In the marine ecosystem, ciliates have been recognized as a dominant group in microzooplankton and known to serve as an effective link between the microbial loop and metazoan food web (Maeda and Carey, 1985; Maeda, 1986; Agatha, 2011). Recent investigations show that aloricate oligotrich ciliates occur in high numbers and consume significant quantities of autotrophic and heterotrophic microbial production (Quevedo and Anad?n, 2000; Fileman and Leakey, 2005; Bojanic et al., 2006). The oligotrichs have been recorded with Butschli’s classification scheme since 1889 and about 300 species have been reported (excluding tintinnids) up to the present (Agatha, 2004a; Agatha and Str?der-Kypke, 2007; Xu et al., 2009). However, the taxonomic composition and ecological characteristics of the oligotrichs in Korean coastal waters are poorly known. To date, only 18 aloricate oligotrichs were described (only about 6% of recorded species in the world) (Suzuki and Han, 2000; Jeong et al., 2004; Moon et al., 2004; Kim et al., 2005, 2008, 2010; Ma and Choi, 2005; Lee et al., 2006, 2011).
The genus
1932; Maeda and Carey, 1985; Lynn et al., 1988; Montagnes et al., 1988, 1990, 2002; Montagnes and Lynn, 1991; Petz and Foissner, 1992; Lynn and Gilron, 1993; Martin and Montagnes, 1993; Petz et al., 1995; Agatha and Riedel-Lorje , 1997; Kim and Taniguchi, 1997; Montagnes and Humphrey, 1998; Song and Bradbury, 1998; Lei et al., 1999; Song et al., 2000; Agatha, 2003; Granda and Montagnes, 2003; Modeo et al., 2003; Agatha, 2004b; Agatha et al., 2005; Xu et al., 2006, 2007, 2009; McManus et al., 2010; Liu et al., 2011). Several studies have described with their ecological data concerning the occurrences at different water temperatures and salinities, as well as the morphologic data. According to their studies,
As a serial report on the taxonomical study of oligotrich ciliates in Korean coastal waters, we describe morphologically six
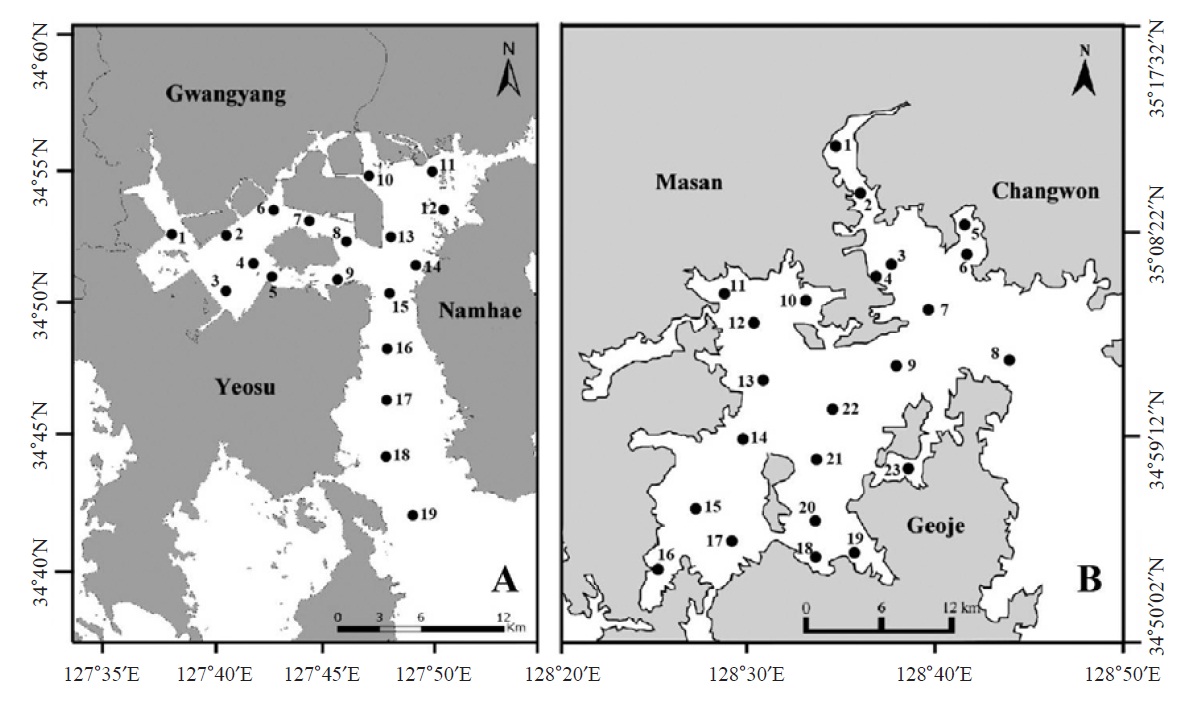
Ciliate plankton was investigated seasonally in Gwangyang Bay (19 sites in Fig. 1A) and Jinhae Bay (23 sites in Fig. 1B) from August 2010 to February 2012 (August and November in 2010; February, May, August, and November in 2011; February in 2012). To collect ciliate cells, surface sea water was sampled by a bucket and fixed using Bouin’s solution in the field (at final concentration 10%, v/v). Water temperature and salinity were measured
>
Cell identification and counting
The water samples fixed were analyzed by the quantitative protargol stain (QPS), following the protocol of Montagnes and Lynn (1993). In brief, 50 mL of fixed water samples were filtered through 0.8 μm in pore size membrane filters (Startorius Stedin Biotech, Goettingen, Germany) and filtered samples were covered with a thin layer of agar to avoid the loss of cells during the next step of preparation. Then, the membranes were bleached, protargol impregnated and mounted. Under a light-field microscope (Zeiss Axioskop II; Carl Ziess, Jena, Germany), the protargol impregnated cells were identified and counted at the magnification of 400-1,600×.
The terminology and classification for the ciliate taxonomy are according to Montagnes and Lynn (1991) and Agatha (2004a) respectively.
Class Oligotrichea Butschli, 1889
Subclass Oligotrichia Butschli, 1889
Order Oligotrichida Butschli, 1889
Family Strombidiidae Faure-Fremiet, 1970
1*Genus Strombidium Claparede and Lachmann, 1859
2*Strombidum emergens (Leegaard, 1915) Kahl, 1932 (Table 1, Figs. 2,3)
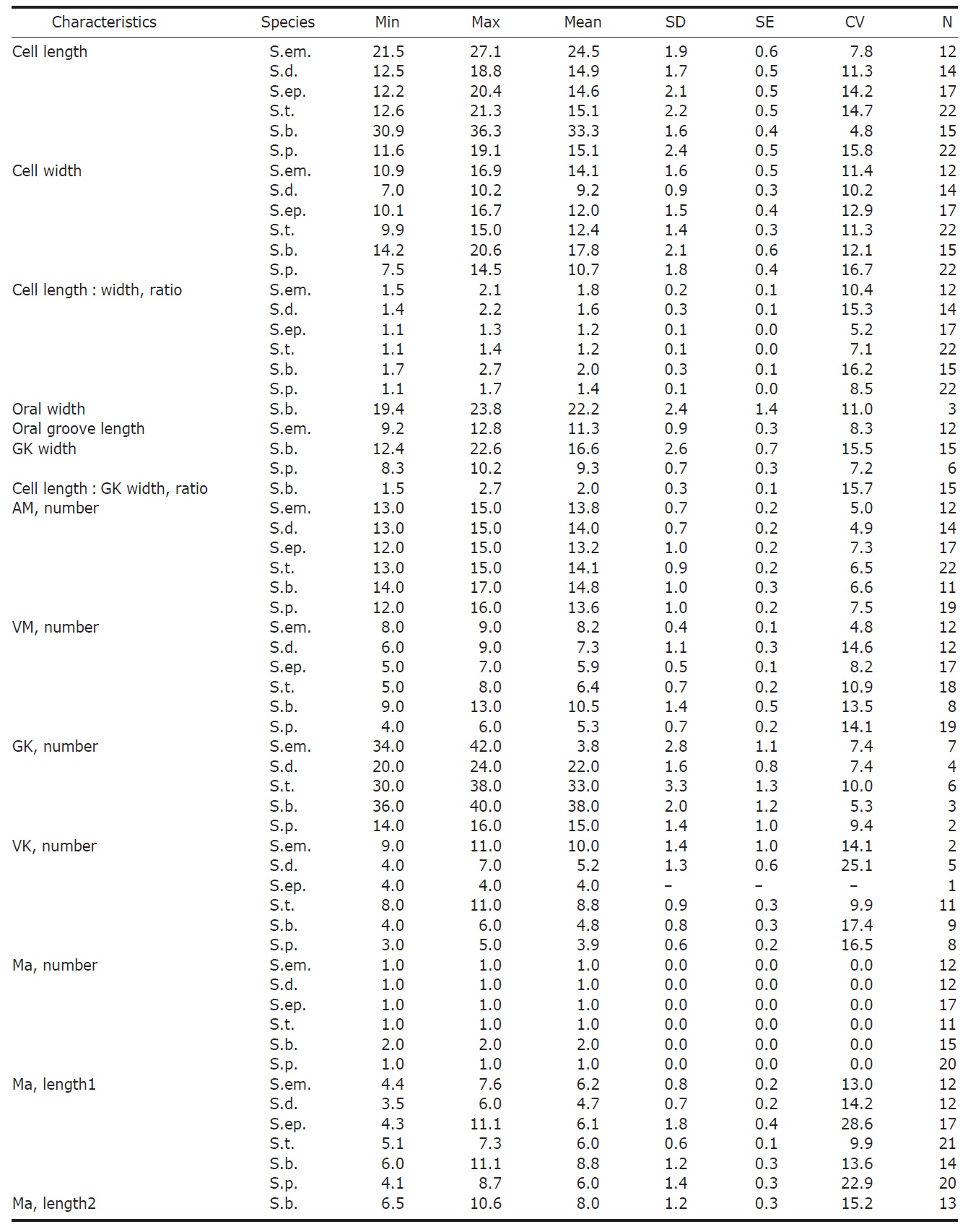
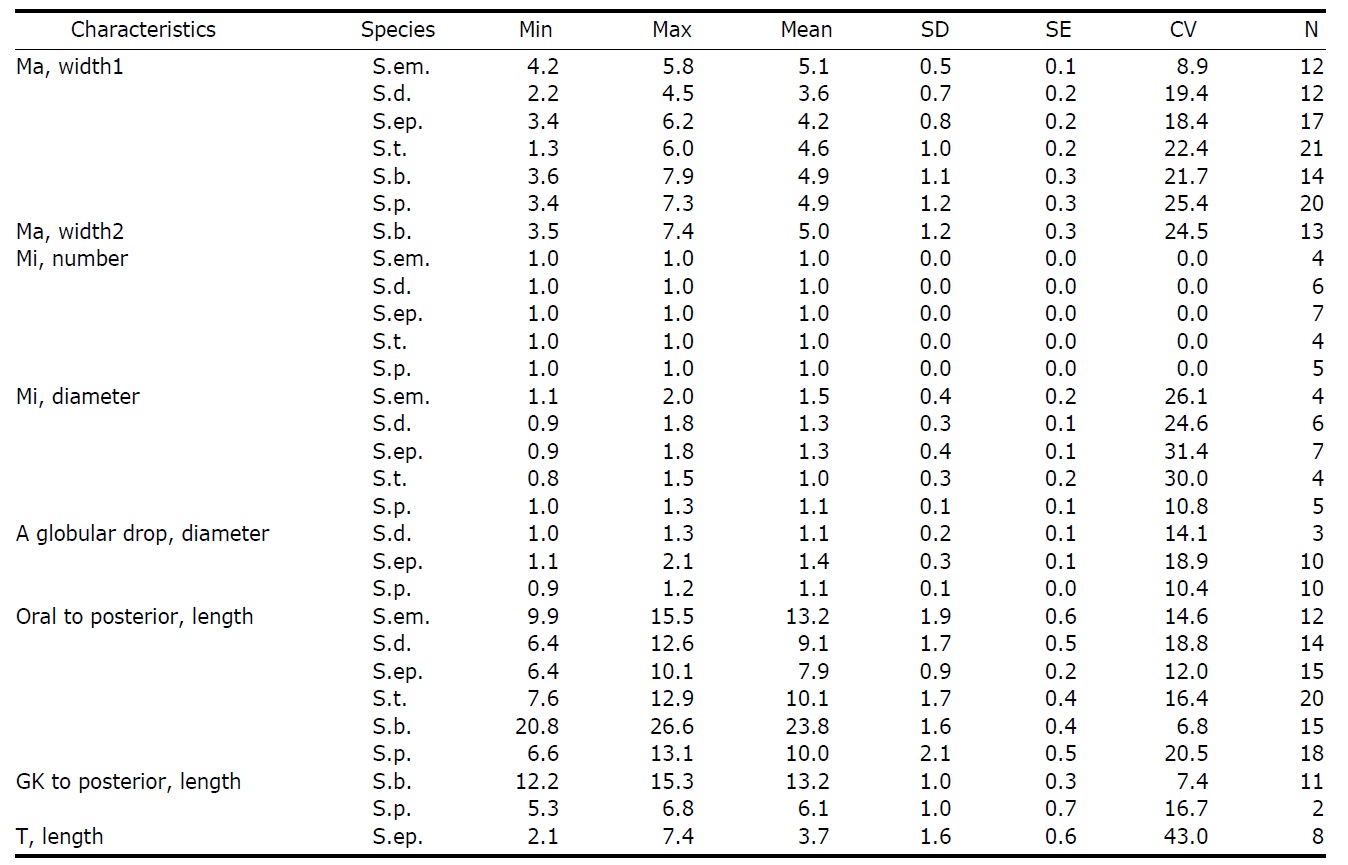
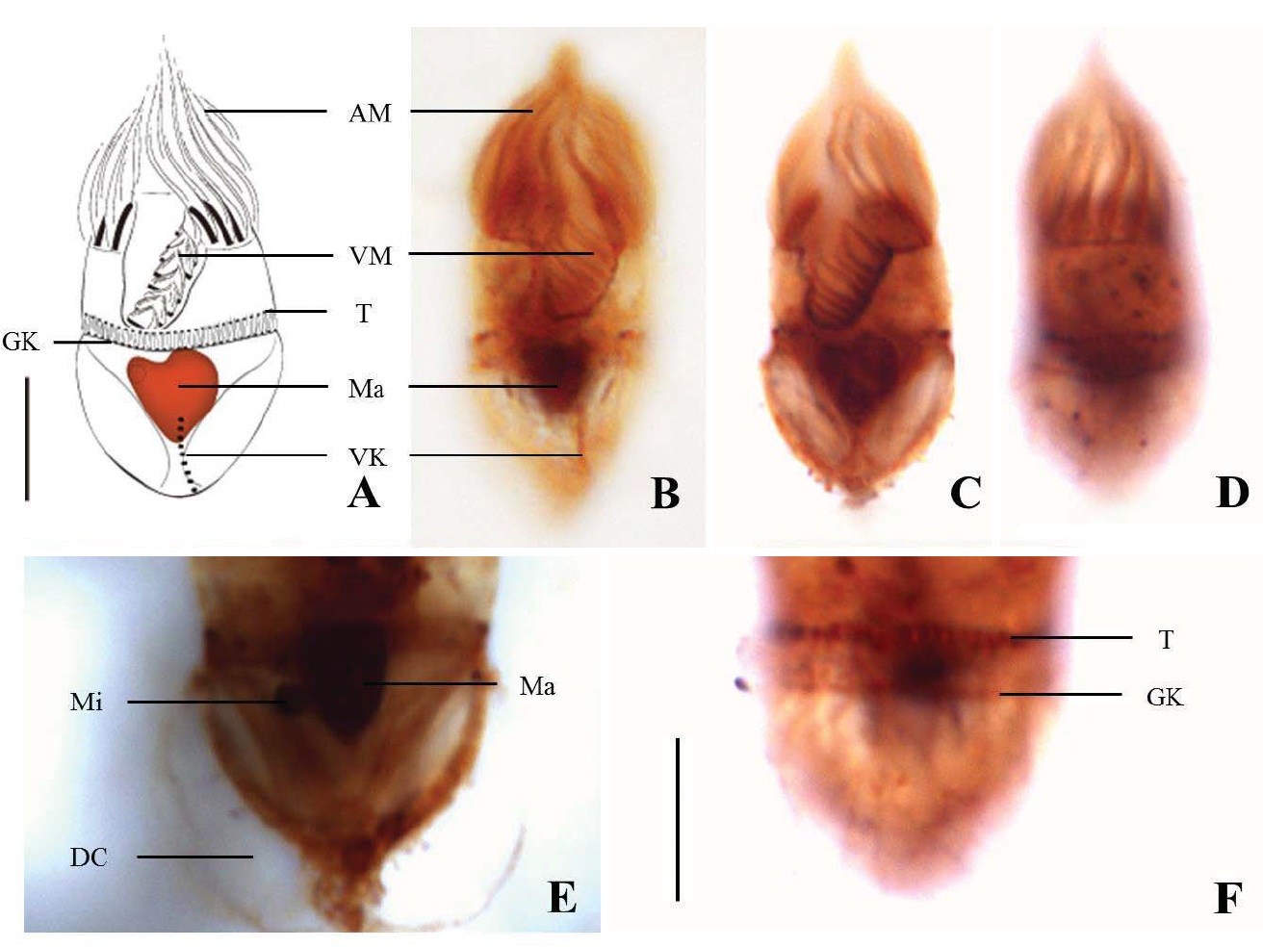
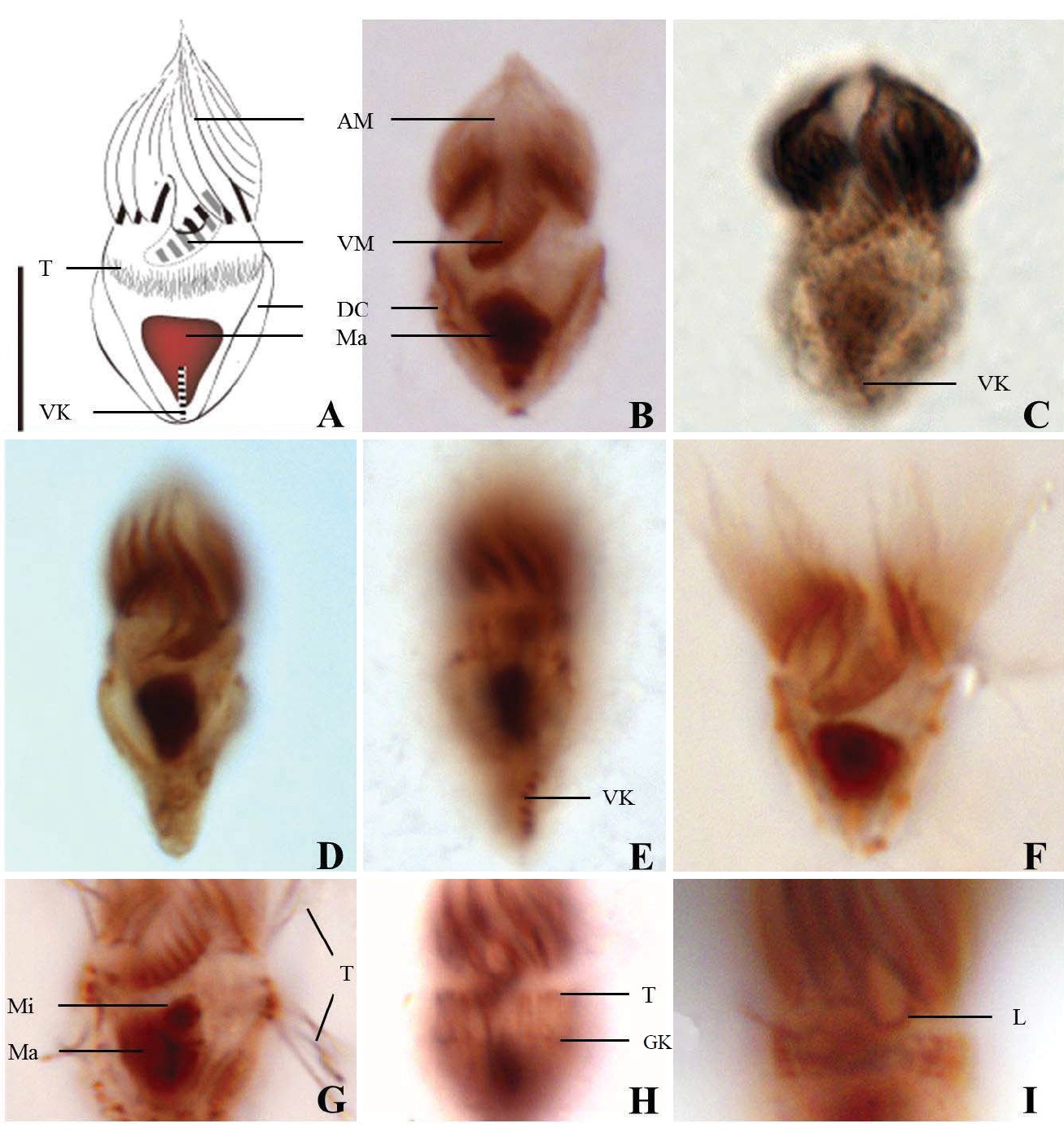
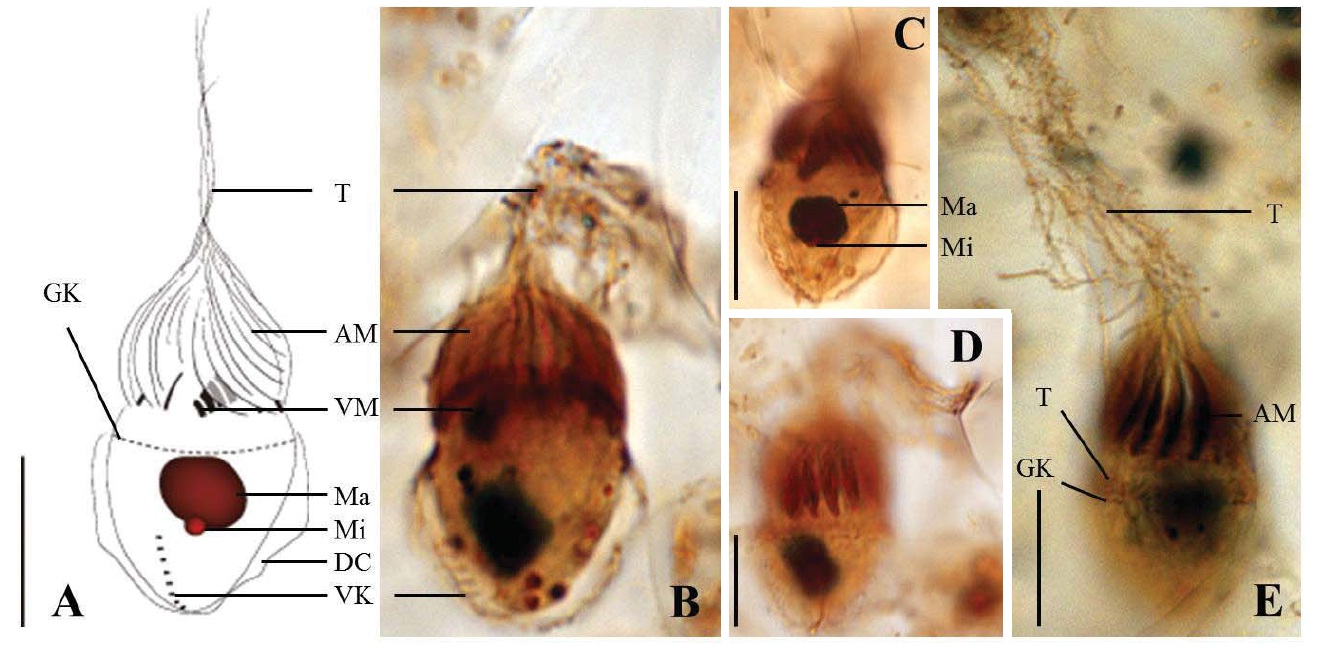
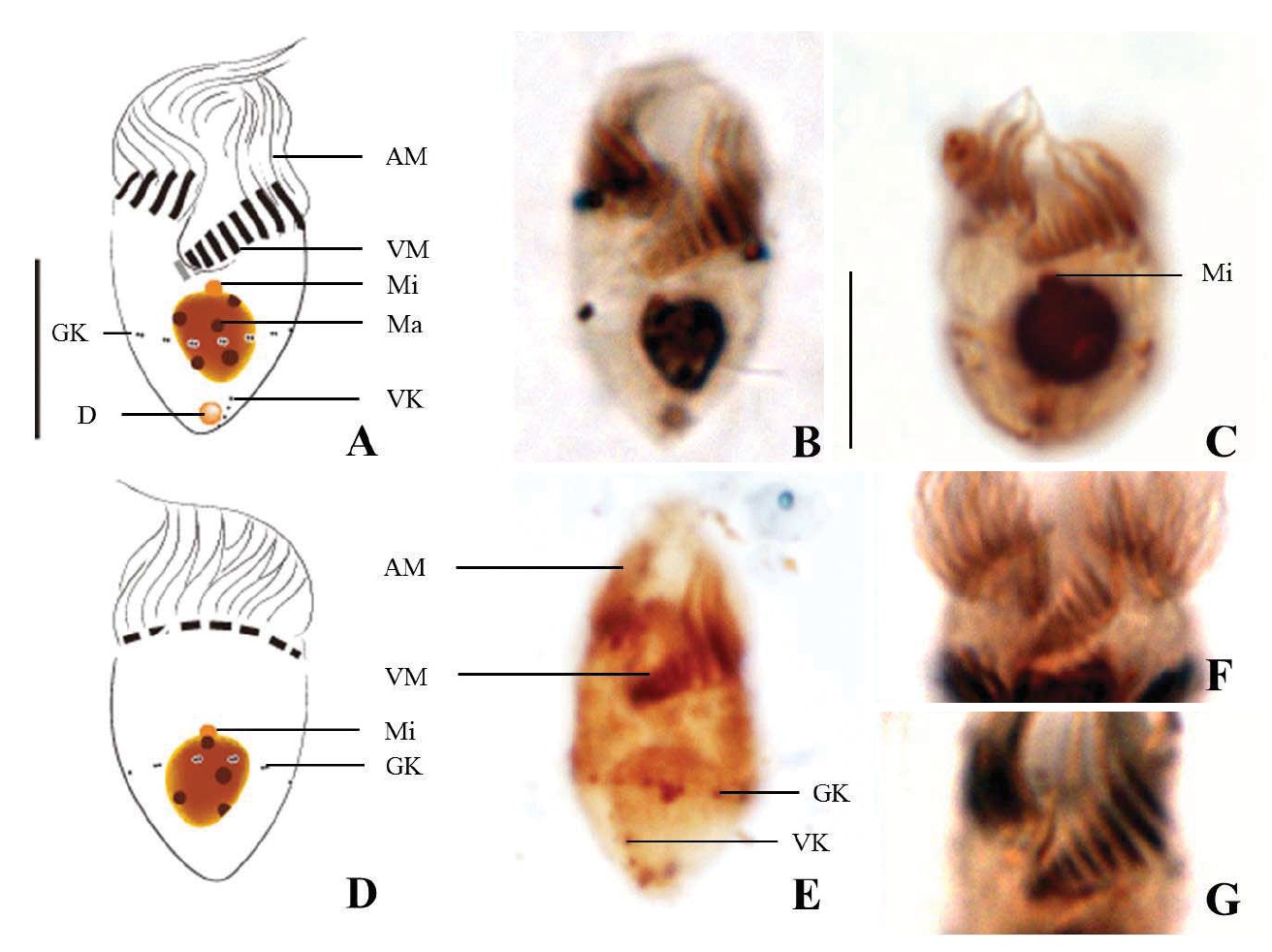
Diagnosis. Body long cone-shaped; deep and open oral groove on ventral surface, extending posteriorly to about halfway down the cell; size 22-27 μm in length and 11-17 μm in width after protargol impregnation; anterior membranelles composed of 13-15 polykinetids; ventral membranelles distinctly separated from anterior membranelles, composed of 8-9 polykinetids; girdle kinety completely surrounding the cell, composed of 34-42 dikinetids; ventral kinety composed of 9-11 dikinetids; one heart-shaped macronucleus, 6×5 μm in size; one spherical micronucleus, 1-2 μm in diameter.
Morphological description of the Korean population. Long cone-shaped body, cylindrical anterior portion and conical posterior portion, no apical protrusion detected. Numerous trichites inserting above girdle and extending to posterior of body, not in bundles (Fig. 2F). Distended cell surface covered from girdle to posterior pole of the cell. No polygonal surface recognizable in protargol impregnated specimen (Fig. 2C-E). Size 22-27 μm in length and 11-17 μm in width after protargol impregnation (average 24.5×14.1 μm , n= 12).
Oral groove relatively deep and open on ventral surface, extending to about halfway down the cell (2/5-1/2 of the body). Anterior membranelles distinctly separated from ventral membranelles. Bases of anterior membranelles connected by fibres system, the cilia of anterior membranelles torchlike spiral, composed of 13-15 polykinetids (average 14 polykinetids, n= 12). Ventral membranelles lying on the left wall of oral groove that forms a sunken pit, composed of 8-9 polykinetids (average 9 polykinetids, n=12). No endoral membrane detected (Fig. 2A-C).
Somatic ciliature composed of girdle kinety and ventral kinety, no extra kinety detected. Girdle kinety, horizontal and equatorial, completely surrounding the cell and composed of 34-42 dikinetids (average 38 dikinetids, n= 7). Vertical ventral kinety, slightly tilted toward the right, composed of 9-11 dikinetids (average 10 dikinetids, n=2).
One macronucleus, heart-shaped, located below oral groove and contained many spherical nucleoli, 4-8 μm in length and 4-6 μm in broad width (average 6.2×5.1 μm , n= 12). One micronucleus attached on macronucleus, 1-2 μ m in diameter.
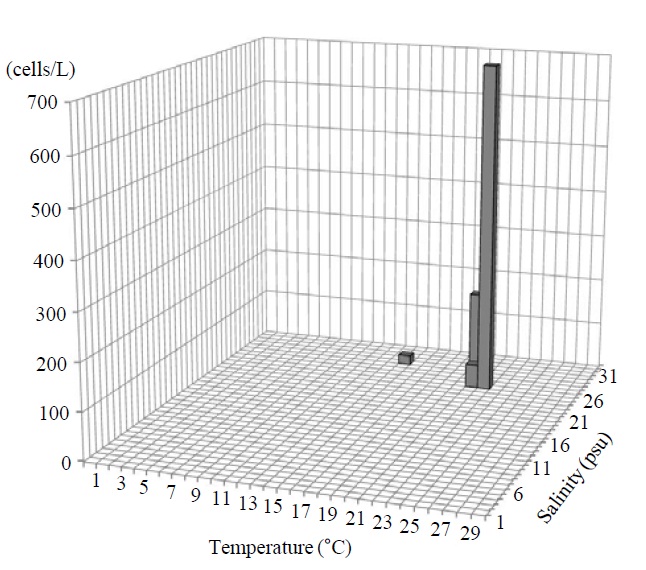
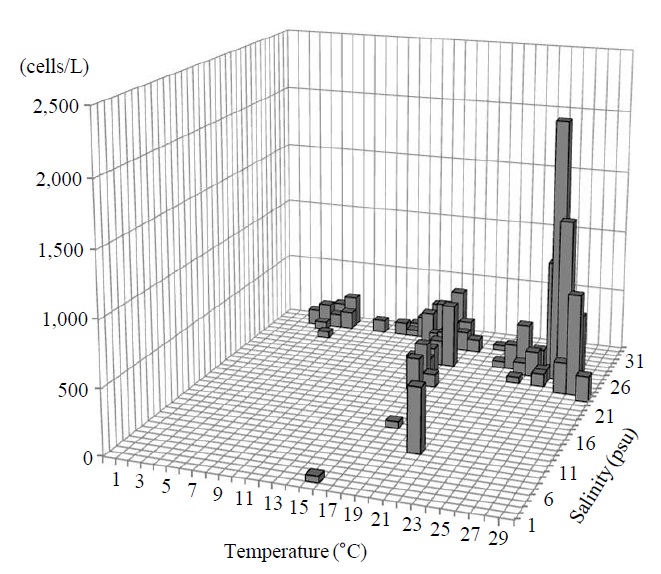
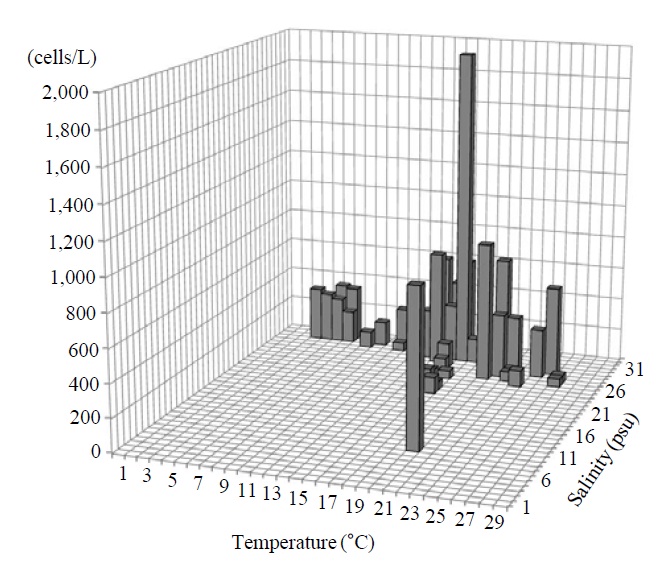
Occurrence and ecological characteristics. The Korean population was collected from Jinhae Bay (August, 2011) and Gwangyang Bay (November, 2011) recording the water temperatures of 22-23℃ and 15℃ and the salinities of 27-28 psu and 31 psu respectively. A maximum abundance (700 cells/L) was observed in August, 2011 from Jinhae Bay (Fig. 3).
The Argentina population was collected from Puerto Cuatreros where the water temperature and salinity were 9-12℃ and 30-33 psu respectively (Pettigrosso, 2003).
Comparison with other populations.
This population is similar to
Compared with the Antarctic population described by Petz et al. (1995), the two populations have many differences; the cell size is considerably smaller (22-27×11-17 μm in the Korean population vs. 90×49-52 μm in the Antarctica population), a smaller number of ventral kinety (9-11 dikinetids vs. 20 dikinetids), and more ventral membranelles (8-9 polykinetids vs. 4-5 polykinetids). As well, the ventral membranelles of the Antarctica population is inconspicuous (vs. extending to girdle portion in the Korea population), continuous with anterior membranelles (vs. conspicuously separated from anterior membranelles in the Korean population),
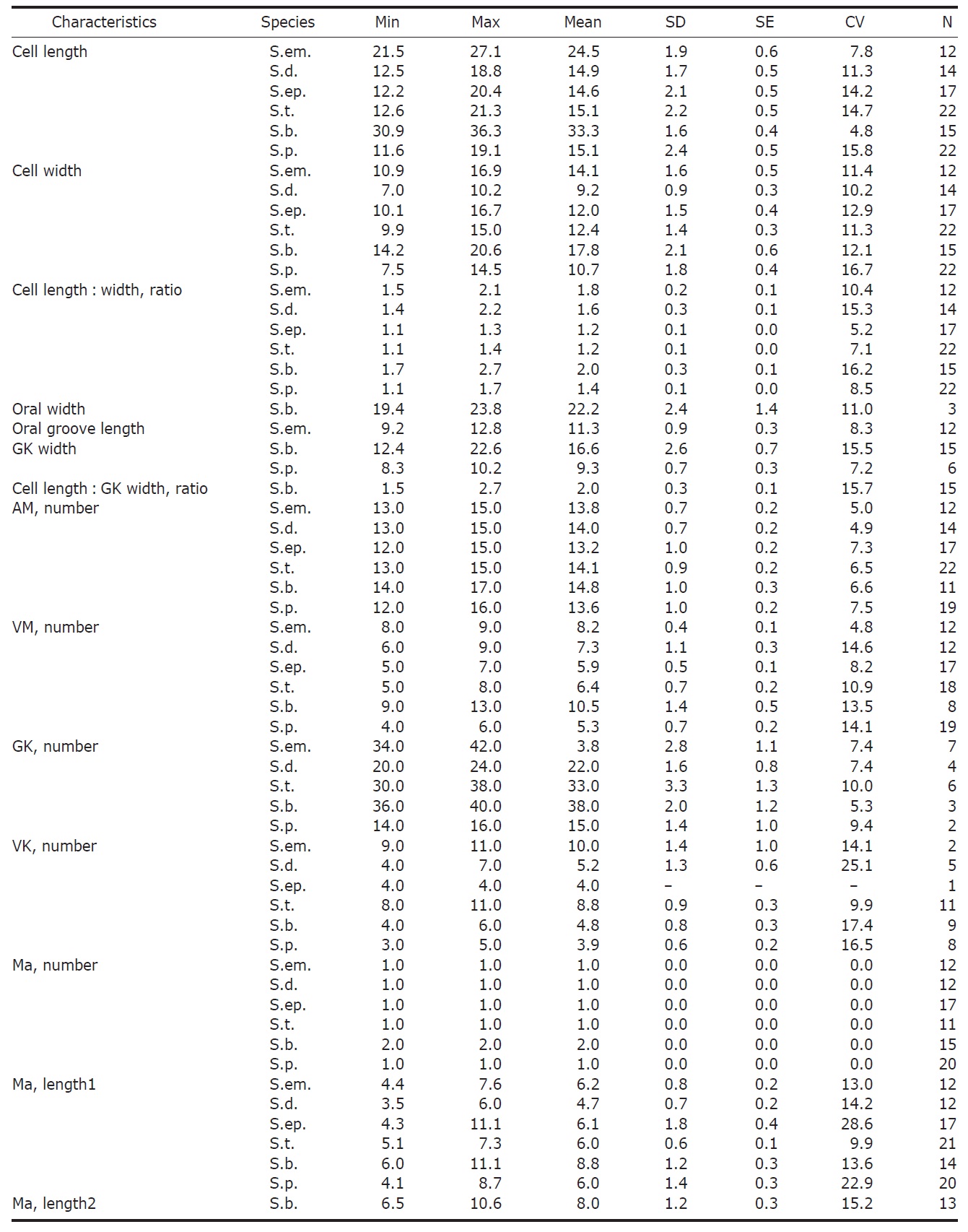
Morphometrical characterization of Strombidium emergens (S.em), S. dalum (S.d.), S. epidemum (S.ep.), S. tressum (S.t.), S. bilobum (S.b.), S. pollostomum (S.p.)
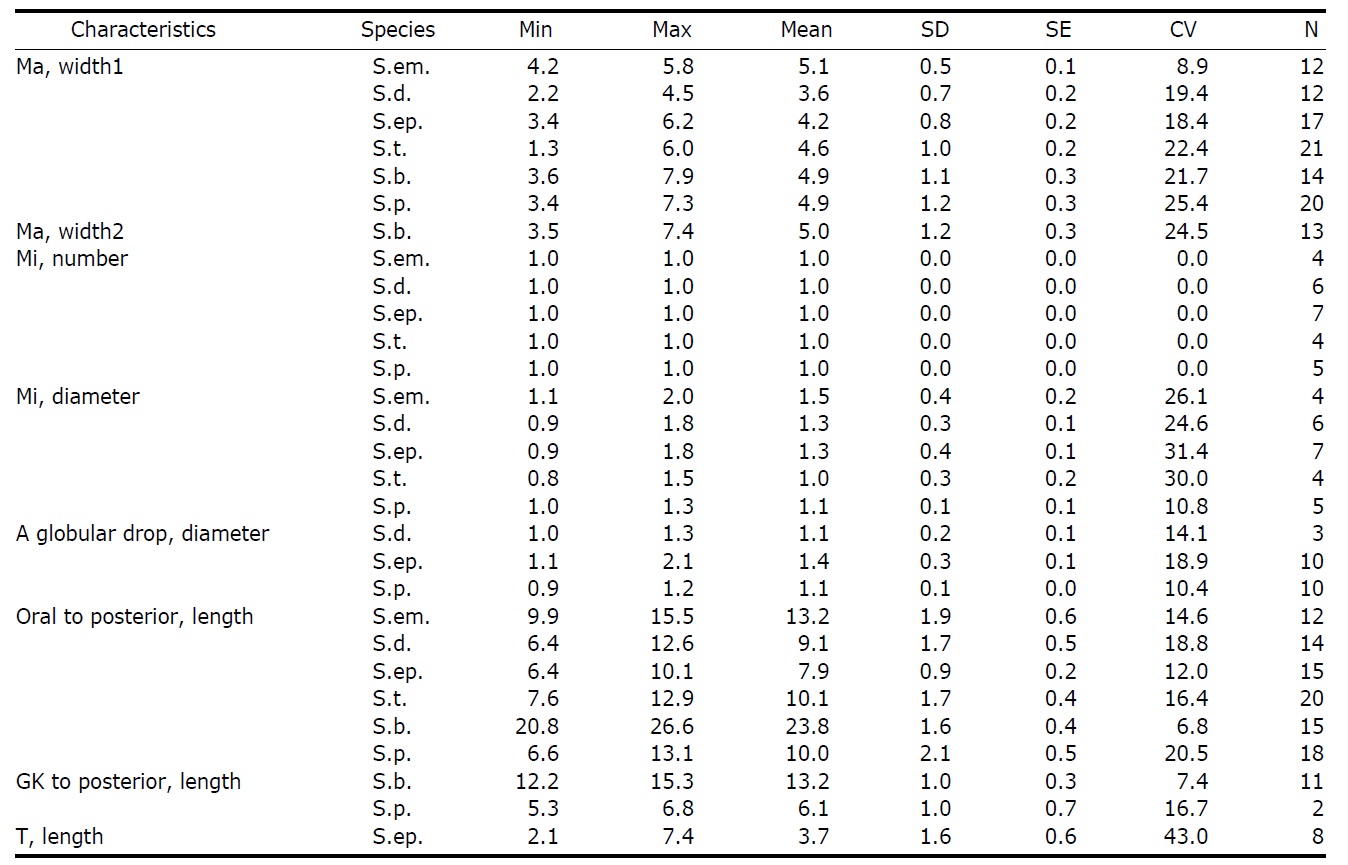
Continued
and covered by cytoplasmic wall (vs. open on ventral side).
1* Strombidium dalum Lynn, Montagnes and Small, 1988 (Table 1, Figs. 4, 5)
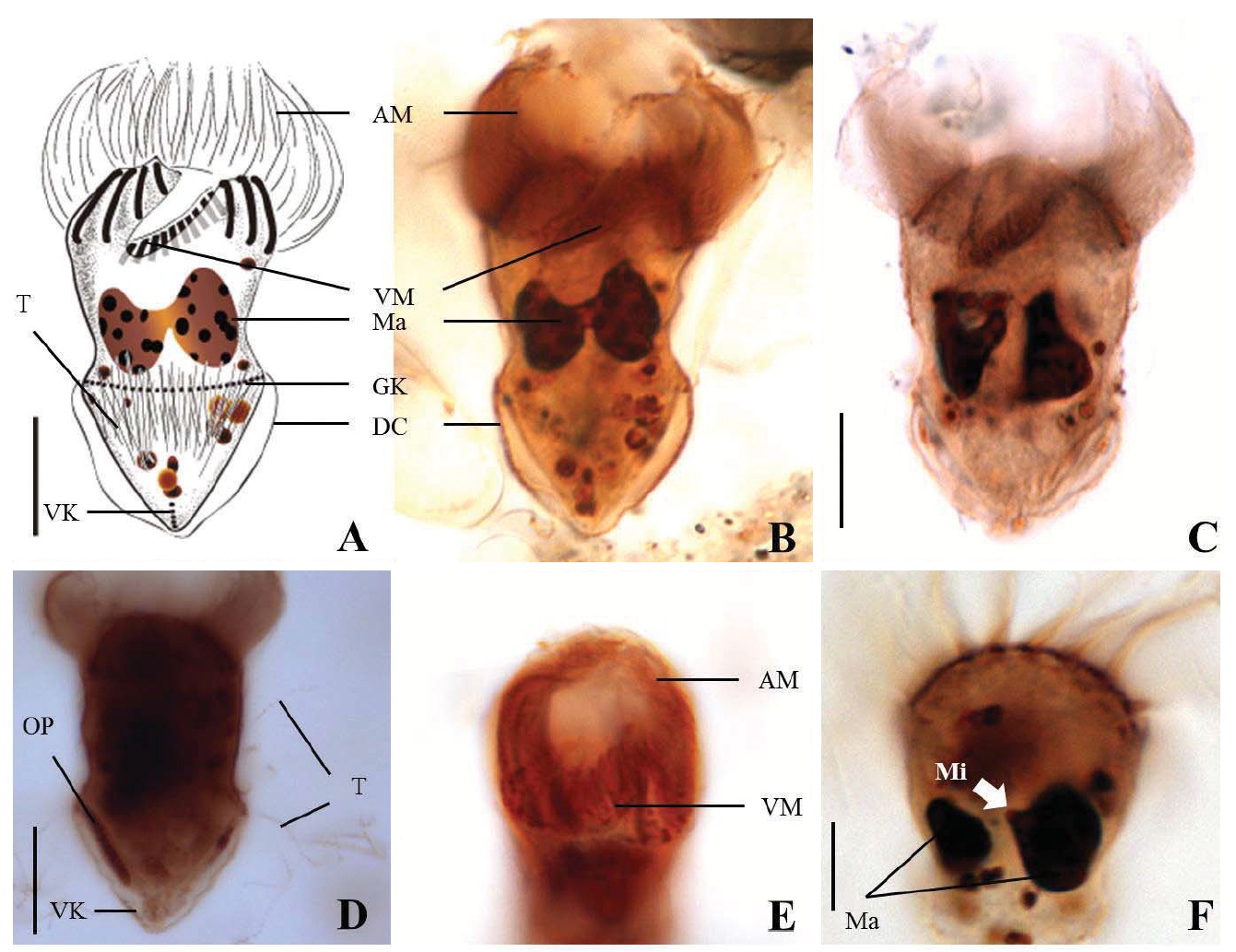
Diagnosis. Body conical to rhombus-shaped; deep oral groove; prominent and numerous trichites, positioned above girdle portion; size 13-19 μm in length and 7-10 μm in width after protargol impregnation; anterior membranelles composed of 13-15 dikinetids; ventral membranelles distinctly separated from anterior membranelles, composed of 6-9 dikinetids; equatorial girdle kinety completely surrounding the cell, composed of 20-24 dikinetids; vertical ventral membranelles composed of 4-7 dikinetids; one inverted triangle-shaped macronucleus located at posterior of cell, 5×4 μm in size.
Morphological description of the Korean population. Body mostly conical to rhombus-shaped, long conical shape from several cells observed (Fig. 4D, E), truncated anterior portion and narrowed posterior portion, no apical protrusion detected (Fig. 4A-C). Small lip on anterior pole of ventral side (Fig. 4I). Trichites, prominent and numerous, but not in bundles, completely surrounding the girdle portion (Fig. 4H, I). Slightly inflated distended cell surface from girdle portion to posterior end, no polygonal cell surface recognizable in protargol impregnated specimen. Size 13-19 μm in length and 7-10 μm in width after protargol impregnation (average 14.9×9.2 μ m, n= 14). Cell length by width ratio, 1.4-2.2 : 1
(average 1.6 : 1, n= 14).
Anterior membranelles distinctly separated from ventral membranelles. The cilia of anterior membranelles, long and spiral (usually torch-like shaped cilia), surrounding the anterior end of cell, composed of 13-15 polykinetids (average 14 polykinetids, n=14). Ventral membanelles lying on deep oral groove that forms a sunken pit, almost enclosed by left wall of oral groove, extending obliquely towards right side of cell and terminating one-third to half of the way down the cell, composed of 6-9 polykinetids (average 8 polykinetids, n=12). No endoral membrane detected.
Somatic ciliature composed of girdle kinety and ventral kinety, no extra kinety detected. Girdle kinety, equatorial, completely surrounding, composed of 20-24 dikinetids (average 22 dikinetids, n=4). Ventral kinety, vertically extended from posterior of cell, composed of 4-7 dikinetids (average 6 dikinetids, n=5) (Fig. 4C, E).
One macronucleus, mostly inverted triangle shaped and
located at posterior end, 4-6 μm in length and 2-5 μm in maximum width (average 4.7×3.6 μm, n= 12). One spherical micronucleus, 1-2 μm in diameter (average 1.3 μm in diameter, n= 6) (Fig. 4G).
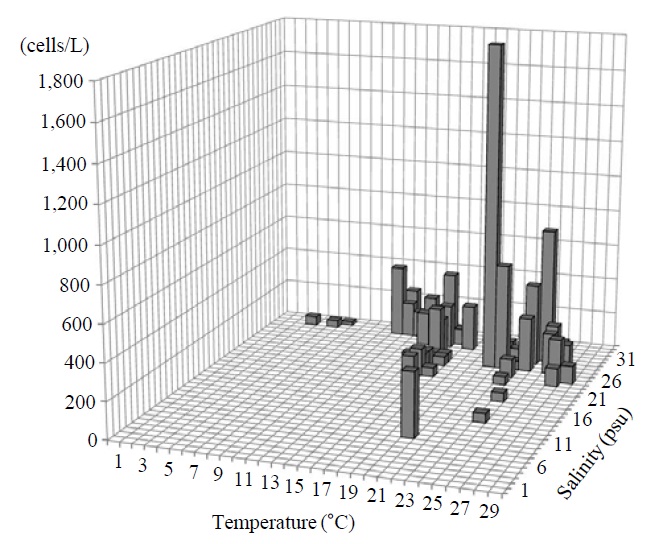
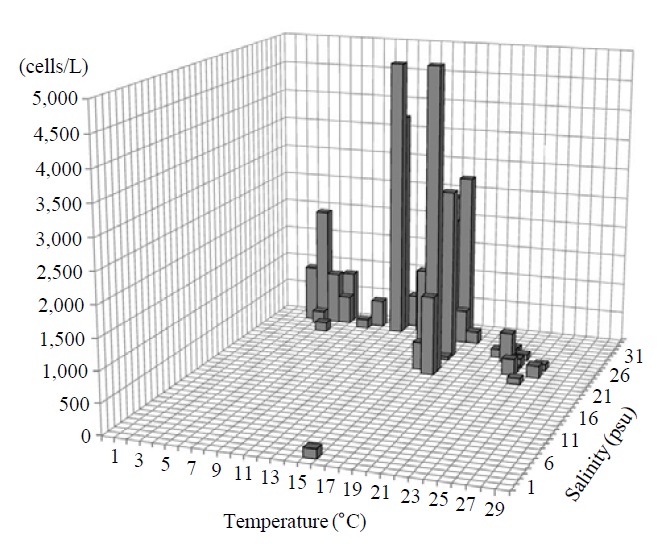
Occurrence and ecological characteristics. The Korean population was collected from Jinhae Bay and Gwangyang Bay in all seasons (water temperature of 4-29℃ and salinity of 9-34 psu). A maximum abundance (1,800 cells/L) was observed in Jinhae Bay in August, 2011 (water temperature of 22℃ and salinity of 27 psu) (Fig. 5).
The Argentina population was collected from Puerto Cuatreros in the lower temperature of 9-13℃ and the salinity of 30-31 psu (Pettigrosso, 2003).
Comparison with other populations. According to original description by Lynn et al. (1988), the cilia of the anterior polykinetids in the Atlantic population have spiral torch-like anterior membranelles. Lynn and Gilron (1993) noted that it may be an artifact of fixing and staining. They observed both torch-like cilia of anterior membranlles and fan-like one from the Jamaican population. In the Korean population, we also observed the two types of anterior membranelles (Fig. 4F). There are no description of the ventral kinety in the Atlantic and the Argentina populations (Lynn et al., 1988; Lynn and Gilron, 1993). However, the Jamaica and the Korean populations appeared the ventral kinety (3 dikineds and 4-7 dikinetids repectively) (Pettigrosso, 2003). Recently, the Canadian population was recorded by Suzuki and Ota (2011). Compared with the Korean population, all morphometric data overlap except for macronucleus size (about 5×4 μm in the Korean population vs. 8×6 μm in the Canadian population).
1*Strombidium epidemum Lynn et al., 1988 (Table 1, Figs. 6, 7)
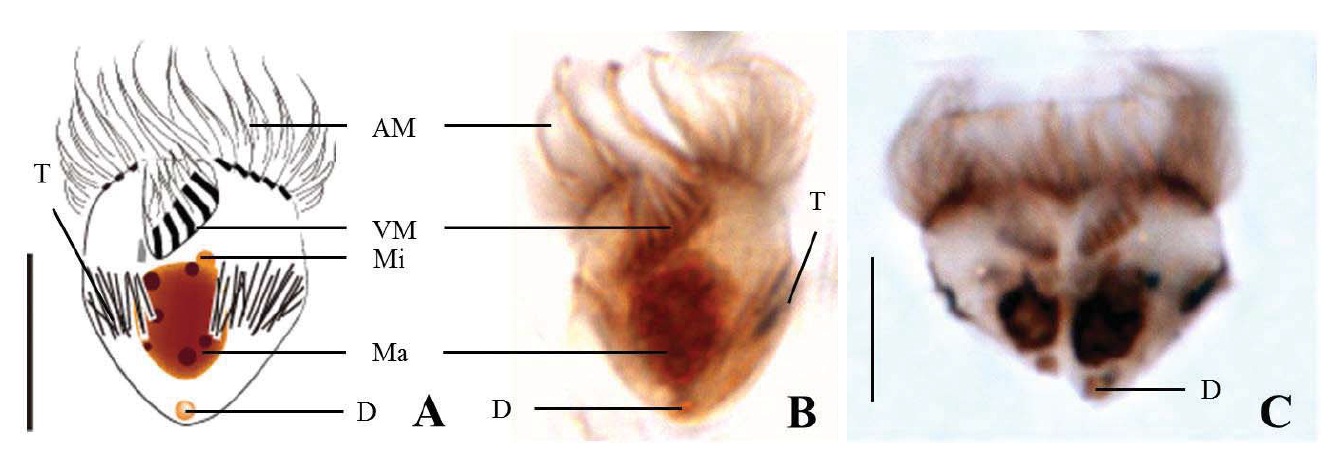
Diagnosis. Body truncate conical; size 12-20 μm in length and 10-17 μm in width after protargol impregnation; prominent trichites surrounding the cell; oral groove opened on ventral surface; anterior membranelles composed of 12-15 dikinetids; ventral membranelles separated from anterior membranelles, composed of 5-7 dikinetids; equatorial girdle kinety, surrounding the cell; one spherical to ovoid macronucleus, 6×4 μm in size; small
Morphological description of the Korean population. Body truncate conical. Size 12-20 μm in length and 10-17 μm in width (average 14.6×12.0 μm, n=17), cell length by width ratio, 1.2 : 1.0 after protargol impregnation. Prominent trichites inserting to anterior of girdle portion and extending internally toward posterior of body, 2-7 μm in length. A tiny globular drop on posterior end of cell, 1-2 μm in diameter (average 1.4 μm in diameter, n=10) (Fig. 6A, B).
Oral groove fairly opened on ventral surface of the cell. Anterior membranelles, equal length surrounding anterior end, composed of 12-15 polykinetids (average 14 polykinetids, n=17). Ventral membranelles are distinctly separated from anterior membranlles and lie on a oral groove which occupies 1/2 of cell length, to the posterior of ventral membranelles to posterior pole of cell distance 6-10 μm(average 7.9 μm, n=15), composed of 5-7 polykinetids (average 6 polykinetids, n=17) (Figs. 6B, 12F). One to two polykinetids of ventral membranelles lying into oral cavity. No endoral membrane detected.
Somatic ciliature composed of girdle kinety and ventral kinetids, no extra kinety detected. Girdle kinety, equatorial, completely surrounding cell. Vertical ventral kinety with 4 dikinetids from posterior end of body.
One spherical to ovoid macronucleus, 4-11×3-6 μm (average 6.1× 4.2 μm, n=17), positioned posterior. One micronucleus on macronucleus, 1-2 μm in diameter (average 1.3 μm in diameter, n=7).
Opisthe (daughter cell) developing on right side of proter (Fig. 6C).
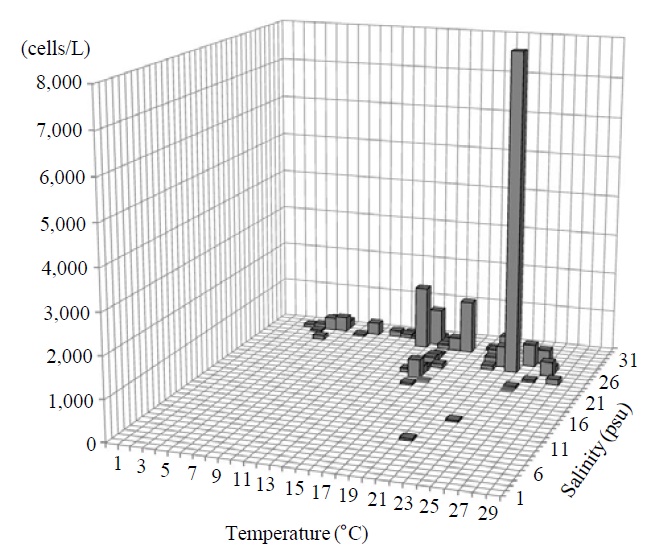
Occurrence and ecological characteristics. This species occurred from Jinhae Bay and Gwangyang Bay in all seasons (water temperature of 4-30℃ and salinity of 2-34 psu). A maximum abundance (2,075 cells/L) of this species was observed in Jinhae Bay on May, 2011 (water temperature of 27℃ and salinity of 27 psu) (Fig. 7).
Comparison with other populations.
les, and ventral membranelles separated from anterior membranelles. However the Atlantic population has slightly smaller cell size than the Korean population (9-14 μm in length vs. 12-20 μm in length) (Lynn et al., 1988; Lynn and Gilron, 1993; Suzuki and Ota, 2011).
A tiny globular drop was positioned in the posterior end of
1* Strombidiuim tressum Lynn et al., 1988 (Table 1, Figs. 8, 9)
Diagnosis. Body ovoid to ellipsoidal; size 13-21 μm in length and 10-15 μm in width after protargol impregnation; long trichites among cilia of anterior memnranelles; long and torchlike spiral anterior membranelles composed of 13-15 polykinetids; ventral membranelles composed of 5-8 polykinetids; supraequatorial girdle kinety composed of 30-38 dikinetids; vertical ventral kinety composed of 8-11 dikinetids; one ovoid macronucleus, 6×4 μm in size; one spherical micronucleus, about 1 μm in diameter.
Morphological description of the Korean population. Body ovoid to ellipsoidal, relatively inflated distended cell surface from girdle portion to posterior pole, apical protrusion not found (Fig. 8A-C). Size 13-21 μm in length and 10- 15 μm in width (average 15.1×12.4 μm, n=22). Trichites locating above girdle kinety, but not extending internally toward posterior of body (Fig. 8E). The long trichites among cilia of anterior membranelles, spirally extending over full length of cell (Fig. 8B, D, E).
Anterior membranelles, as long as almost body length, spirally twisted cilia (torch-like), completely surrounding the cell, and composed of 13-15 polykinetids (average 15 polykinetids, n=22). Ventral membranelles enclosed by the ring of anterior membranelles, composed of 5-8 polykinetids (average 7 polykinetids, n=18) (Fig. 8B, C).
Somatic ciliature composed of girdle kinety and ventral kinety, no extra kinety detected. Girdle kinety, supraequatorial, completely surrounding the cell, composed of 30-38 dikinetids (average 33 dikinetids, n=6). Ventral kinety slight-
ly slanted toward left, composed of 8-11 dikinetids on distended cell surface (average 9 dikinetids, n=11) (Fig. 8B).
One ovoid macronucleus, centrally positioned in cell, 5-7 μm in length and 1-6 μm in width (average 6.0×4.6 μm, n=21). One micronucleus on macronucleus, 1-2 μm in diameter (average 1.0 μm in diameter, n=4) (Fig. 8C).
Occurrence and ecological characteristics. The Korean population was collected from Jinhae Bay and Gwangyang Bay in all seasons (water temperature of 4-26℃ and salinity of 2-34 psu). A maximum abundance (5,250 cells/L) was observed in Jinhae Bay in May, 2011 (water temperature of 18℃ and salinity of 23 psu) (Fig. 9).
Comparison with other population. According to the original description, the torch-like spiral anterior membranelles and the extremely long trichites among the cilia of anterior membranelles are unique characteristics in
1* Strombidium bilobum Lynn and Gilron, 1993 (Table 1, Figs. 10, 11)
Diagnosis. Body subconical with narrowed girdle area; size 31-36 μm in length and 14-21 μm in width after protargol impregnation; apical collar opened on ventral surface; anterior membranelles located on apical collar, composed of 14-17
polykinetids; ventral membranelles covered by apical collar, composed of 9-13 polykinetids; horizontal girdle kinety composed of 36-40 dikinetids; vertical ventral kinety composed of 4-6 dikinetids; two ovoid macronuclear nodules or dumbbell shaped macronucleus, about 8×5 μm in size.
Mophological description of the Korean population. Shape subconical, anterior cylindrical and posterior pointed, most cells slightly narrowed at girdle part (Fig. 10A-C). Size 31-36 μm in length and 14-21 μm in width (average 33.3×17.8 μm, n=15), cell length by width ratio about 2 : 1 after protargol impregnation. Trichites inserting on anterior of girdle kinety and internally extending posterior end of cell. Transparent distended cell surface expanded from girdle kinety to posterior of body (Fig. 10A, B).
Apical collar opened on ventral surface. Anterior membranelles surrounding apical collar and composed of 14-17 polykinetids (average 15 polykinetids, n=11). Ventral membranelles separated from anterior membranelles and obliquely lying on oral groove that extended into oral cavity and almost covered by the apical collar, composed of 9-13 polykinetids (average 11 polykinetids, n=8) (Fig. 10E). Distance from posterior end of ventral membranelles to posterior pole of cell about 21-27 μm(average 23.8 μm, n=15).
Somatic ciliature composed of girdle kinety and ventral kinety, no extra kinety detected. Girdle kinety, subequatorial positioned (3/5 of body length), completely surrounding the cell, composed of 36-40 dikinetids. Distance from girdle kinety to posterior pole of cell distance 12-15 μm (average 13.2 μm, n=11), Ventral kinety, vertically arranged from posterior pole of cell, composed of 4-6 dikinetids (average 5 dikinetids, n= 9) (Fig. 10A, D).
Two ovoid (separating) macronuclear nodules or dumb-
bell shaped (connecting with each other) macronucleus, positioned near the posterior margin of ventral membranelles, one size 6-11 μm in length and 4-8 μm in width, another size 7-11 μm in length and 4-7 μm in width (average 8.8× 4.9 μm, 8.0×5.0 μm, n=15) (Fig. 10B, C, F).
Oral primordium originated below girdle part and left portion of ventral side (Fig. 10D).
Occurrence and ecological characteristics. The Korean population was collected from Jinhae Bay and Gwangyang Bay in all seasons (water temperature of 4-27℃ and salinity of 9-34 psu). The maximum abundance (1,950 cells/L) was observed in Jinhae Bay in May, 2011 (water temperature of 18℃ and salinity of 31 psu) (Fig. 11).
Comparison with other population.
Compared with the Jamaican population, the Korean population has the similar shape, polykinetidal numbers, and bilobed macronucleus. However, there are two different morphological characteristics: the ventral membranelles are distinctly separated from anterior membranelles in the Korean population (vs. anterior membranelles continuous with the ventral membranelles in the Jamaica population) and subequatorial girdle kinety (vs. equatorial girdle kinety).
1* Strombidium pollostomum Lynn and Gilron, 1993 (Table 1, Figs. 12, 13)
Diagnosis. Body hemisphere to semi-ovoid, posterior pointed; size 12-19 μm in length and 8-15 μm in width after protargol impregnation; oral groove opened on ventral surface of cell; anterior membranelles composed of 12-16 polykinetids; ventral membranelles continuous with anterior membranelles, composed of 4-6 polykinetids; subequatorial girdle kinety composed of 14-16 dikinetids; ventral kinety composed of 3-5 dikinetids; one spherical macronucleus, 6×5 μm in size; one micronucleus, about 1 μm in diameter; small Strombidium species (≤20 μm).
Morphological description of the Korean population. Hemisphere to semi-ovoid in body shape, posterior pointed (Fig. 12A-C, E). Cell size 12-19 μm in length and 8-15 μm in width (average 15.1×10.7 μm, n=22), cell length by width ratio=1.4 : 1 after protargol impregnation. No trichites found in body. A tiny globular drop on posterior of cell, 1-2 in number and 1 μm in diameter (average 1.1 μm in diameter, n=10) (Fig. 12A, B).
Oral groove opened on ventral surface of the cell. Anterior membranelles torch-like shaped and slightly sloped toward left side of cell, composed of 12-16 polykinetids (average 14 polykinetids, n=19). Ventral membranelles continuous with anterior membranelles, composed of 4-6 polykinetids (average 6 polykinetids, n=19), from posterior of ventral membranlles to posterior pole of cell distance 7-13 μm(average 10.0 μm, n=18) (Fig. 12B, C, E, G).
Somatic ciliature composed of girdle kinety and ventral kinety, no extra kinety detected. Girdle kinety, equatorial to subequatorial (1/2-1/3 from posterior end of cell), composed of 14-16 dikinetids (average 15 dikinetids, n=2), completely surrounding cell, from girdle kinety to posterior pole of cell distance 5-7 μm(average 6.1 μm, n=2). Ventral kinety vertically extending from posterior end, composed of 3-5 dikinetids (average 4 dikinetids, n=8) (Fig. 12D, E).
One spherical to ovoid macronucleus, containing many granules, 4-9×3-7 μm (average 6.0×4.9 μm, n=20), positioned centrally. One micronucleus on macronucleus, 1 μm in diameter (average 1.1 μm in diameter, n=5) (Fig. 12C).
Occurrence and ecological characteristics. This species was collected from Jinhae Bay and Gwangyang Bay in all seasons (water temperature of 4-28℃ and salinity of 9-34 psu). The maximum abundance (1,550 cells/L) of this species was observed in Jinhae Bay on May, 2011 (water temperature of 15℃ and salinity of 31 psu) (Fig. 13).
Comparison with other population.
This species is very similar to the Jamaica population in shape, the continuous arrangement and polykinetid numbers
of anterior and ventral membranlles. However, the Korean population is bigger than the Jamaican population (12-19×8-15 μm vs. 9-14×7-11 μm) and has a globular drop on posterior of the cell.
Suzuki and Ota (2011) described a species under the name of
Korean name: 1*민소모충속, 2*반나민소모충 Korean name: 1*횃불민소모충 Korean name: 1*범소민소모충(epidemos, Gr.-prevalent, 일반적인, 널리 퍼져있는) Korean name: 1*긴털민소모충 Korean name: 1*아령핵민소모충 Korean name: 1*극소민소모충