Estuarine tidal flats are important sites for production and remineralization of organic matter (OM) and recycling of nutrients [1, 2]. These sites are the transitional zone from land to ocean system, and receive materials, such as OM and nutrients transported from rivers or oceans. Especially, sedimentary organic matter (SOM) is an important food source of macrobenthos in estuary systems. However, an overabundance may lead to reductions in species and biomass of macrobenthos, due to oxygen depletion and buildup of toxic by-products (e.g., ammonia and sulphide), which is associated with the break-down of these materials [3, 4]. Therefore, to accomplish sustainable estuary management consistent with conserving such valuable estuarine ecosystems, better understanding of the sediment environment in estuarine systems is needed. Although the dynamics of OM in estuaries have been studied, it still remains about the sources, fate and role of OM. The reasons for these uncertainties include the complex interactions among the various physical, geological and biochemical factors that define each estuarine ecosystem and control OM in these environments [5, 6].
In the last decade, more attention has been paid to the SOM composition in estuary ecosystem [7, 8]. However, many of these investigations focused on the influential relationship between the role of terrestrial and marine OM. Because of the terrestrial- derived OM with high C/N tends to be less, and chemical properties of OM can affect its consumption and abundance in sediment of estuarine tidal flats. Whereas, according to the some studies, it indicated that the dynamic of OM is influenced by the benthic microorganisms (e.g., bacteria and benthic microalgae) in estuarine sediment, due to high productivity and re-mineralization of OM [9, 10]. For instance, according to Lee et al. [9] in 1998, the degraded OM amount by bacteria in Hiroshima Bay tidal flat indicated about 5-14 times more than macrobenthos. Thus, benthic microorganisms play important roles in the sediment environments, but little is known concerning its role and fate of microorganism in sediment OM dynamic [11, 12].
Isotopic ratios of C can also be helpful in distinguishing between the marine and terrestrial OM. However, if more than two sources are suspected of being present, carbon isotope studies alone cannot detect the contribution of the other sources [13]. Whereas, fatty acid biomarker have also been used as trophic markers to determine the transfer of OM, within food webs, since these fatty acids are fundamental components of cellular material with high biological specificity, which are generally incorporated by higher trophic levels with little modification in its structure [14]. Therefore, we used a combination of fatty acid biomarkers and stable isotope (δ13C) to identify the SOM origins in a Nanakita estuary.
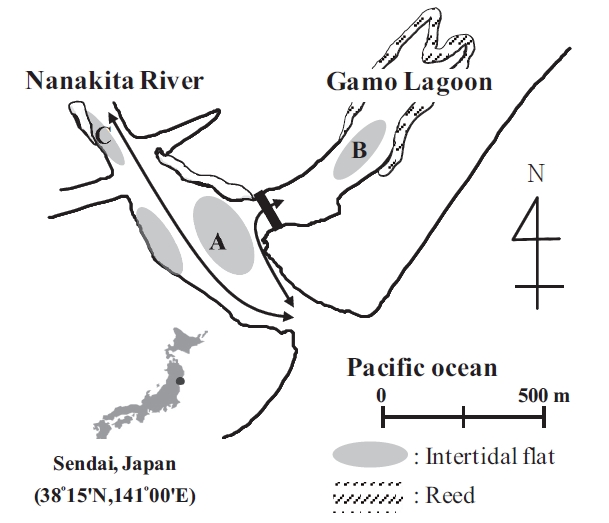
The currently studied tidal flats are located on the north and south banks, within 500 m, and showed diverse sediment properties from sandy to muddy. The fact is thought that sediments have the same OM origin because the tidal flats are located on the same location. However, Sediments are shown obviously with different properties. The purpose of this study is to determine the relative contribution of OM to the sediment of a Nanakita River estuary, with a different sediment characteristic, Nanakita River. We estimated the contribution of organic matters in the sediment by carbon stable isotopes and fatty acids.
This study was conducted in a tidal flat system of the Nanakita River estuary, involving Gamo lagoon on the north-east coast of Honshu Island, Japan (38˚15'N, 141˚00'E) (Fig.1). Nanakita River discharges to the Pacific Ocean at ~15 m3/sec (annual mean). Moreover, the studied tidal flats are located on the north and south banks, within a 500 m distance, and showed diverse sediment properties from sandy to muddy. Whereas, Gamo lagoon extends over 0.22 km2 with 5 ha of tidal flats, at low tides. Reed (
Sediment samples (about 1 g) were acid-treated with 1 N HCl, for 24 hr in pre-combusted glass test-tubes to remove carbonate [8]. Then, samples for carbon stable isotope analysis were combusted in a DELTAplus elemental analysis (Thermo Fisher Scientific, Waltham, MA, USA), and were passed online to an isotope ratio mass spectrometer (Delta plus MAT Finnigan; Thermo Fisher Scientific) to determine 13C/12C. The results are expressed as per mill deviations, relative to the conventional standards, i.e., Pee Dee Belemnite carbonate (PDB) for carbon, as follow:
δ13C (‰) = {(RSample / RStandard) ? 1} × 103 (1)
Where R = 13C/12C. Reproducibility in the analyses was ± 0.2‰ for δ13C.
In this study, terrestrial plant (TP), marine particulate organic matter (POM), estuarine POM, and benthic microalgae (BMA) were assumed as a potential OM of SOM. In case of terrestrial plant, reed (
Marine POM (surface seawater of Sendai Bay) samples were collected with a bucket (1-2 L). The water samples were then filtered through precombusted (2 hr, 450℃) Whatman (Clifton, NJ, USA) GF/F filters to collect POM. The filters were dried in an electric oven (50℃) and the dried filter was washed with 6 N HCl before being kept in a desiccator for half a day, to allow for decarbonization. Benthic microalgae on the surface sediment of the large sand flat as sampled at the stations A, and the benthic microalgae extraction was conducted, following the method described by Riera et al. [11].
Lipids of samples, 10 g dry weights (wt) of sediments were extracted, following the slightly modified version of method of Bligh and Dyer [15] and Meziane and Tsuchiya [16].
Fatty acids methyl esters (FAMEs) were separated and quantified by gas chromatography (GC-17A; Shimadzu Inc., Kyoto, Japan), equipped with a flame ionization detector. Separation was performed with an free fatty acid phase (FFAP)-polar capillary column (100 m × 0.25 mm internal diameter) with He as a carrier gas (flow rate, 1.4 mL/min). After injection at 150℃, the oven temperature was raised to 150℃ at a rate of 4℃ per minute, then to 230℃ at 4℃ per minute, and finally held constant for 50 min. The flame ionization was held at 250℃. Most FAMEs peaks were identified by comparing their retention times with those of authentic standards (Supelco Inc., Bellefonte, PA, USA). For some samples, peaks of FA were identified with a GC-mass spectrometry (Table 1).
TOC content of sediment samples was determined for dried (105℃, 24 hr), homogenized and HCl-treated sediments, using a
[Table 1.] Fatty acids used as biomarkers for different food sources

Fatty acids used as biomarkers for different food sources
Heraeus Vario-el CHN-analyzer (Vario-eL III; Elementar Analysensysteme GmbH, Hanau, Germany). Chlorophyll
To determine whether the sediment properties differed between the stations, one-way analysis of variance (ANOVA) was calculated once with all data. All data were checked for normality and equality of variance and transformed, if necessary. Statistical analyses were conducted with SPSS ver. 17.0 software (SPSS Inc., Chicago, IL, USA).
3.1. Chemical Properties of Sediment
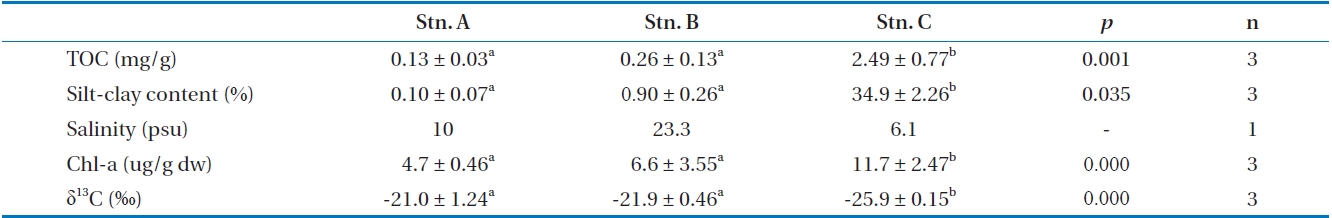
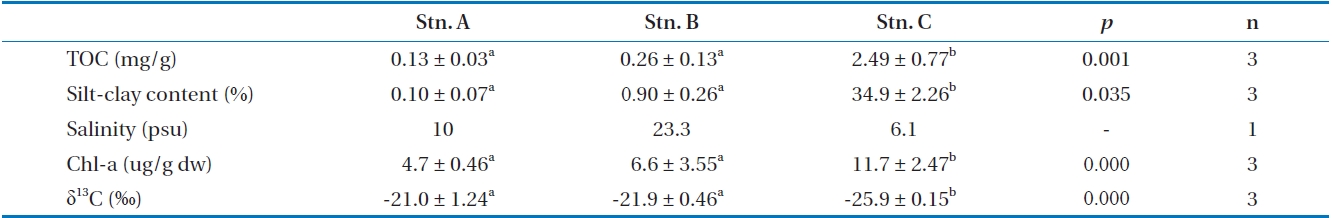
Table 2 shows sediment characteristics from the intertidal flats along the Nanakita estuarine and Gamo lagoon. Concentrations of TOC in sediments from the Nanakita estuary were relatively low, 1.3-24.9 mg/g. It is observed in Table 1 that TOC has the remarkable distribution differences along the Nanakita estuary (ANOVA,
Chlorophyll
[Table 2.] Sediment characteristics in Nanakita River estuary
Sediment characteristics in Nanakita River estuary
and sandy tidal flats more turbulent hydrodynamic conditions (e.g., Sakamaki and Nishimura [5]). In addition, muddy sediments generally showed high C:N and N:P ratios, compared with sandy sediments [21].
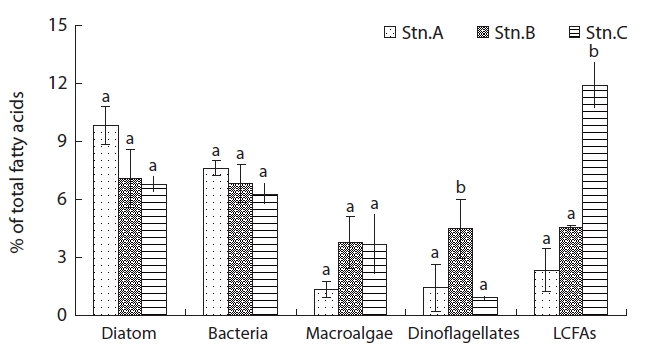
From these results, station A as sand flat showed a low amount algae-derived OM, but station C indicated a high amount OM with benthic microalgae and terrestrial plant, derived OM as muddy flat. Whereas, station B as sandy-muddy flat showed intermediate characteristic of station A and C.
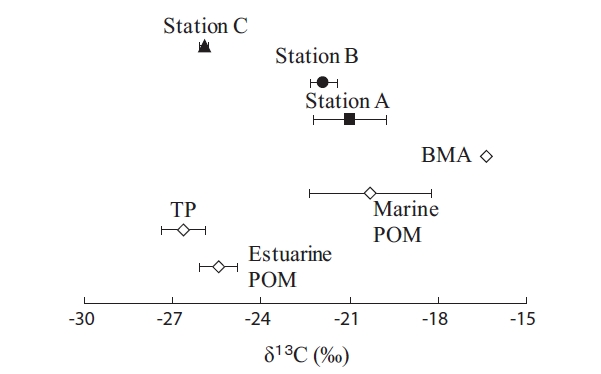
The Fig. 2 shows that δ13C values of potential OM sources and SOM in this study. The δ13C isotopic ratio for TP (δ13C = -26.3‰) was significantly lower than Marine POM (δ13C = -20.3‰). This large difference showed ease of the δ13C stable isotope analysis for the estimates of sediment’s potential OM. The δ13C value for Estuarine POM (δ13C = -25.5‰) were between TP and Marine POM, showing a mixture of material originating from terrestrial and marine OM [22]. Thus, phytoplankton production in rivers of Japan is usually small because water retention time is short, due to the steep slope [23]. Moreover, Kanaya [24] showed an average δ13C values (-26.0‰ ± 0.1) of riverine particulate organic matter in Nanakita River (a tidal river as same as our study area). Therefore, the freshwater phytoplankton would not be the major contributor to Estuarine POM in our study area.
The δ13C values of SOM in the study area showed stations A (-21.0‰), B (-21.9‰), and C (-25.9‰) (ANOVA,
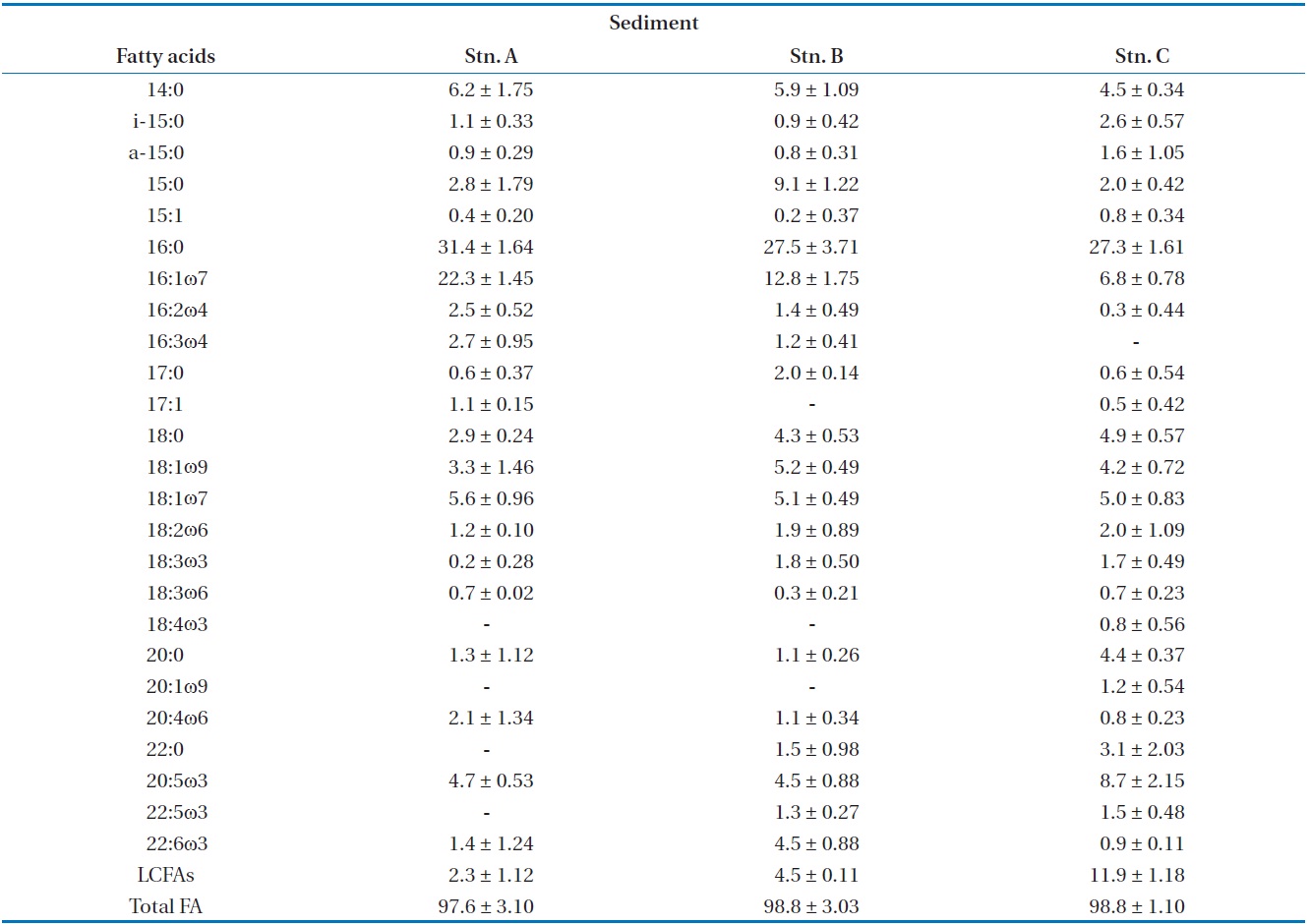
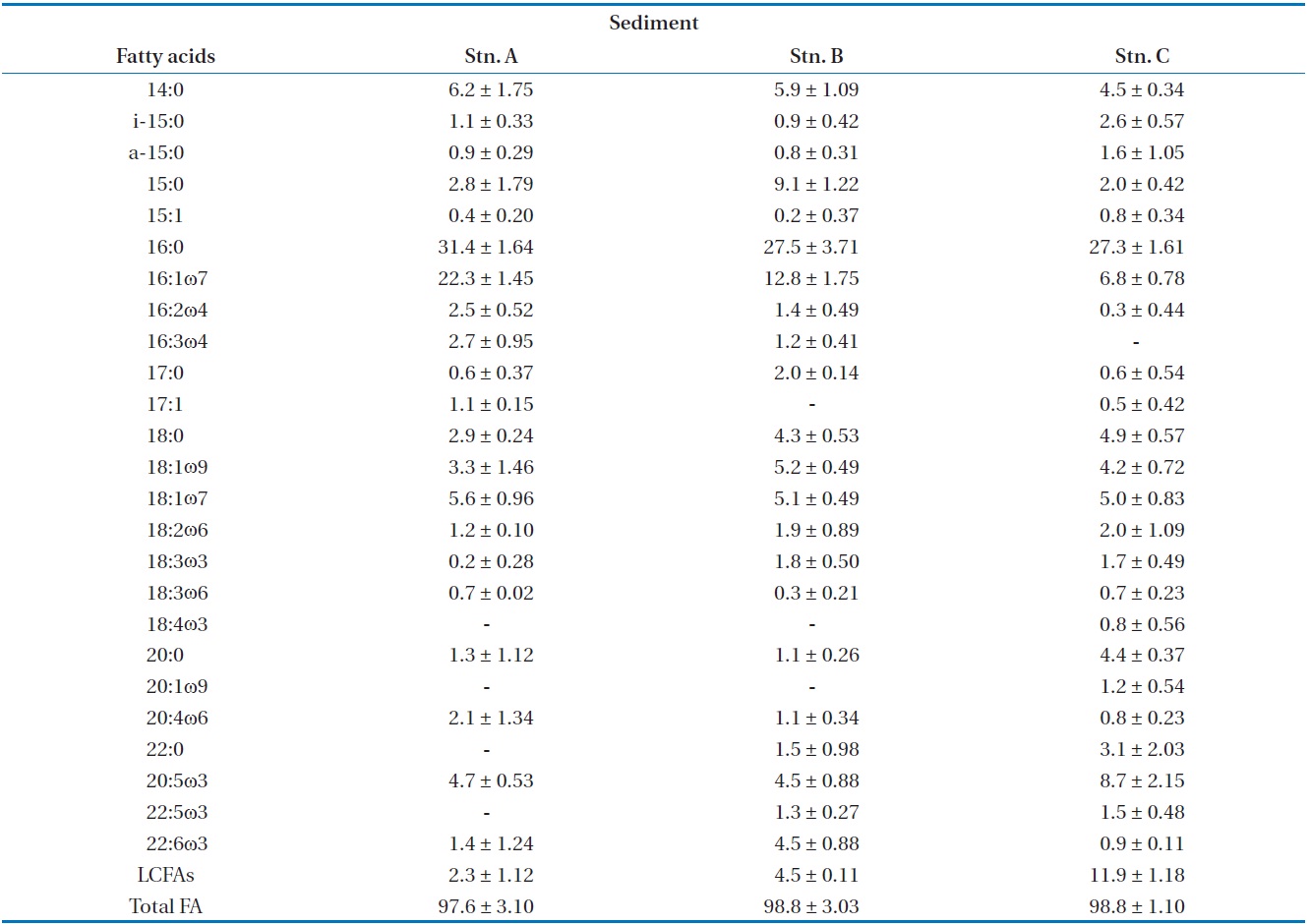
The most ratio fatty acids were 16:0 and 16:1ω7 in surface sediments (Table 2). The fatty acid ratio for diatoms appeared with no significant difference in spatial variation (
[Table 3.] Percentage composition of individual fatty acids in sediments of study stations
Percentage composition of individual fatty acids in sediments of study stations
algal-originated carbon to total sediment carbon may be higher in station A. This is in an agreement with the findings of Ogawa et al. [27] in this study station. In the previous studies, sand sediments showed relatively low C:N and N:P molar ratios which may indicated easily degradable OM of a high nutritional value relatively rich in nitrogen and phosphorus [21]. Hence, the close correlation between benthic microalgae and bacteria fatty acids indicates that bacteria are largely dependent on organic matter from a benthic microalgae source (r = 0.867,
This study was to determine the spatial variation of OM origin to sediment of estuarine tidal flats, with different sediment properties by carbon stable isotope and fatty acid biomarkers. Sediments of stations A and B were enriched in marine OM, diatom, and bacteria. Whereas, the OM at station C is dominated by terrestrial sources, along with diatom and bacteria, including high contents of OM. Moreover, microorganism-derived OM indicated high contribution to SOM composition, in all the study stations. Overall, the amount and origin of SOM showed obviously different relative properties in Nanakita estuary tidal flats, suggesting that the different transport characteristic of OM.