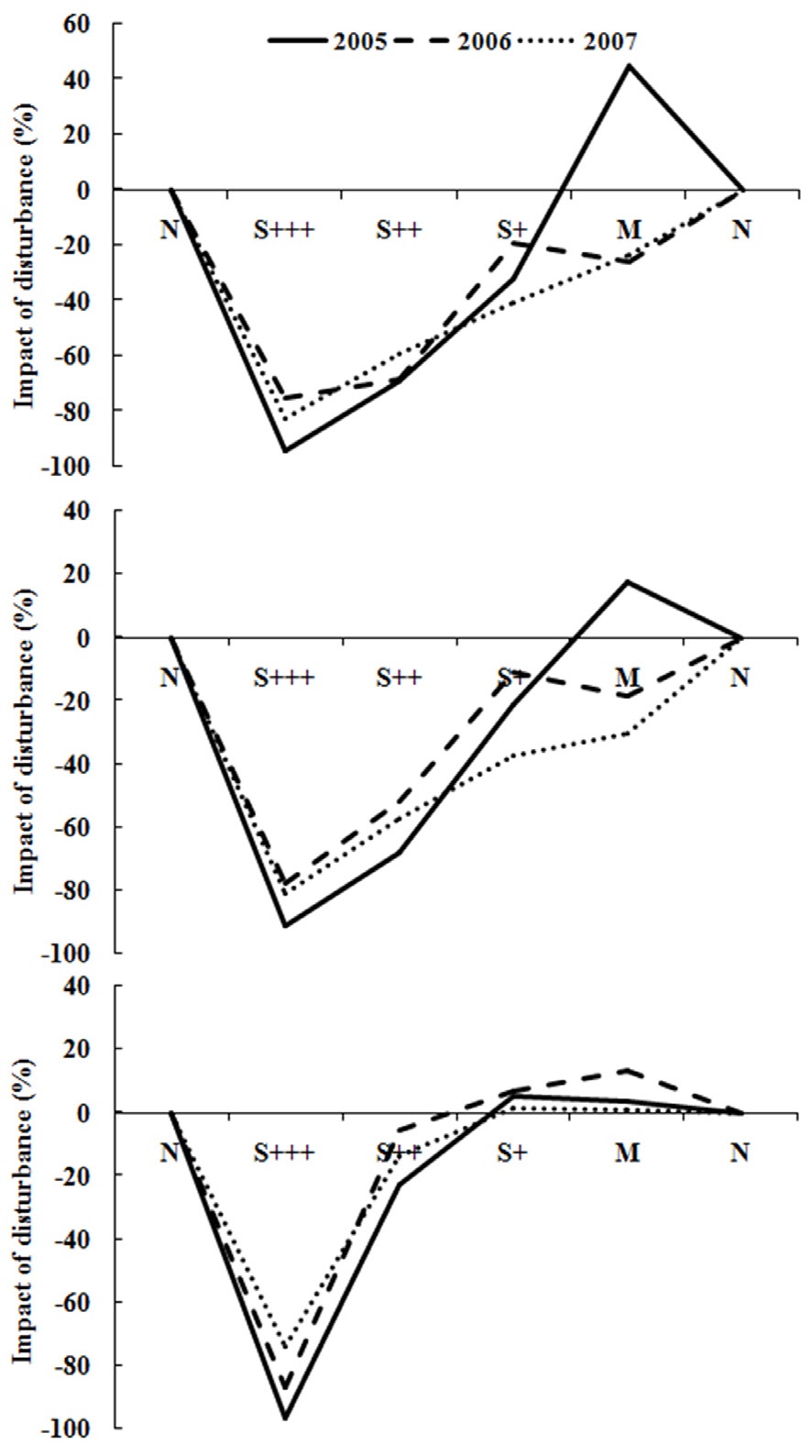
Fig. 4 showed the resilience pattern of reorganization of oribatid mite communities. Impacts of disturbance in severely damaged sites were more than 90% reduction of the abundance and species richness while minor damaged sites showed around 20% increase (Fig. 4a and 4b) after 5 years of disturbance in 2005. In species divesrity and disturbance graph (Fig. 4c), it was more clear that the diversity was much lower in severely disturbed sites, but similar or higher in less damaged sites. And the diversity showed increasing trend as the recovery phase elapsed.
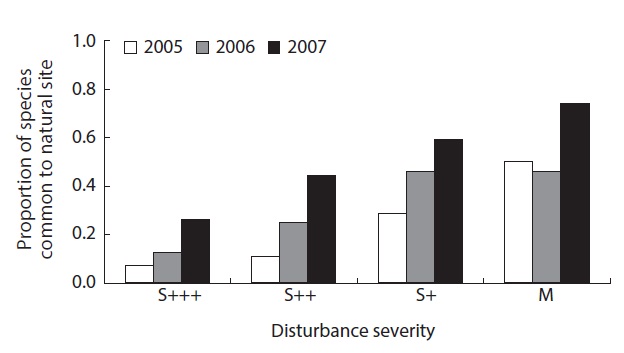
Proportions of the species common to the reference natural site (N) where no fire disturbance occurred were between 7 - 74%, with increasing pattern of this proportion as the disturbance severity decreased. As the time progressed, the proportion also increased from 24%, 32% and 51% in 2005, 2006 and 2007, respectively, eventhough there was not significant different (Fig. 5).
This study demonstrates that the abundance, species richness and diversity showed negative impacts from the fire disturbance. Disturbance in ecosystem can be distinguished as three types relative to their effects on organisms and ecosystems (Bengtsson 2002). Frequent or regular
fire and tilling). Large
This study showed the typical resilience pattern of soil oribatid mite communities after the massive fire disturbance, even though the temporal resilience was not pronounced. If the further long term study (e.g., 10, 20 and 50 years after disturbance) on the reorganization of oribatid mite community after the disturbance, then demonstration of the resilience and elasticity could be possible. However, the amplitude was partly explained from the existing data. Diversity is a key component in ecological resilience which is better than abundance or species richness because the diversity serves as a shortcut indication of the ecosystem functioning (Bengtsson 2002). In severely disturbed sites, diversity was heavily impacted almost leading the oribatid mite community extinct, while recovery also took place in lightly disturbed sites. Also it was clear that the relative impacts were decreasing as the time after disturbance increased.
In the reorganization phase, species structure changed dramatically. The local diversity after disturbance includes the survivors and recolonizers or invaders. The proportion of the species common to the reference natural sites increased from ap. 25% to more than 51% in three years. The value may indicate the naturalness of the diversity.
While recovering phase, minor disturbance site showed even higher abundance and species richness supporting the intermediate disturbance hypothesis. Intermediate disturbance hypothesis (IDH) states that diversity will be highest at sites that have had an intermediate frequency of disturbance that prevents competitive exclusion and will be lower at sites that have experienced very high or very low disturbance. In this study, it was clear that the diversity was higher in less severe or minor damaged sites
[Table 2.] Fire impact according to fire type in abundance and species richness on soil fauna
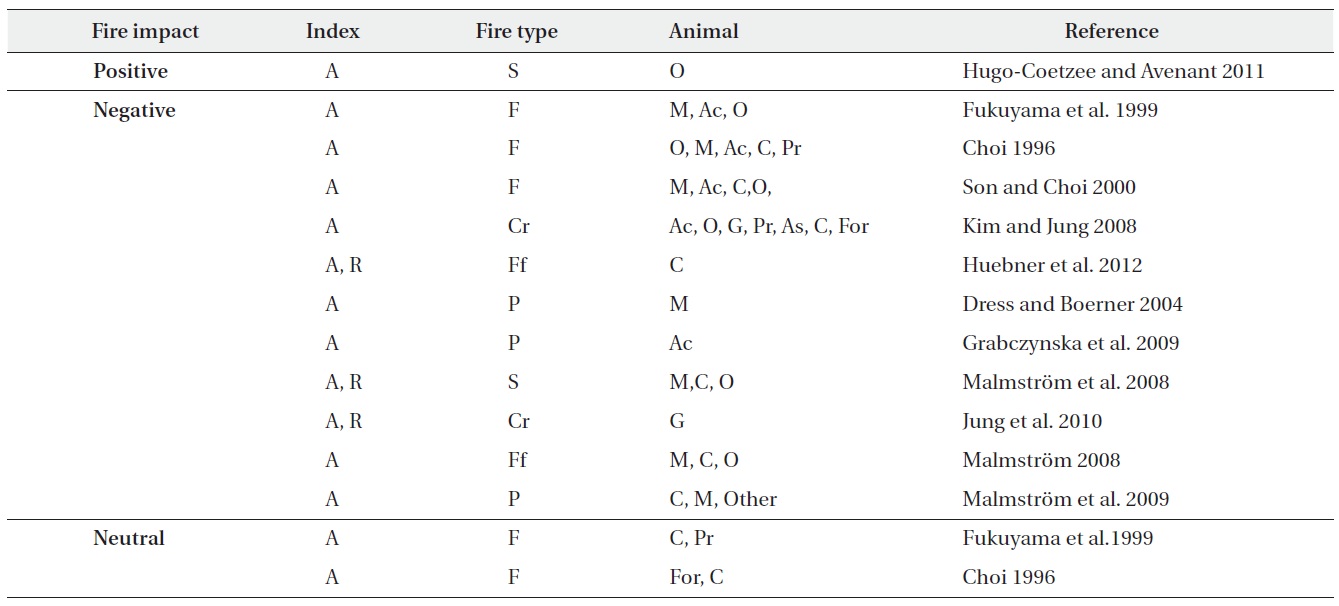
Fire impact according to fire type in abundance and species richness on soil fauna
than in non-damaged sites (N) or severely damaged sites (S+++ or S++) during the recovering phase after the fire disturbance. This is contrasting to Schwilk et al. (1997) or Collins (1992). They suggested that the IDH may not hold true in the Mediterranean-climate shrublands of South Africa or North American grasslands. Our study was an example of temperate pine forest. Further designed study is requested to test if the disturbance response may differ depending on the ecosystem and the target organism groups.
In conclusion, the study reports significant reduction of abundance, diversity and species richness of soil dwelling oribatid mites when faced with the fire disturbance. In reorganizing phase, partial data supported the intermediate disturbance hypothesis and the pattern of resilience after disturbance.