The quantitative evaluation of soil respiration following fertilizer application is a key process for understanding carbon dynamics in forest ecosystems, because nitrogen fertilization showed a positive effect on the soil carbon pool among forest management practices (Johnson and Curtis 2001). However, fertilizer application in forest soils has shown to increase, decrease, or to have no effect on soil respiration. Gallardo and Schlesinger (1994) reported an increase in soil respiration when nitrogen was added to forest soils in central North Carolina, while soil respiration was significantly lower for fertilized than for unfertilized plots due to reduced root (Haynes and Gower 1995, Olsson et al. 2005) and microbial respiration (Phillip and Fahey 2007). Also, nitrogen fertilization had a significant negative effect on soil respiration in a hardwood plantation, but no effect was observed in a coniferous plantation (Lee and Jose 2003, Samuelson et al. 2009). Since soil respiration results from two main sources, root respiration (autotrophic respiration) and the microbial decomposition of organic matter (heterotrophic respiration), these conflicting reports could be due to the result of fertilizer- induced differences in carbon fixation and allocation patterns among tree species, or soil-specific differences in the microbial decomposition of organic matter (Raich and Tufekcioglu 2000, Lee and Jose 2003) and mycorrhizal colonization (Phillips and Fahey 2007).
Fertilizer application was found to result in a decrease or an increase in heterotrophic respiration. For example, heterotrophic respiration was reduced after nitrogen applications in pine forests (Franklin et al. 2003), while root respiration would be expected to increase along with an increase in forest production after fertilizer application (Bowden et al. 2004). Lee and Jose (2003) suggested that reductions in heterotrophic respirations following fertilizer application could be offset by increases of fine root production. In contrast to this result, fertilization increased heterotrophic respiration, microbial biomass carbon, and microbial activity in a loblolly pine plantation (Samuelson et al. 2009).
Total soil and heterotrophic respiration responded differently to environmental variables such as nutrient availability, soil water content and soil temperature (Raich and Tufekcioglu 2000, Noh et al. 2010, Bond-Lamberty et al. 2011). Although these factors are potentially important regulators of total soil and heterotrophic respiration, experimental data in relation to fertilizer application or soil temperature are limited in Korea forest ecosystems. Red pine (
This study was conducted in approximately 40-year-
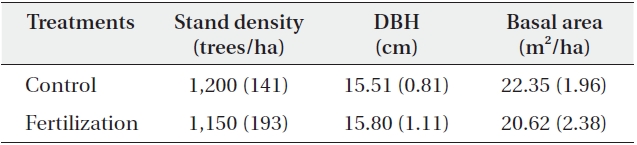
[Table 1.] General stand characteristics of the study plots (N = 6)
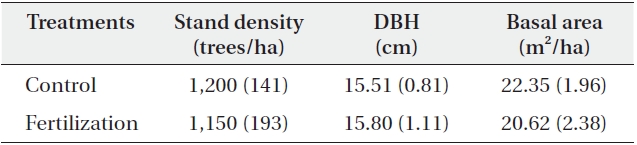
General stand characteristics of the study plots (N = 6)
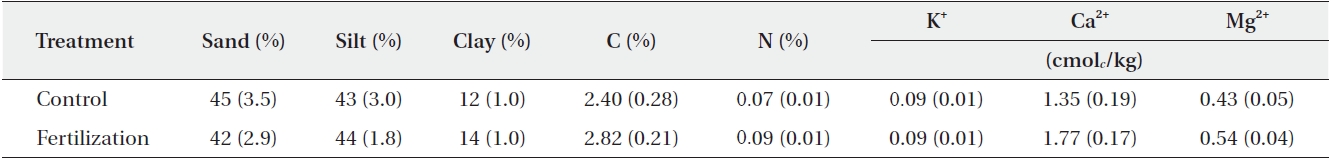
[Table 2.] Soil property before fertilizer application in study plots (N = 6)
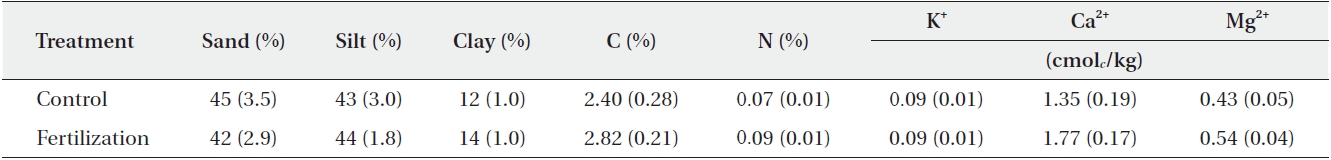
Soil property before fertilizer application in study plots (N = 6)
old natural red pine stands in the Wola National Experimental Forest, which is administered by the Southern Forest Research Center, Korea Forest Research Institute. The average annual precipitation and temperature in this area are 1,490 mm/y and 13.1℃, respectively. The soil is a slightly dry, dark-brown forest soil (mostly Inceptisol, United States Soil Classification System) originating from sandstone or shale with a silt loam texture. The site index of dominant pine trees indicates low forest productivity (site index, 8-10 at 20-year-old base age) suggesting poor soil fertility. The treatment plots were established on the same facing slopes and aspects under similar environmental conditions to minimize spatial variation in soil properties. The experimental design consisted of a completely randomized block design with 2 blocks (35°12′32″ N, 128°10′23″ E, 180 m; 35°12′26″ N, 128°10′25″ E, 195 m) involving 12 plots (plot size = 10 × 10 m): 2 treatments [fertilized, control] × 2 blocks × 3 replicated plots) in mature red pine stands, which were identified based on homogeneity between the sites. Fertilizer application (N:P:K = 113:150:37 kg/ha) was based on the guidelines of forest fertilization in Korea (Joo et al. 1983). Urea, fused superphosphate, and potassium chloride fertilizers were used as sources of N, P, and K, respectively, and were applied manually in 21 April 2011. Tree densities were similar between the fertilizer and the control treatments (Table 1). The mean diameter at breast height was 15.80 cm in the fertilizer and 15.51 cm in the control treatments, whereas the stand basal area was slightly higher in the control (22.35 m2/ha) than in the fertilizer (20.62 m2/ha) treatment (Table 1). The understory tree species in each stand were
A root exclusion collar with a trenching was used to separate heterotrophic respiration (Vogel and Valentine 2005, Bond-Lamberty et al. 2011) from total soil respiration. Trenching was performed by excavating the outside edges of a columnar soil of 50 cm diameter and 30 cm deep about one month (24 March 2011) prior to fertilizer application. The soil depth to 30 cm involved the bottom of B horizon and the top of C horizon in a shallow soil of
the study site. In addition, the trenching depth cut down most live roots. Polyvinyl chloride (PVC) collars (50 cm inner diameter, 30 cm high, 4 mm thick) were inserted into the columnar soil (total 12 plots:6 plots in control and 6 plots in fertilizer treatments) and were backfilled with excavated soil . Seedlings and herbaceous vegetation inside the collars were manually removed, while litter fall was retained within the collars during the study period.
In this study, we assumed that total soil respiration was regarded as soil CO2 efflux omitted from outside of the trenched location, while heterotrophic respiration was regarded as soil CO2 efflux omitted inside of the PVC collars in each plot (Lee et al. 2010). Four replicate measurements (2 inside PVC collars and 2 outside the trenched locations) of each plot were performed monthly between 1000 and 1230 h during the study period by using an infrared gas analyzer system (Model EGM-4 environmental gas monitor systems; PP Systems, Hitchin, UK) equipped with a flow-through closed soil respiration chamber (Model SRC-2; PP Systems). Soil temperature from each plot was measured at 8 cm depth adjacent to the soil respiration chamber, using a digital soil temperature probe (Summit SDT 200; Summit, Incheon, Korea).
Data were analyzed by two-way analysis of variance (ANOVA) to determine the significance of main effects (fertilizer treatments [F], trenching treatments [T]) and their interactions (F × T) at a significance level of
Soil CO2 efflux rates = B0eB1ST
, where
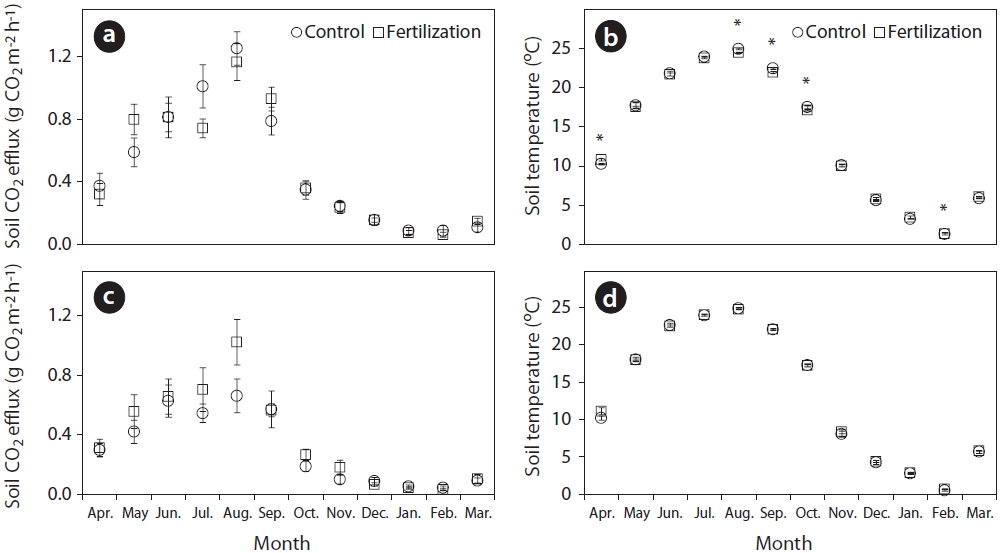
Monthly rates of total soil and heterotrophic respiration were not significantly affected (
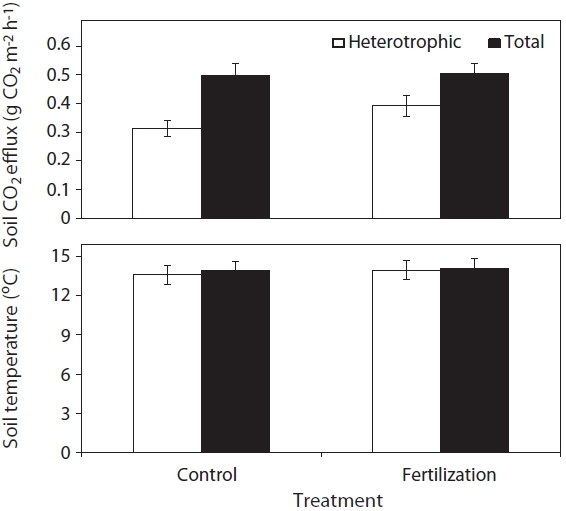
Annual mean total soil respiration rates in this study were similar between the fertilizer treatments (0.504 g CO2 m-2 h-1) and control treatments (0.501 g CO2 m-2 h-1) (Fig. 2). Many studies have observed decreases in soil respiration rates due to reduced microbial biomass (Lee and Jose 2003) and fine root production (Haynes and Gower 1995, Olsson et al. 2005, Phillips and Fahey 2007) because soil environmental changes in response to fertilizer application are closely related to microbial activity and nutrient availability (Lee and Jose 2003, Kim 2008). However, the decrease in the soil CO2 efflux following fertilizer application could be compensated for by the increased decomposition of organic matter with the change of carbon and nitrogen ratio in forest floor and mineral soil layers (Bolan et al. 1996, Kim et al. 2002, Kim 2008). In addition, heterotrophic respiration rates in the fertilizer (0.393 g CO2 m-2 h-1) represented 78% of total soil respiration rates, and 62% in the control (0.312 g CO2 m-2 h-1) treatments. Lower root respiration in fertilizer (0.111 g CO2 m-2 h-1) compared with the control (0.189 g CO2 m-2 h-1) treatments could be due to the decreased allocation of carbon to roots in response to increased nutrient availability (Haynes and Gower 1995, Phillips and Fahey 2007). Similarly, Samuelson et al. (2009) reported that fertilizer application increased heterotrophic respiration, microbial biomass, and carbon and microbial activities, with reduced fine root biomass. The proportion of heterotrophic respiration rates in the control treatments of this study was comparable to about 66% of total soil respiration in temperate coniferous forests in Korea (Lee et al. 2010).
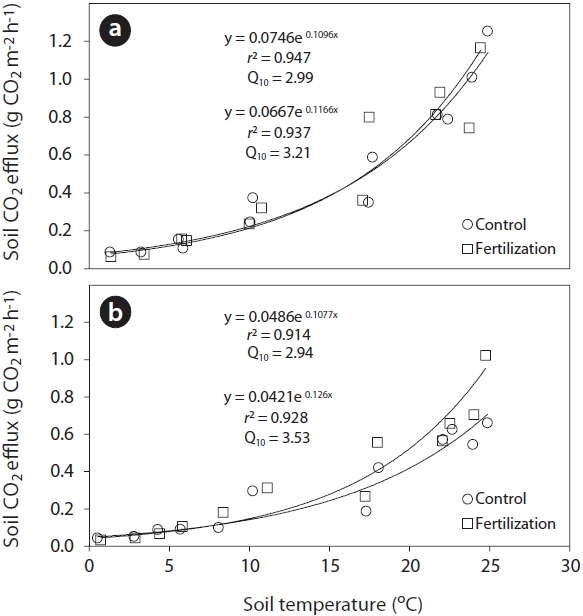
An exponential regression has been widely used to describe the relationship between soil respiration and temperature following fertilizer application (Bowden et al. 2004, Kim 2008). In this study, the exponential relationships between total soil or heterotrophic respiration rates and the corresponding soil temperature at a depth of 8 cm (Fig. 3) were highly significant (total soil respiration:
Sensitivity of soil respiration to soil temperature following trenching was commonly expressed by the coefficient
(2.94) treatments. The high
Fertilizer application induced changes in heterotrophic respiration, while total soil respiration rates were little affected in red pine stands. Both total soil and heterotrophic respiration rates in fertilizer and control treatment showed high sensitivity by soil temperature. Further long-term studies are needed to examine the controlling factors, such as root and microbial activities, related to total soil and heterotrophic respiration following fertilizer application.