The venoms from rattlesnakes in America and from salmusa in Asia have toxic effects on the vascular tract and affect blood circulation [1]. The poisonous components in the snake venom, either enzymes or non-enzymatic substances, cause drastic changes in the circulation of blood and thus lead to outbreaks of hemorrhage. The activities of the toxins include coagulation of blood, inhibition of blood coagulation, platelet activation, anti-platelet function, fibrinolysis activation, and hemorrhage. Blood coagulation has been reported to be inhibited by fibrin(ogen)olytic enzymes from the snake venom of the families of
The fibrin(ogen)olytic enzyme in the snake venom is defined as an enzyme that can hydrolyze fibrin and fibrinogen to liquify insoluble fibrin-rich clot and to remove fibrinogen from blood [2]. The enzyme is also an endoproteinase, which can be classified as either serine proteinase or metalloproteinase based on the structure of active site. The metalloproteinase mostly requires zinc at the active site and is mostly on α- chain fibrinogenase [1], but there are some exceptions [3]. However, most serine fibrin(ogen)olytic enzymes preferentially hydrolyze β-chain over α-chain of fibrinogen [4-6].
The fibrin(ogen)olytic enzyme from snake venom has received attention as a therapeutic agent of oriental pharmacopuncture medicine as well as Western medicine to treat obstructive thrombosis and acute stroke by removing fibrinogen and fibrin clots from the blood [7]. Thus, numerous fibrin(ogen)olytic enzymes have been isolated from snake venoms, and their abilities to treat cardiovascular disease without side effects have been evaluated [1]. One of the potential side effects of fibrin(ogen)olytic enzymes for clinical application is hemorrhage [2].
The objective of this study were to isolate a fibrin(ogen)olytic enzyme from the snake venom of
2.1. Isolation of fibrinolytic enzyme from snake venom
The snake venom of
2.2. Sodium dodecyl sulfate-polyacrylamide gel
electrophoresis
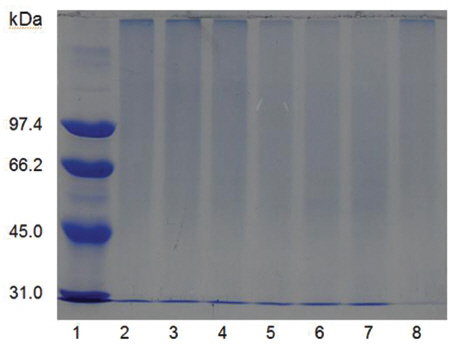
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed according to the procedure described by Laemmli [12]. The molecular weight markers (Bio-Rad, USA) for SDSPAGE were phosphorylase b (97.4 kDa), serum albumin (66.2 kDa), ovalbumin (45.0 kDa), carbonic anhydrase (31.0 kDa), trypsin inhibitor (21.5 kDa), and lysozyme (14.4 kDa). β-Mercaptoethanol (5%) was added to the sample buffer used for all SDS-PAGE except for that in (Fig 5), lane C. Iodoacetate (0.15 M), instead of β-mercaptoethanol (5%), was added to the sample buffer in (Fig 5), lane C. Iodoacetate (0.15 M) was added after heating at 100℃ for 5 min in (Fig 5), lane B.
2.3. Protein concentration determination
The protein concentration was determined using BCA Protein Assay Reagent (Pierce, USA). Bovine serum albumin was used for the calibration line.
2.4. Protein hydrolysis analysis
Fibrinogen and gelatin was hydrolyzed using the isolated fibrinolytic enzyme and then subjected to SDS-PAGE. An aliquot (0.2 ml) of 10 mg/ml proteins in 50 mM Tris-HCl pH 7.6, 0.15 M NaCl was mixed with 0.05 ml of 0.2 mg/ml fibrinolytic enzyme. The mixture was incubated at 37 ℃ for 6 h. An aliquot (10 ㎕) was taken at 0.5, 1, 2, 4, and 6 h, and was added to the sample buffer containing β-mercaptoethanol. The mixture was heated at 100 ℃ for 5 min before SD SPAGE.
The experiment to determine the effect of the fibrinolytic enzyme on hemorrhage reaction beneath back skins of mice was approved by the Animal Experiment Ethics Committee of Sangji University (Approval document No. 2102-9). An aliquot (0.1 ml) of diluted fibrinolytic enzyme in 50 mM Tris-HCl pH 7.6, 0.15 NaCl was injected subcutaneously to Institute of Cancer Research (ICR) mice of 6-week-old (Daehan Biolink, Korea). The mice were sacrificed using cervical dislocation after 6 h and the skin around the injection site was stripped off to determine the diameter of the hemorrhage zone formed beneath the skin. Two diameters (r1 and r2) at a right angle were measured. The hemorrhagic area was calculated following formula of 0.785r1r2. The hemorrhagic areas were measured in duplicate by injecting two mice, and the average was calculated.
2.6. Amino acid composition analysis
The amino acid composition of the fibrinolytic enzyme was analyzed by Korea Basic Science Institute. The PICO-tag procedure of high performance liquid chromatography (HPLC) was used to analyze PITC-labeled amino acids. The HPLC column was Waters Pico-tag Column. The HPLC instruments (Waters Corporation, USA) consisted of 510 HPLC pump, gradient controller, and 2487 UV detector.
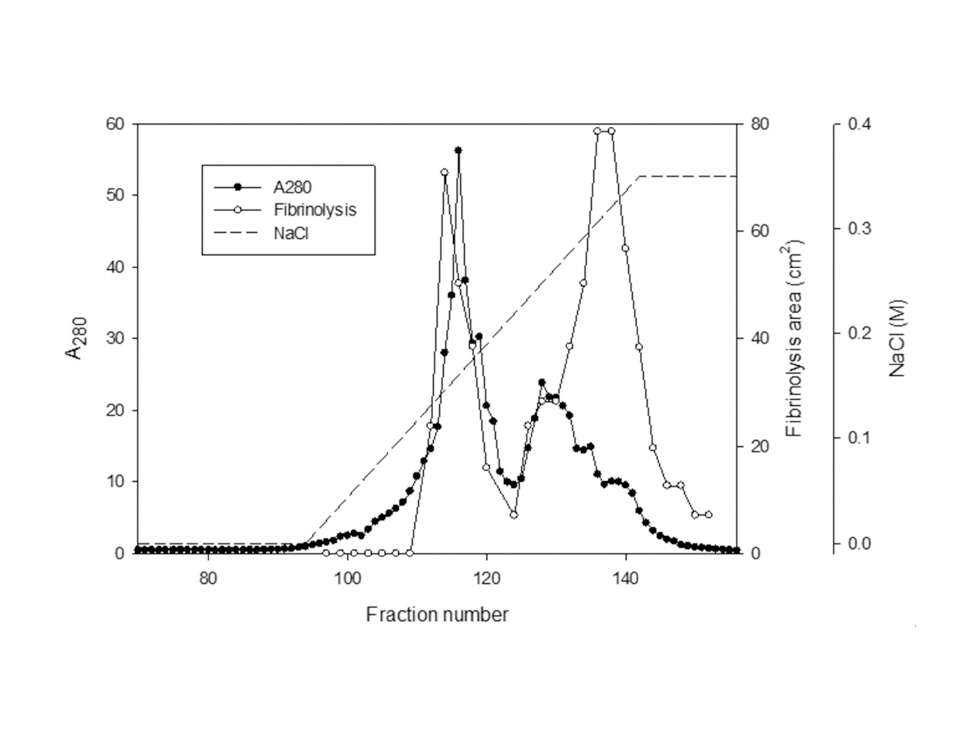
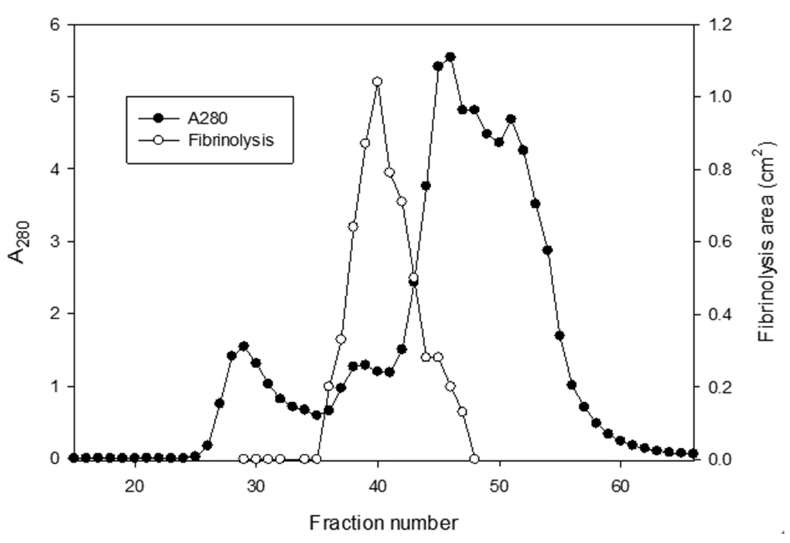
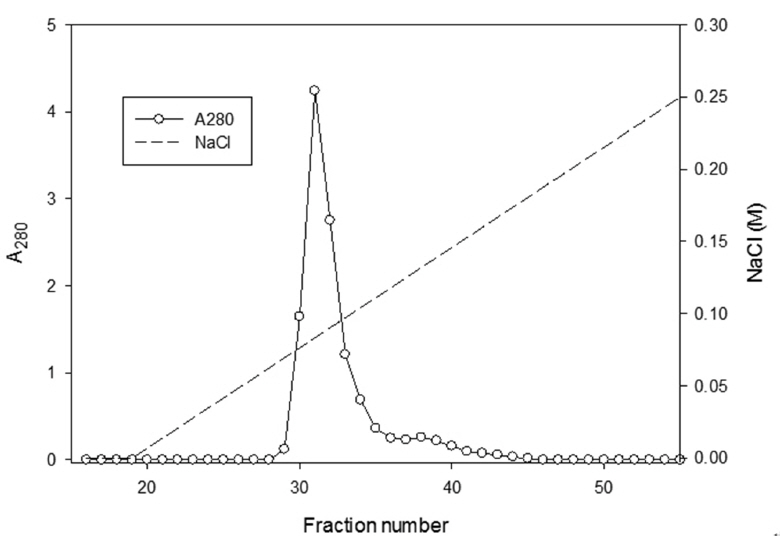
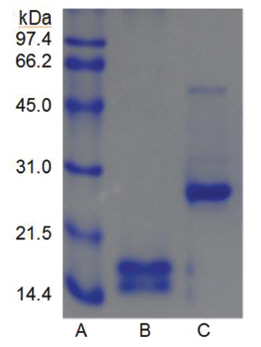
The sample containing the two polypeptides with the molecular weights of 15 kDa and 18 kDa described by Choi [11] was obtained from chromatography of
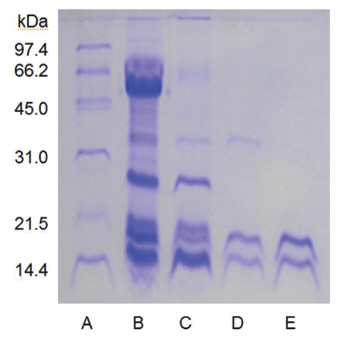
When the sample was heated in the sample buffer containing iodooacetate before electrophoresis, a major polypeptide with molecular weight of 27 kDa and a minor polypeptide with molecular weight of 50 kDa were detected in SDS-PAGE (Fig 5), lane C. The minor polypeptide with the molecular weight of 50 kDa seemed to be a dimer consisting of two FE-27kDa linked by a disulfide bond. Since iodoacetate, one of alkylating agent of sulfhydryl group, prevents formation of linkage between polypeptides and of thus polymeric proteins, the dimeric polypeptide should not be an artifact of SDS-PAGE.
When the sample was heated in the sample buffer containing ß-mercaptoethanol and then iodoacetate was added to the sample before electrophoresis, the two polypeptides with the molecular weights of 15 kDa and 18 kDa were detected in SDS-PAGE (Fig 5), lane B. These results suggested that the fibrinolytic enzyme with the molecular weight of 27 kDa consisted of the two disulfide bond-linked polypeptides. The fibrinolytic enzyme was then designated FE-27kDa in this study.
However, the apparent molecular weight of 27 kDa obtained from SDS-PAGE was less than the calculated molecular weight of 33 kDa obtained by adding the molecular weights of the two polypeptides. The discrepancy between the two molecular weights may be due to the fact that the two polypeptides linked by a disulfide bond could not be converted to a fullyextended linear structure of a polypeptide with the same molecular weight, but were converted to a linear structure lined side by side with the two polypeptides in the gel of SDS-PAGE without ß-mercaptoethanol. Thus, the hydrodynamic volume of the linear structure lined with the two polypeptides should be less than that of the fully-extended linear structure, which would result in a lower apparent molecular weight of 27 kDa. The structure of FE-27kDa was similar to that of brevinase isolated from the snake venom of a Korean snake,
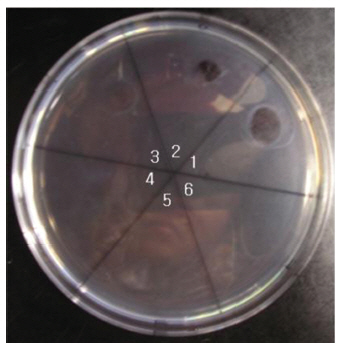
The areas of the fibrinolysis zones in the fibrin plate assay were 0.71 cm2 and 0.33 cm2 at 3 mg/ml and 2 mg/ml of FE-27kDa, respectively (Figs 6,1) and 2). The fibrin plate assay showed that the minimum concentration of FE-27kDa to form a fibrinolysis zone was about seven times higher than that (0.3 mg/ml) of the fibrinolytic enzyme with a molecular weight of 54 kDa (FE-54kDa) [11], suggesting that the fibrinolytic activity of FE-27kDa was weaker than that of FE- 54kDa.
Protease inhibitors were added to the solution containing FE-27kDa (2.5 mg/ml) in the fibrin plate assay. The relative fibrinolysis was the ratio of the area of the enzyme treated with protease inhibitor to that of a control without treatment (Table 1) (Table 1) shows that among the chelate compounds, which remove metal ions from the enzyme, EDTA inhibited fibrinolysis completely, 1, 10-phenanthroline inhibited it strongly, and EGTA did not inhibit it. PMSF, a serine protease inhibitor, inhibited fibrinolysis completely. However, TLCK, an inhibitor of trypsin and trypsin-like protease, did not show any effect. Dithiothreitol and cysteine, which cleave disulfide bonds, inhibited fibrinolysis strongly, but iodoacetate did not have any effect on it. These results suggest that FE-27kDa is not a trypsin-like enzyme, but a serine proteinase. The disulfide bond is required for fibrinolytic activity, and metal ions are needed to promote fibrinolysis. The inhibition pattern of FE- 27kDa is similar to that of brevinase reported by Lee et al.[6]. Brevinase is moderately inhibited by aprotinin, bezamidine, EDTA, EGTA, and 1, 10- phenanthroline. The fibrinolytic activity is completely inhibited by PMSF, Pefabloc, and dithiotreitol. These results indicate that brevinase is also a serine proteinase requiring a disulfide bond for its fibrinolytic activity. However, FE-54kDa [11] is a metalloproteinase containing a disulfide linkage. The enzyme is inhibited by chelating agents such as EDTA, EGTA, and 1, 10-phenanthroline and by reducing agents such as dithiothreitol and cysteine.
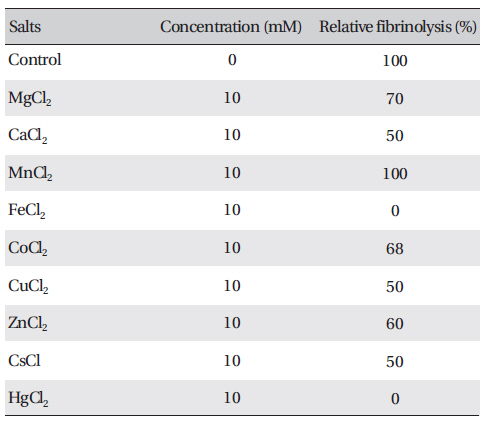
The effects of metal ions on the fibrinolytic activity of FE-27kDa were determined (Table 2) The concentration of salts added to FE-27kDa (2.5 mg/ml) was 10 mM. (Table 2) shows that Hg2+ and Fe2+ inhibited fibrinolysis completely and that Mg2+, Ca2+, Co2+, Cu2+, Zn2+ and Cs+ inhibited it moderately. Mn2+ was the only metal ion that did not show any effect on fibrinolysis. The inhibition by metal ions of FE-27kDa activity in this study was different from that of brevinase [6]. The fibrinolytic activity of brevinase is completely inhibited by Zn2+, but is unaffected by Ca2+, Co2+, Cu2+, Fe2+, Mg2+, Mn2+, K+, and Na+.
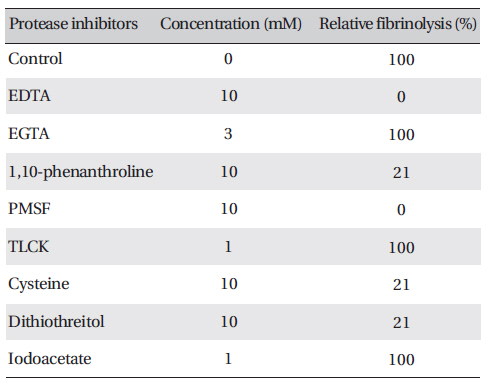
Effects of protease inhibitors on the fibrinolytic activity of FE-27kDa from G. b. siniticus venom
[Table. 2] Effects of salts on the fibrinolytic activity of FE-27kDa from G. b. siniticus venom
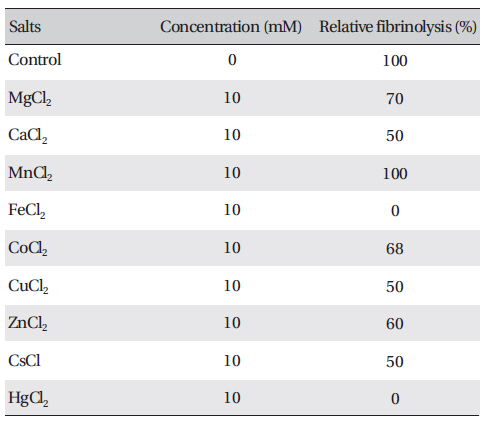
Effects of salts on the fibrinolytic activity of FE-27kDa from G. b. siniticus venom
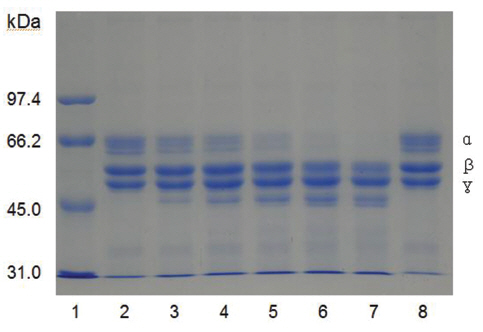
Fibrinogen was mixed with FE-27kDa and incubated at 37 ℃ for a certain time. The sample was subjected to SDS-PAGE to determine the fibrinogenolytic activity of FE-27kDa (Fig 7) The SDS-PAGE pattern showed that α-chain of fibrinogen decreased initially after 30 min (Fig 7), lane 3 and disappeared after 360 min (Fig 7), lane 7. The intensity of β-chain of fibrinogen was weakened after 240 min (Fig 7), lane 6. However, there was no change for γ-chain throughout the time of the incubation. These results showed that FE-27kDa hydrolyzed α-chain initially and then β- chain slowly. However, brevinase preferentially cleaves β-chain of fibrinogen and more slowly α-chain [6]. The concentration ratio of FE-27kDa to fibrinogen was 0.5% in the assay, suggesting that the fibrinogenolytic activity of FE-27kDa was weaker than that of brevinase whose concentration ratio to fibrinogen was 0.025%. FE-54kDa [11] preferentially cleaved α-chain of fibrinogen and slowly cleaved β-chain. The concentration ratio of FE- 54kDa to fibrinogen was 0.0025%, suggesting that it had very strong fibrinogenolytic activity.
Gelatin was mixed with FE-27kDa and incubated for a certain time. The sample was subjected to SDS-PAGE to determine the gelatin hydrolytic activity of FE-27kDa (Fig 8) According to the SDS-PAGE results, the gelatins incubated for 0 min (Fig 8), lane 2 and 30 min (Fig 8), lane 3 and the gelatin that was not incubated (Fig 7), lane 8 showed smear patterns of polypeptides that were strongly localized at molecular weights of more than 97.4 kDa. However, the polypeptide bands at molecular weights between 45.0 kDa and 66.2 kDa were intensified after more than 4 h, but there were no polypeptides with molecular weights lower than 45.0 kDa. These results suggest that even though the hydrolysis of the gelatin by FE-27kDa was evident, the enzyme did not hydrolyze the gelatin extensively. However, FE-54kDa hydrolyzed high-molecular-weight polypeptides of the gelatin extensively into low molecular- weight polypeptides with molecular weights less than 45.0 kDa [11].
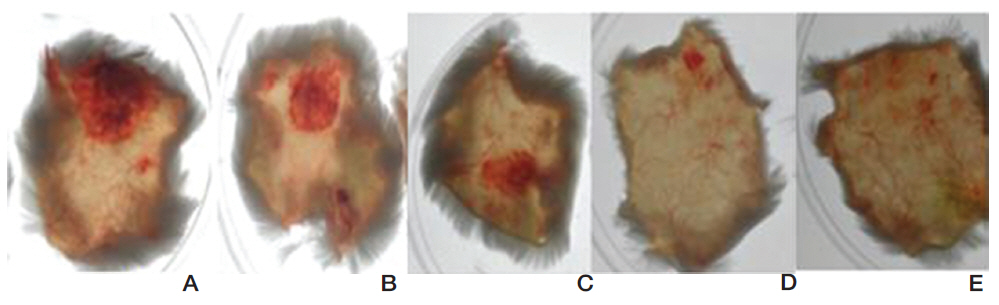
FE-27kDa was injected subcutaneously into shaved back skins of mice. The mice were sacrificed after six hour and then the diameter of the hemorrhage zone (Fig 9) formed beneath the skin was measured. When FE-27kDa was administered to a mouse at dosages of 100 μg, 50 μg, 25 μg, and 10 μg, the areas of the hemorrhage zones were 2.5 cm2, 1.5 cm2, 0.9 cm2, and 0.2 cm2, respectively (Fig 9, A-D). The results showed that the areas and the intensities of the hemorrhage zones were correlated with the dosages of FE-27kDa and that the minimum dosage of FE-27kDa to induce hemorrhage was 10 μg.
[Table. 3] Amino acid composition of FE-27kDa from G. b. siniticus venom
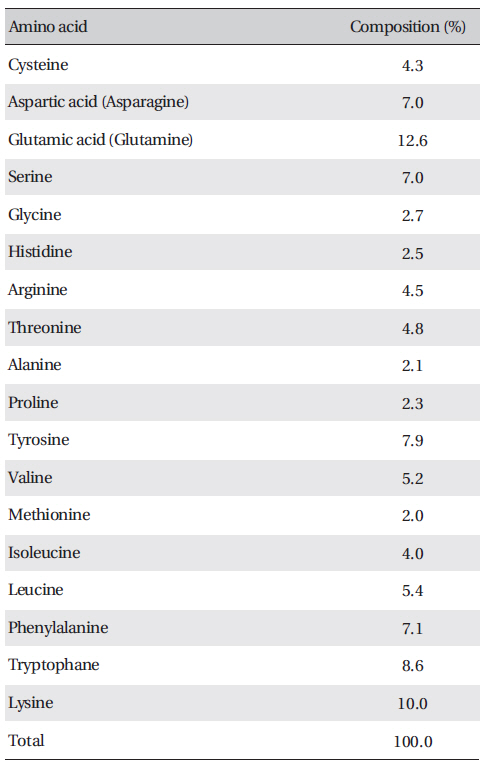
Amino acid composition of FE-27kDa from G. b. siniticus venom
Most of the fibrin(ogen)olytic enzymes from snake venom are able to induce hemorrhage, which results from proteolysis of basement membrane proteins [17, 18]. The hemorrhagic activity of FE27kDa showed a sunbursting radiating pattern of the hemorrhagic phenomenon beneath the skin [19]. However, the hemorrhagic activity of FE-27kDa was relatively weaker than that of FE-54kDa [11]. When FE-54kDa at dosages of 40 μg, 20 μg, 10 μg, and 5 μg was administered to the mice, the areas of the hemorrhage zones were 1.1 cm2, 0.9 cm2, 0.36 cm2, and 0.38 cm2, respectively (results not shown). These results showed that FE-54kDa had a higher hemorrhagic activity than FE-27kDa because a hemorrhage zone was still formed at a dosage of 5 μg FE-54kDa. The strong gelatin hydrolytic activity of FE- 54kDa might lead to the formation of a hemorrhage zone at low concentrations. When the activities in fibrinolysis, gelatin hydrolysis and hemorrhage were compared, FE-54kDa was overall more active than FE- 27kDa.
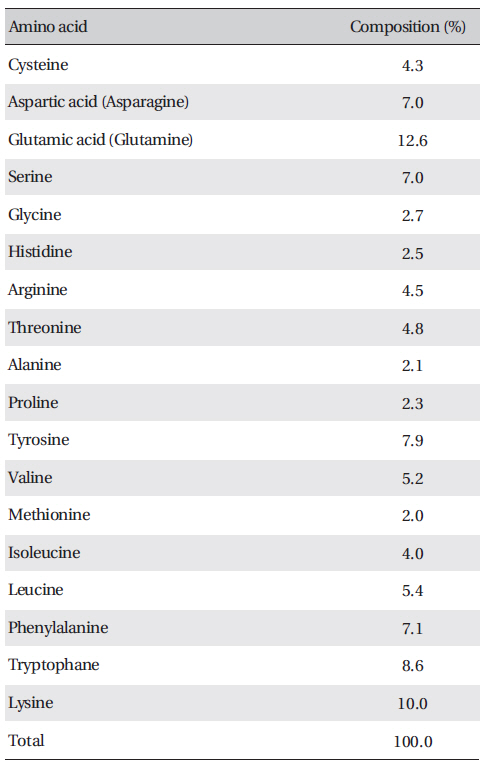
The amino acid composition of FE-27kDa is shown in (Table 3) FE-27kDa contained cysteine at a concentration of 4.3%, which was in accord with inhibition of fibrinolysis by a reducing agent, such as cysteine and dithiothreitol. Among the amino acids, lysine and glutamic acid (glutamine) were major components with concentrations of more than 10%.
Many studies have been reported the isolation of a non-hemorrhagic fibrin(ogen)olytic enzyme that could be presumably used to treat obstructive thrombosis and acute stroke resulting from cardiovascular diseases. Didisheim and Lewis [20] proposed that the activity to hydrolyze fibrin clots of the snake venom from
This study showed that FE-27kDa, a serine proteinase, had fibrin(ogen)olytic activity and gelatin hydrolytic activity and caused hemorrhage beneath the skin of a mouse. The results suggested that the possible use of FE-27kDa as a pharmacopuncture agent in Oriental medicine should be limited due to its relatively low fibrinolytic activity and potential side effects of hemorrhage.