Phylum Ciliophora Doflein, 1901
Subphylum Postciliodesmatophora Gerassimova and Servin, 1976
Class Heterotrichea Stein, 1859
Order Heterotrichida Stein, 1859
1*Family Condylostomatidae Kahl in Doflein and Reichenow, 1929
2*Genus Condylostoma Bory de Saint-Vincent, 1824
3*Condylostoma curva Burkovsky, 1970 (Table 1, Figs. 1,2)
Condylostoma curva Burkovsky, 1970: 58; Song et al., 2003: 457.
Condylostoma arenarium: Kahl, 1932: 455 (fig. 6 only); Dragesco and Dragesco-Kerneis, 1986: 393.
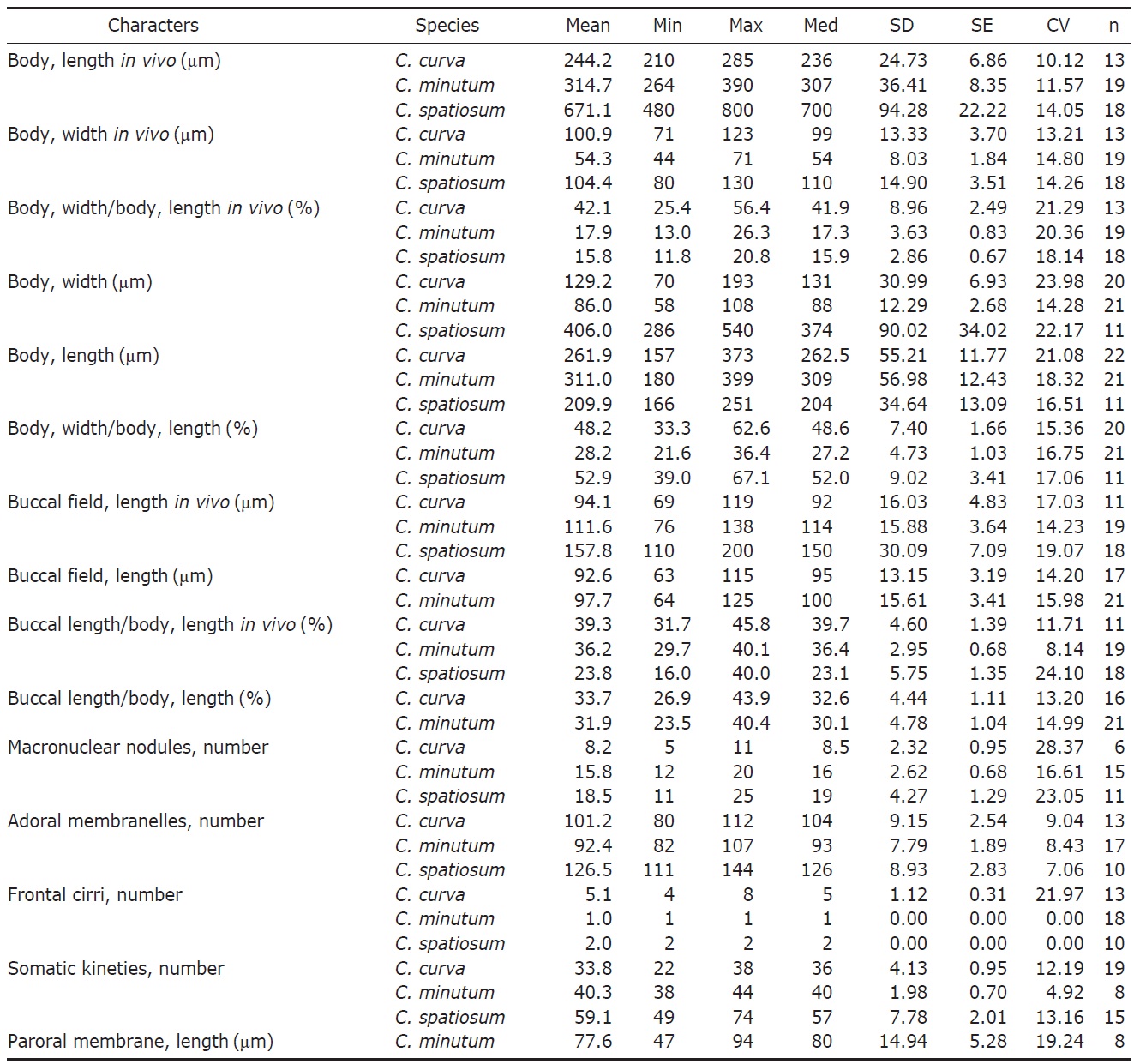
Material examined. Thirteen living and 22 protargol impregnated specimens were observed respectively and analyzed biometrically.
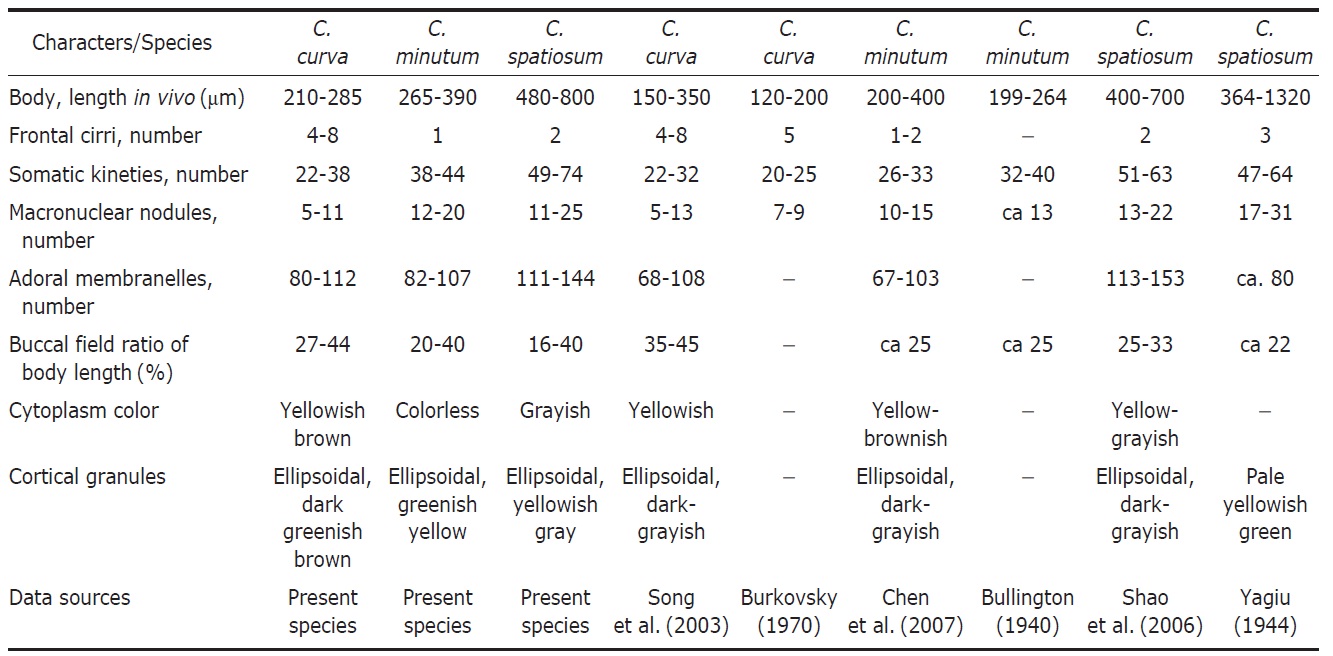
Diagnosis. Body size about 245×100 μm in vivo, oval to elongated ellipsoidal shaped, buccal cavity about 40% of body length, macronuclear moniliform with 5-11 nodules, yellowish brown cytoplasm, dark greenish brown cortical granules, 80-112 adoral membranelles, 4-8 frontal cirri, 22-38 somatic kineties.
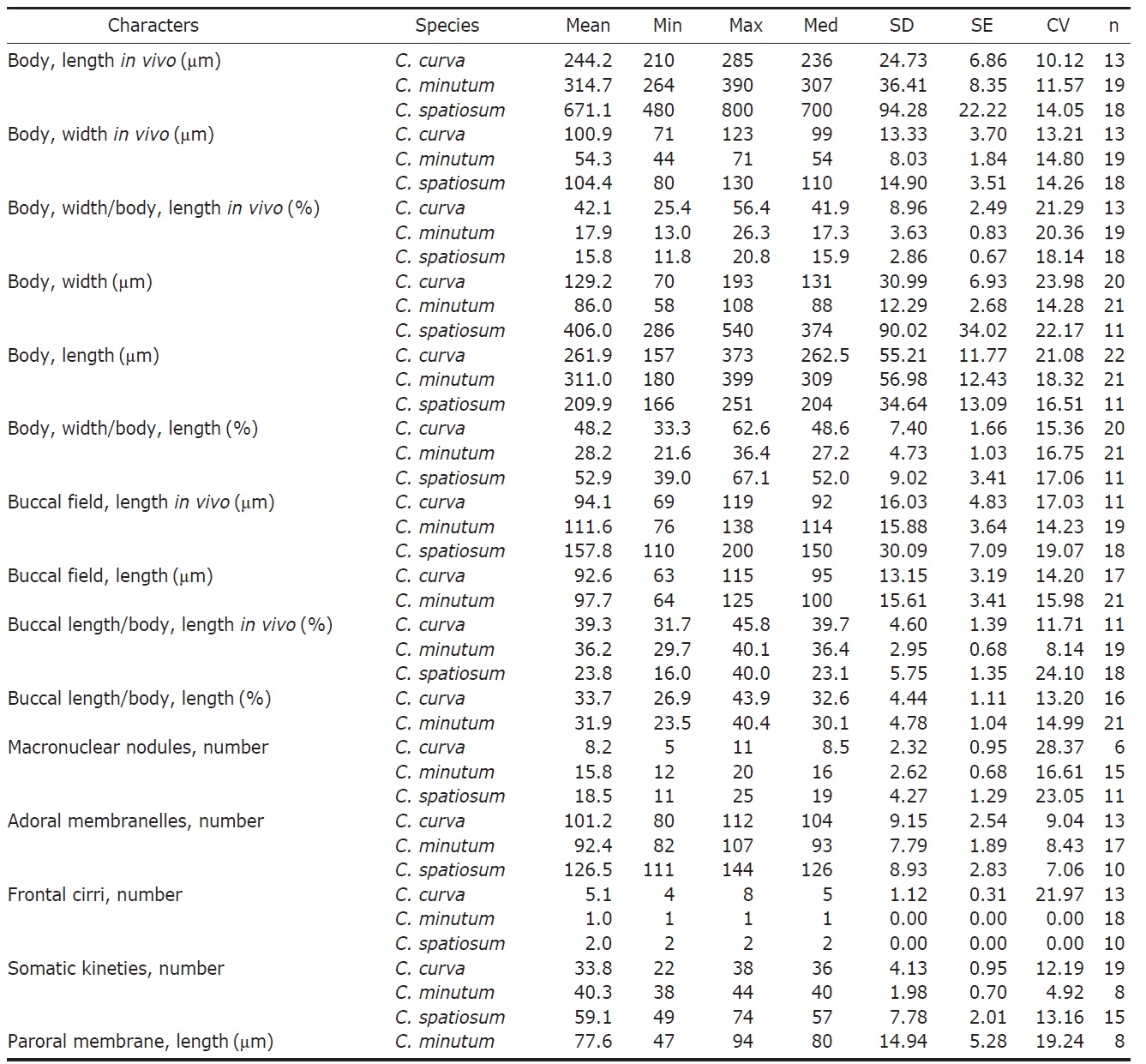
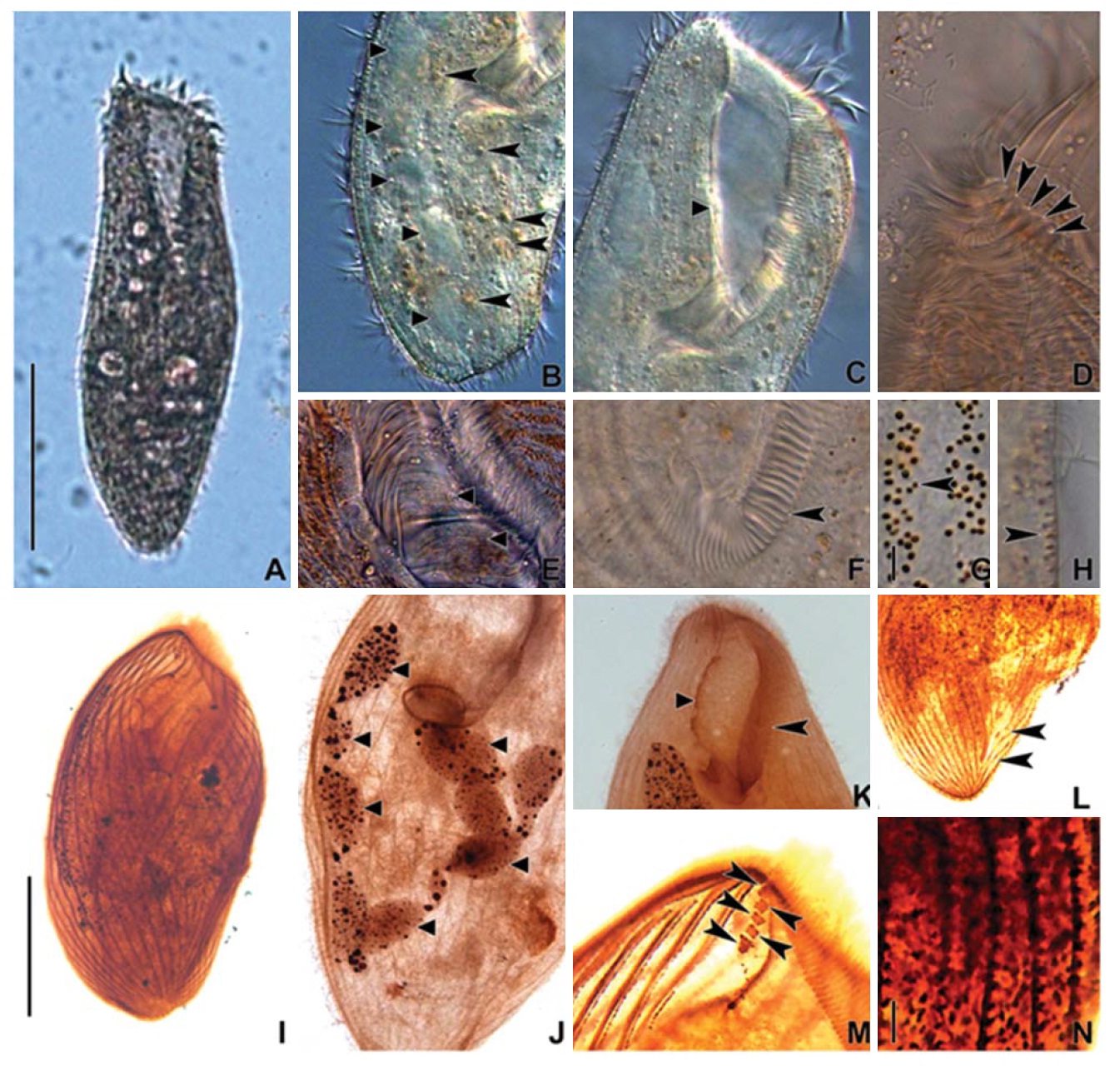
Description. Body size 210-285×70-125 μm, usually about 245×100 μm in vivo (Table 1). Shape elongated ellipsoidal, dorsoventrally slightly flattened, anterior end truncated and slanted left side, posterior end gradually tapered and narrowly rounded, widest in mid-body (Figs. 1A, 2A). Oral cavity fairly deep with transversely striated inner wall (Fig. 2E). Macronucleus aligned with the right margin of the body, distinctly nodulated and connected by fine threads, composed of 5-11 usually 8 nodules with many small nucleoli, anteriormost macronuclear nodule size about 25×15 μ m in impregnated preparations (Figs. 1A, C, 2B, J). No contractile vacuole. Cortex very flexible and contractile. Dark greenish brown cortical granules ellipsoidal, size about 0.5-1 μm, arranged 1-3 irregular rows in between kineties (Figs. 1F, G, 2G, H, N). Cytoplasm slightly yellow with some lipid-like droplets and food vacuoles (Fig. 2A, B). Movement moderately slow, usually gliding on bottom.
Somatic kineties arranged longitudinally 22-38 rows, usually commenced near the buccal field, formed suture posteriorly, consisted of dikinetids and somatic cilia about 10 μm long in vivo (Table 1, Figs. 1B, C, 2I, L). Four to eight frontal cirrus near apical end of right margin, on outside of buccal cavity and 15-20 μm long in vivo (Table 1, Figs. 1A, B, E, 2D, M).
The adoral zone of membranelles conspicuously covered the left side of buccal field, occupied 32-46% of the body length with the proximal portion extending spirally into the cytopharynx, consisting of 80-112 adoral membranelles (Table 1, Figs. 1A, B, E, 2C, F, K). The paroral membrane located on the inner side of buccal cavity, conspicuously long and smoothly undulated and composed of cilia about 25 μm in length in vivo (Figs. 1A, E, 2C, K).
Distribution. Africa (Benin), Asia (China, Korea), Europe (Germany, Russia).
Remarks. The accurate identification of Condylostoma species is difficult because they have high variations in many characters, fewer unique characters, overlapping of characteristics in many features, and inappropriate literature caused by previous insufficient descriptions (Kahl, 1932; Bullington, 1940; Villeneuve-Brachon, 1940; Yagiu, 1944; Faure-Fremiet, 1958; Burkovsky, 1970; Dragesco and Dragesco-Kerneis, 1986; Song et al., 2003; Shao et al., 2006; Chen et al., 2007).
We identified Condylostoma curva Burkovsky, 1970 based on three main characters of the body shape, the color of cortical granules and the pattern of frontal cirri. The original population of C. curva described by Burkovsky (1970) was
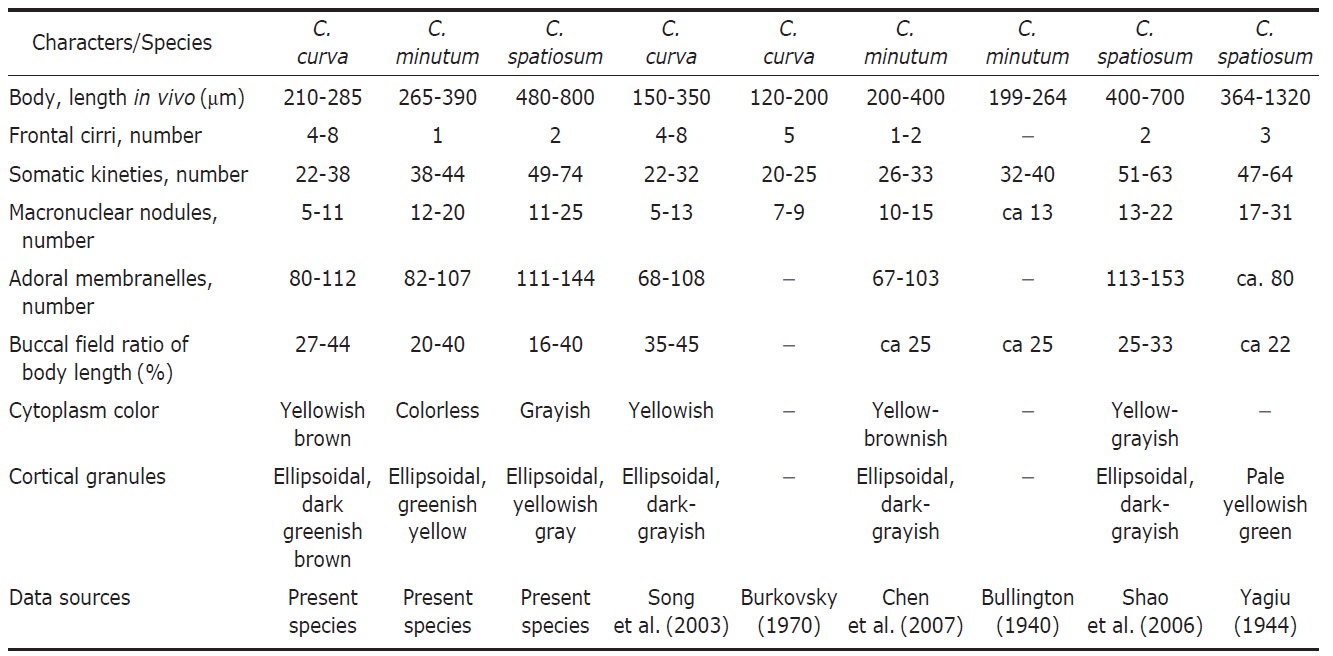
collected from Russia. We compared the Korean and the Russian populations those are slightly different in contractile vacuole (absent vs. present) and body size (210-285 μm vs. 120-200 μm). However, Song et al. (2003) mentioned that the presence of the contractile vacuole in Burkovsky (1970) was a food vacuole. The Chinese populations of Song et al. (2003) actually corresponded to the Korean population of C. curva in terms of all morphological key characters (Table 2, Fig. 7C-F).
The descriptions of the two populations of Condylostoma arenarium sensu Kahl (1932) and Dragesco and Dragesco-Kerneis (1986) are in agreement with C. curva. The one of Kahl’ s populations is similar to C. curva in the body size, body shape, shape of cortical granules, 4-5 frontal cirri and being from a brackish habitat. The African population of Dragesco and Dragesco-Kern?is is similar to C. curva in the body shape and the buccal field ratio (Fig. 7A, B) (Kahl, 1932; Dragesco and Dragesco-Kern?is, 1986; Song et al., 2003).
Condylostoma curva Burkovsky, 1970 differs from C. magnum Spiegel, 1926, C. spatiosum Ozaki and Yagiu in Yagiu,
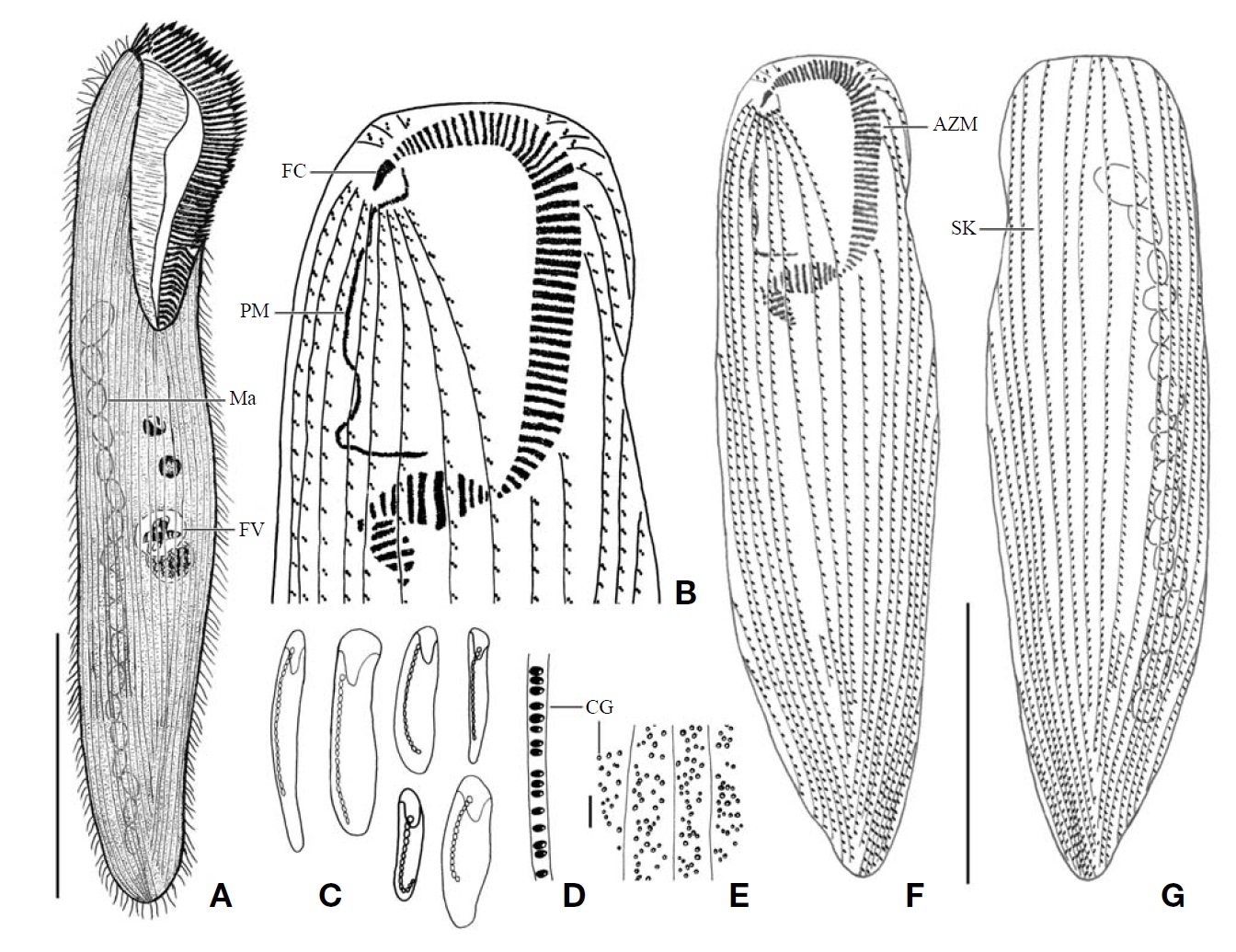
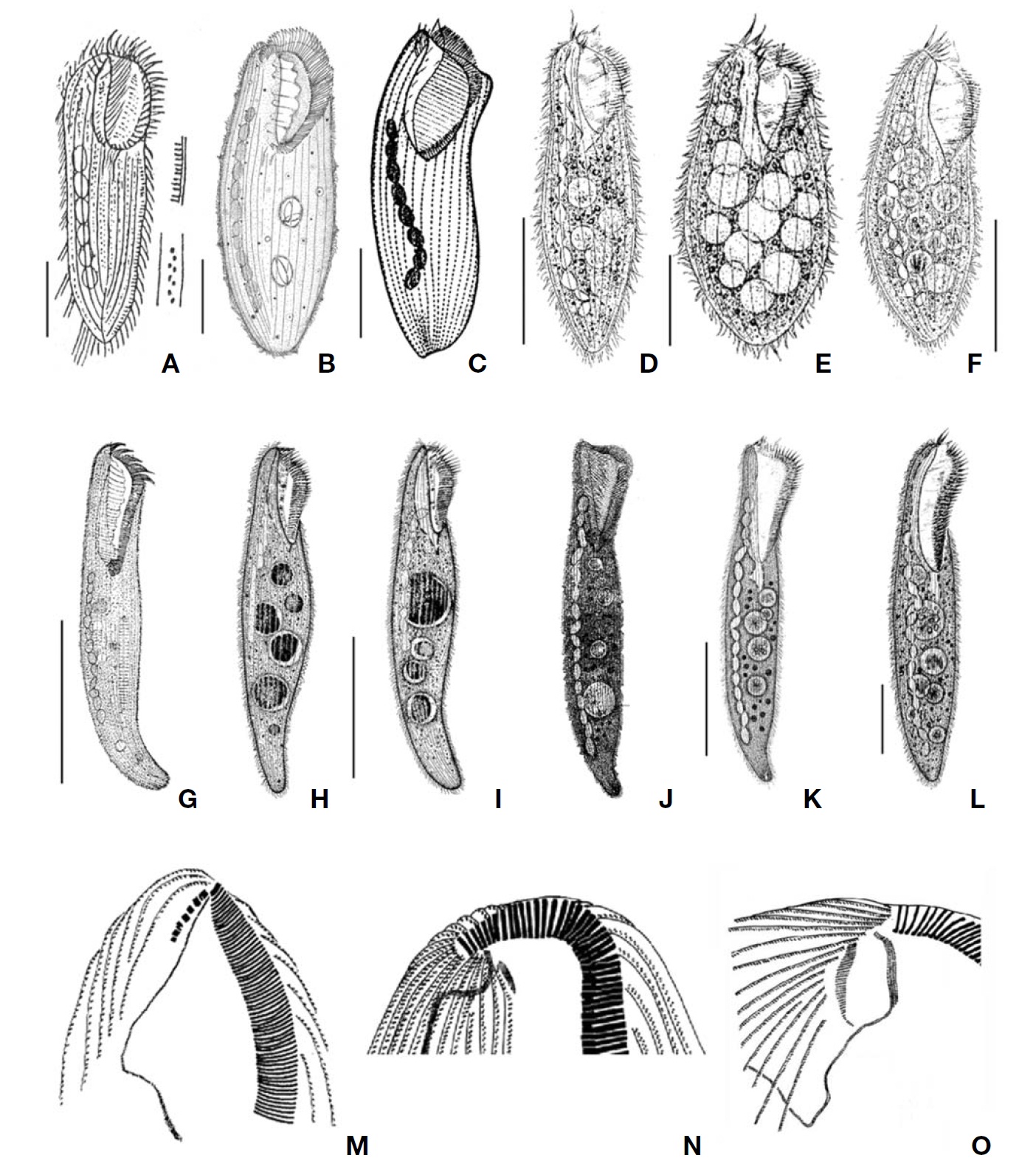
[Fig. 1.] Morphology and infraciliature of Condylostoma curva from live specimens (A, D, F, G) and after protargol impregnation (B, C, E). A, Ventral view of a typical individual; B, Ventral view of impregnated specimen; C, Dorsal view of impregnated specimen; D, Varied body shapes and macronuclear patterns; E, Ventral view of buccal field; F, Pattern of cortical granules; G, Lateral view of cortical granules. AZM, adoral zone of membranelles; CG, cortical granule; FC, frontal cirrus; FV, food vacuole; Ma, macronucleus; PM, paroral membrane; SK, somatic kineties. Scale bars: A, C=100 μm, F=5 μm.
[Fig. 2.] Microphotographs of Condylostoma curva from live specimens (A-H) and after protargol impregnation (I-N). A, Ventral view of a typical individual; B, Moniliform macronuclear nodules (triangular arrowheads) and cytoplasmic inclusion (arrowheads); C, Buccal field (triangular arrowhead); D, Frontal cirri (arrowheads); E, Buccal field to indicate the stripes of inner wall (triangular arrowheads); F, Proximal end of adoral zone of membranelles (arrowhead); G, Pattern of cortical granules (arrowhead); H, Lateral view of cortical granules (arrowhead); I, Ventral side view; J, Impregnated macronuclear nodules (triangular arrowheads); K, Paroral membrane (triangular arrowhead) and adoral zone of membranelles (arrowhead); L, Suture (arrowheads); M, Frontal cirri (arrowheads); N, Impregnated cortical granules. Scale bars: A, I=100 μm, G, N=5 μm.
1944 and C. granulosum Bullington, 1940 in body length in vivo (210-285 μm vs. 200-400 μm vs. 400-800 μm vs. 400-700 μm), number of frontal cirri (4-8 vs. 1-2 vs. 2 vs. 2 vs. 1-3), number of adoral membranelles (80-112 vs. 150-200 vs. 113-153 vs. 123-210) (Petz et al., 1995; Song and Wilbert, 1997; Song et al., 2003). Condylostoma curva is similar to C. minutum Bullington, 1940 and C. arenarium Spiegel, 1926 in body length in vivo, number of somatic kineties, number of macronuclei. However, C. curva and C. minutum are different in number of frontal cirri (4-8 vs. 1-2); C. curva differs from C. arenarium in number of adoral membranelles (80-112 vs. 60-70) (Borror, 1963; Chen et al., 2007).
1*Condylostoma minutum Bullington, 1940 (Table 1, Figs. 3,4)
Condylostoma minutum Bullington, 1940: 193; Chen et al., 2007: 299.
Material examined. Twenty living and 21 protargol impregnated specimens were observed respectively and analyzed biometrically.
Diagnosis. Body size about 315×55 μm in vivo, elongated ellipsoidal shaped, buccal cavity about 35% of body length, macronucleus moniliform with 12-20 nodules, 82-107 adoral membranelles, 1 frontal cirrus, 38-44 somatic kineties.
Description. Body size 265-390×45-70 μm, usually about
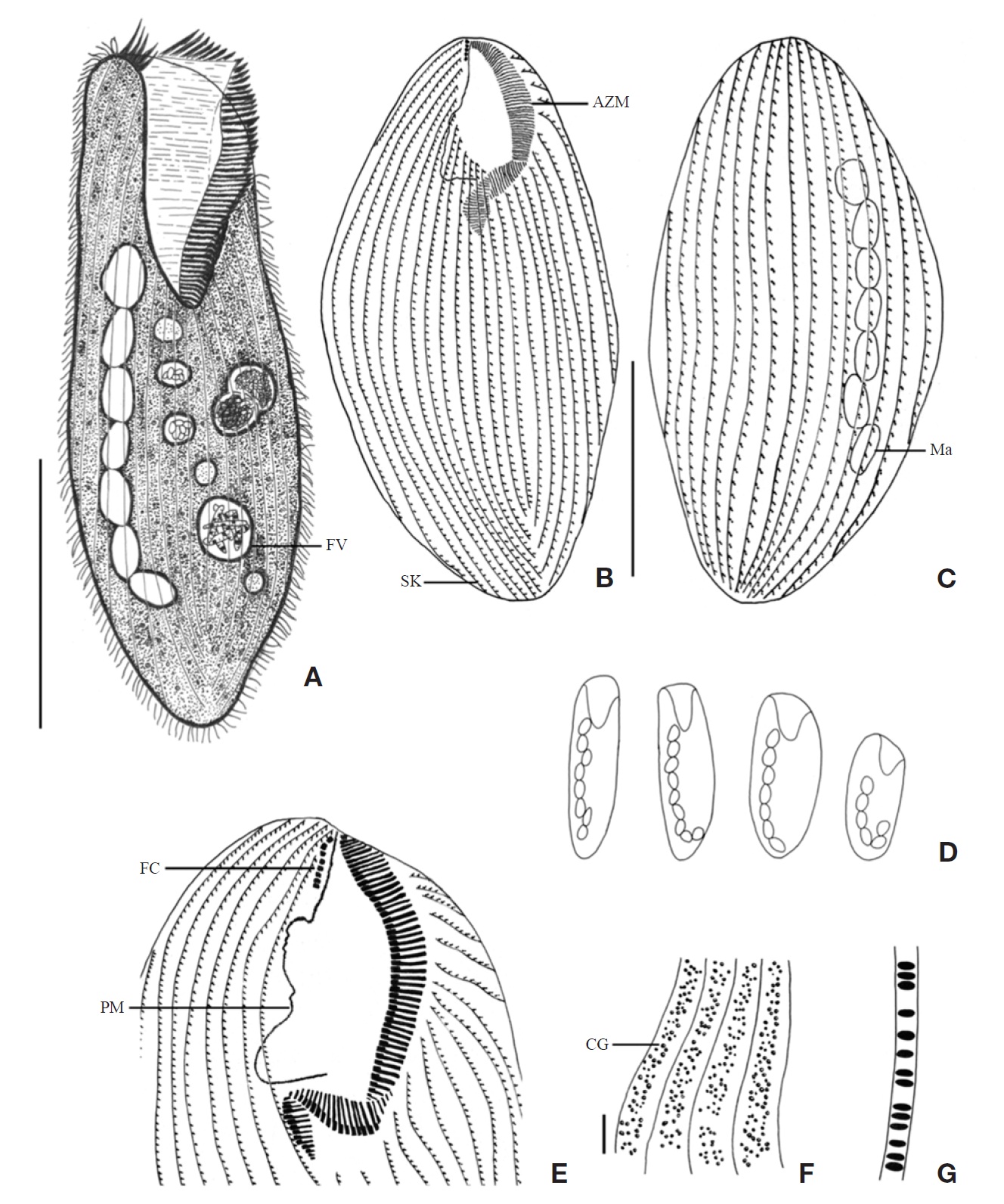
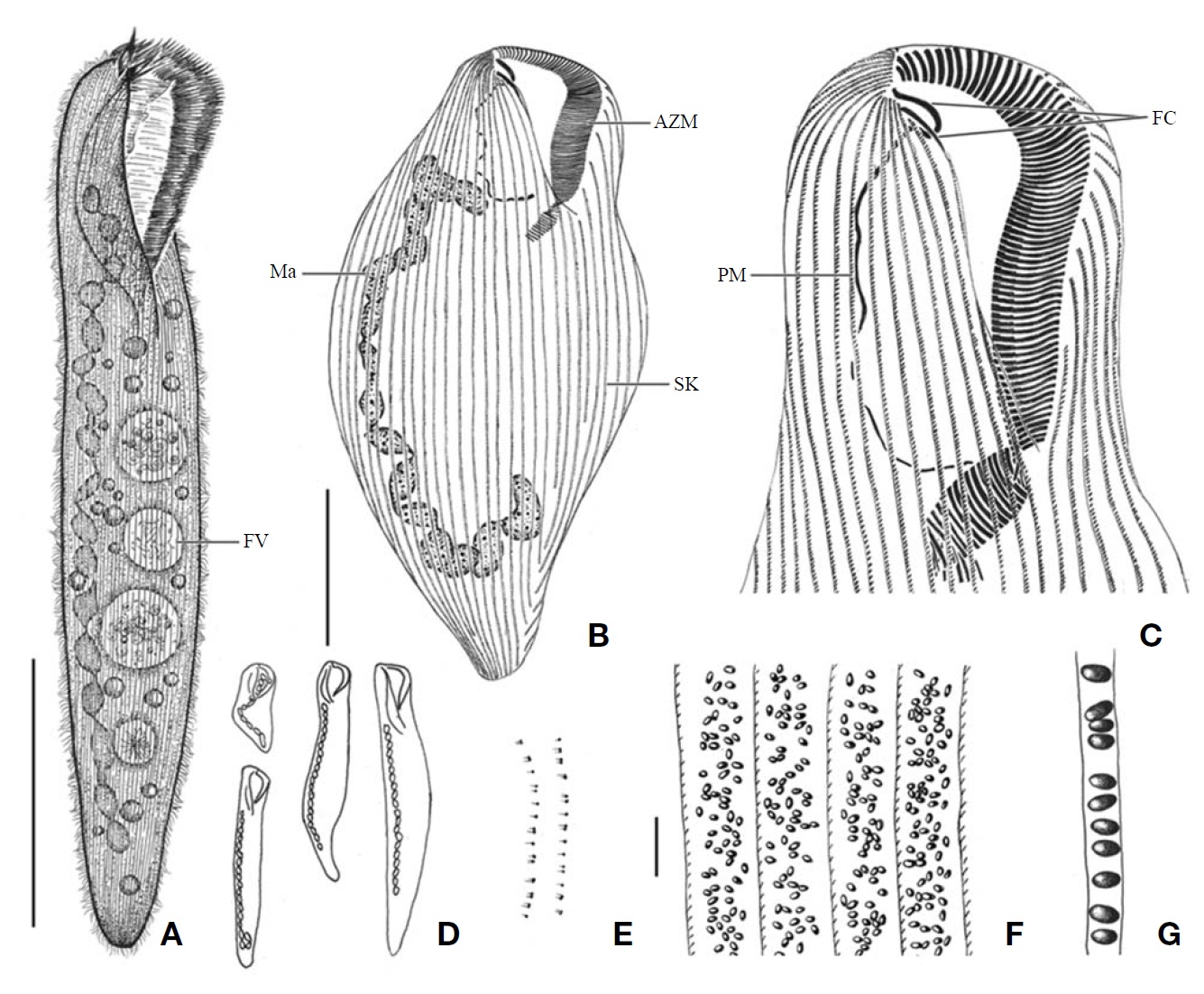
[Fig. 3.] Morphology and infraciliature of Condylostoma minutum from live specimens (A, C-E) and after protargol impregnation (B, F, G). A, Ventral view of a typical individual; B, Ventral view of buccal field; C, The various body shape and macronuclear nodules pattern; D, Lateral view of cortical granules; E, Ventral view of cortical granules; F, Ventral view of impregnated specimen; G, Dorsal view of impregnated specimen. AZM, adoral zone of membranelles; CG, cortical granule; FC, frontal cirrus; FV, food vacuole; Ma, macronucleus; PM, paroral membrane; SK, somatic kineties. Scale bars: A, G=100 μm, E=5 μm.
315×55 μm in vivo (Table 1). Shape elongated ellipsoidal, dorsoventrally slightly flattened, anterior end truncated and slanted left side, posterior end gradually tapered and narrowly rounded, widest in mid-body (Figs. 3A, C, 4A). Oral cavity fairly deep with transversely striated inner wall (Fig. 4A, G). Macronucleus aligned with the right margin of the body, distinctly nodulated and connected by fine threads, composed of 12-20 usually 16 nodules with many small nucleoli, anterior-most macronuclear nodule size about 28×18 μm in protargol impregnated preparations (Figs. 3A, G, 4B, J). No contractile vacuole. Cortex very flexible and contractile. Greenish yellow ellipsoidal cortical granules size about 1 μm, arranged irregular rows in between kineties (Figs. 3D, E, 4F,
[Fig. 4.] Microphotographs of Condylostoma minutum from live specimens (A-H) and after protargol impregnation (I-M). A, Ventral view of a typical individual; B, Moniliform macronuclear nodules (trianglular arrowheads); C, Paroral membrane (trianglular arrowhead); D, Food vacuole (trianglular arrowhead); E, Frontal cirrus (trianglular arrowhead); F, H, Cortical granules; G, Striated inner wall on buccal cavity (arrows); I, Ventral side view; J, Moniliform macronucleus (trianglular arrowheads); K, Suture of posterior end (trianglular arrowheads); L, To show adoral zone of membranelles (arrow), frontal cirrus (trianglular arrowhead), paroral membrane (arrowhead); M, Impregnated cortical granules between somatic kineties. Scale bars: A, I=100 μm, F, H, M=5 μm.
H, M). Cytoplasm colorless with some food vacuoles (Figs. 3A, 4D). Movement moderately slow, usually gliding on bottom.
Somatic kineties arranged longitudinally 38-44 rows, usually commenced near the buccal field, formed suture posteriorly, consisted of dikinetids and somatic cilia about 10 μm long in vivo (Figs. 3F, G, 4K). One frontal cirrus near apical end of right margin, on outside of buccal cavity and 15-20 μm long in vivo (Figs. 3A, B, F, 4E, L).
The adoral zone of membranelles conspicuously covered the left side of the buccal field, occupied 30-40% of the body length, with the proximal portion extending spirally into the cytopharynx, consisting of 82-107 adoral membranelles (Figs. 3A, B, F, 4A, I, L). The paroral membrane located on the
[Fig. 5.] Morphology and infraciliature of Condylostoma spatiosum from live specimens (A, D-G) and after protargol impregnation (B, C). A, Ventral view of a typical individual; B, Ventral view of impregnated specimen; C, Ventral view of buccal field; D, Various body shape and macronuclear nodules pattern; E, Infraciliature of somatic dikinetids; F, Pattern of cortical granules; G, Lateral view of cortical granules. AZM, adoral zone of membranelles; FC, frontal cirrus; FV, food vacuole; Ma, macronucleus; PM, paroral membrane; SK, somatic kineties. Scale bars: A=200 μm, B=100 μm, F=5 μm.
inner side of the buccal cavity, total length 47-94 μm in impregnated preparations, conspicuously long and smoothly undulated with a cilia about 25 μm in length in vivo (Figs. 3B, 4C).
Distribution. Asia (China, Korea), South America (Mexico).
Remarks. The Korean population of Condylostoma minutum closely resembles the Gulf of Mexico and Chinese populations of it with respect to body size, number of fragments of the macronucleus, number of adoral membranelles, ratio of adoral zone of membranelles. However, this Korean population differs from the Chinese population in number of somatic kineties (38-44 vs. 26-33) and number of frontal cirri (1 vs. 1-2) (Table 2, Fig. 7G-I) (Bullington, 1940; Chen et al., 2007).
Condylostoma minutum differs from C. magnum and C. spatiosum in body length in vivo (265-390 μm vs. 400-800 μm vs. 400-700 μm), number of frontal cirri (1 vs. 2 vs. 2), number of somatic kineties (38-44 vs. 47-56 vs. 51-63), number of adoral membranelles (82-107 vs. 150-200 vs. 113-153) (Song and Wilbert, 1997; Chen et al., 2007).
1*Condylostoma spatiosum Ozaki and Yagiu in Yagiu, 1944 (Table 1, Figs. 5,6)
Condylostoma spatiosum Ozaki and Yagiu in Yagiu, 1944: 163; Shao et al., 2006: 11; Chen et al., 2007: 306; Wilbert and Song, 2008: 990.
Material examined. Eighteen living and 11 protargol impregnated specimens were observed respectively and analyzed biometrically.
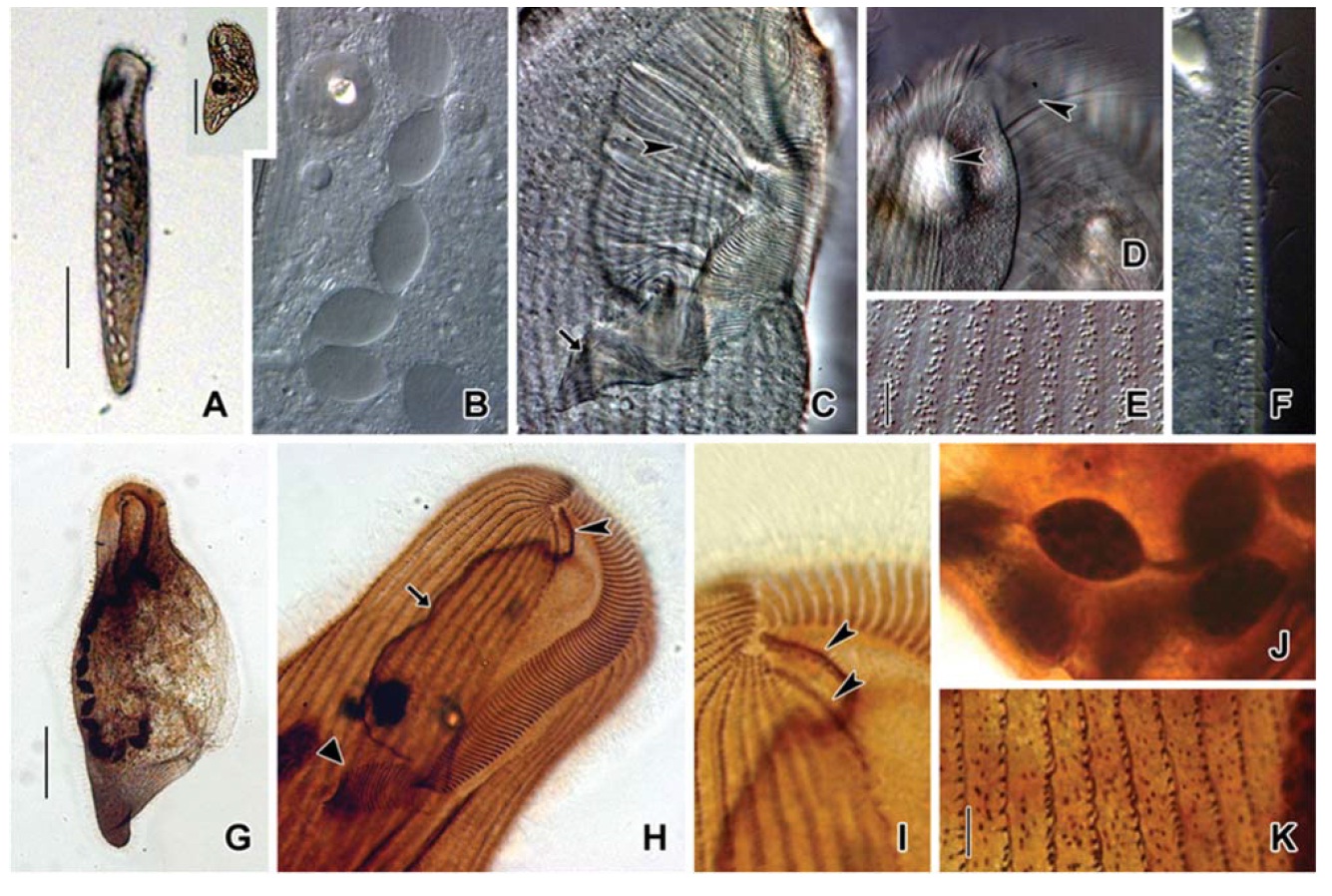
[Fig. 6.] Microphotographs of Condylostoma spatiosum from live specimens (A-F) and after protargol impregnation (G-K). A, Ventral side view; B, Moniliform macronuclear nodules; C, Buccal field to indicate the stripes of inner wall (arrowhead) and the cytopharynx (arrow); D, Location of frontal cirri (arrowheads); E, Pattern of cortical granules; F, Lateral view of cortical granules; G, Ventral side view in impregnated specimen; H, Ventral view of frontal cirri (arrowhead), paroral membrane (arrow) and cytopharynx (triangular arrowhead); I, Location of frontal cirri (arrowheads); J, Macronuclear nodules and threads; K, Cortical granules in impregnated specimen. Scale bars: A=200 μm, Inset in A=200 μm, E, K=5 μm, G=100 μm.
Diagnosis. Body size about 670×105 μm in vivo, elongated ellipsoidal shaped, buccal cavity about 25% of body length, macronucleus moniliform with 11-25 nodules, 111-144 adoral membranelles, 2 frontal cirri, 49-74 somatic kineties.
Description. Body size of 480-800×80-130 μm, usually about 670×105 μm in vivo (Table 1). Shape elongated ellipsoidal, dorsoventrally slightly flattened, anterior end truncated and slanted left side, posterior end gradually tapered and narrowly rounded, body margin slightly parallel (Figs. 5A, D, 6A). Oral cavity fairly deep with transversely striated inner wall (Figs. 5A, 6C). Macronucleus aligned usually with the right margin of the body, distinctly nodulated and connected by threads, composed of 11-25 usually 19 nodules with many small nucleoli in impregnated preparations (Figs. 5A, B, 6B, J). No contractile vacuole. Cortex very flexible and contractile. Slightly yellowish ellipsoidal cortical gran-
[Fig. 7.] A-F, M, Condylostoma curva Burkovsky, 1970 (A, from Kahl, 1932; B, from Dragesco J, Dragesco-Kern?is A, 1986; C, from Burkovsky, 1970; D-F, from Song et al., 2003); M, The apparatus of frontal cirri (from Song et al., 2003); G-L, N, C. minutum Bullington, 1940 (G, from Bullington, 1940; H, I, Chen et al., 2007); N, The apparatus of frontal cirrus (from Chen et al., 2007); JL, O, C. spatiosum Ozaki and Yagiu in Yagiu, 1944 (J, from Ozaki and Yagiu in Yagiu, 1944; K, L, from Shao et al., 2006); O, The apparatus of frontal cirri (from Chen et al., 2007). Scale bars: A-C, E, F=50 μm, D, G, I, L=100 μm, K=200 μm.
ules about 0.5-0.8×0.7-1.5 μm in size and about 1-5 irregular rows in between kineties (Figs. 5F, G, 6E, F, K). Cytoplasm colorless with some small inclusions 4 μm in vivo, lipid-like droplets and occasionally large food vacuoles (Figs. 5A, 6A). Movement moderately slow, usually gliding on bottom.
Somatic kineties arranged longitudinally in 49-74 rows, mostly commenced near the buccal field, formed suture posteriorly, consisted of dikinetids and somatic cilia about 12μm in length in vivo (Fig. 5B, E). Frontal cirri near the apical end of right margin, two membrane-like cirri were located, respectively, on outside of the buccal cavity, about 40 μm in length in vivo and on the apical end of the paroral membrane and connected with paroral, about 20 μm in length in vivo (Figs. 5C, 6D, H, I, 7O).
The adoral zone of membranelles which conspicuously covered the left side of the buccal field, occupied 16-40% of the body length, the proximal portion extending spirally into the cytopharynx, consisting of 111-144 adoral membranelles (Figs. 5A, C, 6C, H). The paroral membrane located on the inner side of the buccal cavity in impregnated preparations, was conspicuously long and smoothly undulated (Figs. 5C, 6H).
Distribution. Asia (China, Japan, Korea), Antarctica
Remarks. This Korean population of Condylostoma spatiosum Ozaki and Yagiu in Yagiu, 1944 closely resembles with the Chinese and Japanese populations which have similar characters in body size (480-800 vs. 400-700 vs. 364-1,320), number of frontal cirri (2 vs. 2 vs. 3), and number of somatic kineties (49-74 vs. 51-63 vs. 47-64). The number of adoral membranelles is conspicuously less than the others in the Japanese population. The colors of cortical granules are different (Korean, yellowish gray; Chinese, dark gray; Japanese, yellowish green). However, this can be a variable character because it is depends on the subjective or the microscopic field. The Korean population has a variation in the pattern of cortical granules dense or loose (Table 2, Fig. 7J-L) (Yagiu, 1944; Shao et al., 2006; Chen et al., 2007).
This present species is different from the Condylostoma magnum Spiegel, 1926 in number of adoral membranelles (111-144 vs. 150-200) (Song and Wilbert, 1997; Shao et al., 2006).
Korean name: 1* 터진입섬모충과, 2*터진입섬모충속, 3*노랑터진입섬모충
Korean name: 1* 작은터진입섬모충
Korean name: 1* 넓은터진입섬모충