Piezoelectric and electrostrictive actuators are widely used in applications requiring precision displacement control or high generative force (i.e., optical stage, precision mechatronic systems, and semiconductor devices) [1,2]. In particular, multilayer actuators (MLAs) using Pb(Zr,Ti)O3 (PZT) ceramics have attracted much attention because of their merits, such as rapid operation, low power consumption, high precision control, and little noise [3,4] compared with conventional electromagnetic actuators.
Currently, most MLAs adopt expensive AgPd alloys as internal electrodes because they are stable between piezoelectric ceramic layers throughout a high-temperature sintering process. Therefore, there is ample necessity to explore inexpensive conducting materials as an internal electrode for low-cost and low-firing MLAs. For utilization of Ag as an inner electrode, co-firing temperature should be lower than the melting point of Ag, which makes it difficult to attain excellent piezoelectric properties, since they are generally improved with microstructural evolution. To overcome such problems, the low-temperature sintering of PZT ceramics has been attempted by a variety of approaches:sol-gel method [5], reactive sintering [6], fine powders prepared by high-energy ball-milling [7,8], and low-melting sintering aids,such as Li2O [9], LiBiO2 [10], LiSbO3 [11], Li2CO3 [12], and CuO [13,14].
In contrast, piezoelectric ceramics should possess high electric field-induced strain (EFIS) properties if they are to be applied to MLAs. It was reported that Pb(Mg1/3Nb2/3)O3-Pb(Zr,Ti)O3 (PMN-PZT) ceramics exhibit high piezoelectric constants as well as EFIS near the morphotropic phase boundary [14-17]. Moreover, our recent study on Pb(Fe1/2Nb1/2) added PMN-PZT complex perovskite ceramics demonstrated enhancement in their dielectric and pyroelectric properties by adding Li2CO3 as a sintering aid [15]. Therefore, this study investigates the effects of Li2CO3 on the sintering behavior and piezoelectric properties of 0.2Pb(Mg1/3Nb2/3)O3 - 0.5Pb(Zr0.475Ti0.525)O3 - 0.3Pb(Fe1/2Nb1/2) (hereinafter, PMN-PFN-PZT) ceramics and the possibility of
their application to low-firing MLAs using Ag inner electrodes.
Ceramic powder, with a composition of PMN-PFN-PZT, was synthesized using the columbite precursor method, which consists of a two-stage calcination process. In the first step, a mixture of MgO, Fe2O3, Nb2O5, ZrO2, and TiO2 powders (99.5% purity; Cerac Specialty Inorganics, Milwaukee, WI, USA) was properly weighed, according to the formula, and ball-milled with zirconia balls and deionized water for 24 hours. The mixed powders were dried and then calcined at 1,100℃ for 2 hours, to form a columbite phase of (Mg,Fe)Nb2(Zr,Ti)O6. In the second step, a stoichiometric amount of PbO, with 99.5% purity, (supplied by the same company) was added and mixed with calcined powders by ball-milling for 24 hours again. After drying, it was re-calcined at 850℃ for 2 hours. Before milling the calcined powders, Li2CO3 was added as a sintering aid in the range of 0.05 to 0.3 wt%. Ceramic pellets with a 12-mm diameter were prepared by dry pressing at 98 MPa and fired at temperatures between 800℃ and 200℃ for 2 hours, in air.
MLAs were prepared using 0.1 wt% Li2CO3 added PMN-PFN-PZT powders and thick film Ag. Piezoelectric ceramic sheets were obtained by tape casting ceramic slurry containing a mixture of piezoelectric powders, organic binders, a plasticizer, and solvents. The green sheets were cut into 10 × 10 cm2 using a knife cutting machine. Ag paste was screen-printed on the ceramic sheet, and then laminated with 12-layers of ceramic sheets. After cutting into 10 (W) × 15 (L) mm2, the samples were soaked at 500℃ for 6 hours, in air, to remove organic additives, and then co-fired at 850-950℃ for 5 hours, in air.
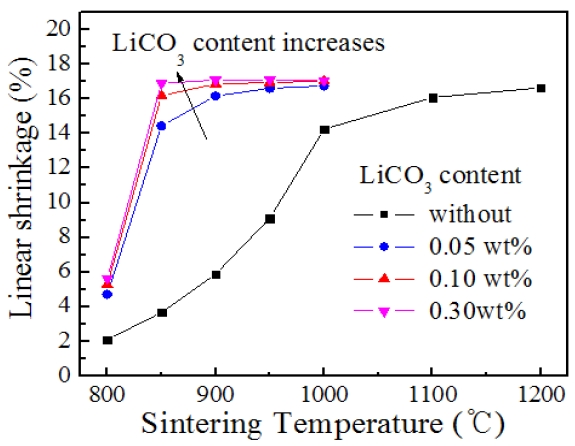
Figure 1 shows the firing shrinkage of PMN-PFN-PZT ceramics as a function of sintering temperature (
The effect of Li2CO3 on the sintering behavior is more clearly understood from microstructural observation. Figure 2 displays
the surface micrographs of specimens sintered at 900℃ for 2 hours. As seen in Fig. 2(a), a specimen sintered without L2CO3 reveals little grain growth and densification because the particle size is almost similar to that of a green compact (~0.5 μm), and many pores can be also observed. However, Li2CO3 added specimens show dense microstructures and significant grain growth;the average grain size for 0.05, 0.1, and 0.3 wt% Li2CO3 is 1.4, 2.2, and 1.8 μm, respectively.
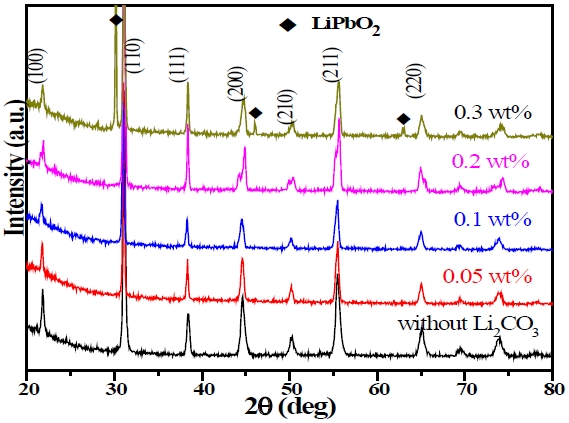
Such an enhancement in density and grain size by Li2CO3 was also observed in our previous work on PMN-PZT ceramics with added Li2CO3 [7] or Li2O [9]. A possible explanation is that sintering is enhanced by formation of a transient liquid phase at the initial stage [16], which is in turn dissolved into a matrix at a later stage of sintering. In this work, a low-melting Li compound was also detected from X-ray diffraction (XRD) analysis at high Li2CO3 levels, as shown in Fig. 3, which compares XRD reflections of specimens for different Li2CO3 amounts. All specimens correspond to perovskite symmetry, while a secondary phase that is identified as LiPbO2 is observed when the amount of Li2CO3 reaches 0.3 wt%.
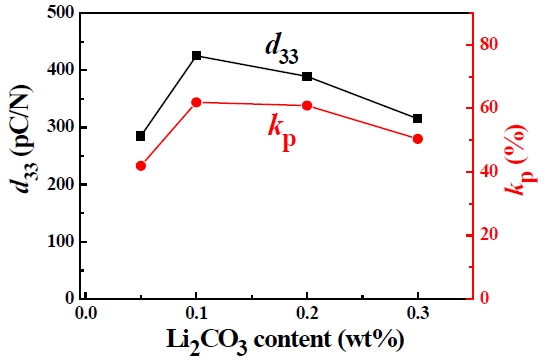
Figure 4 presents the piezoelectric constant
MLAs were fabricated by laminating PMN-PFN-PZT green sheets with a screen-printed Ag-ceramic electrode. After cofiring,the dimension of a MLA was about 8 (W) × 12 (L) × 1(T) mm3, in which 12 ceramic and 11 Ag inner electrode layers are alternatively stacked. The fractured surface of specimens sintered at 850, 900, and 950℃ was observed using a scanning electron microscope and the results are provided in Fig. 5. Lowmagnification micrographs (left column of Fig. 5) show well laminated multilayers with clear interfaces and a uniform thickness of about 80 μm in piezoelectric layers.
In the case of inner electrode layers, the morphologies are
more clearly seen in higher-magnification micrographs (right column). The Ag thickness is not uniform in a range of 3-5 μm, moreover, their connectivity is broken when
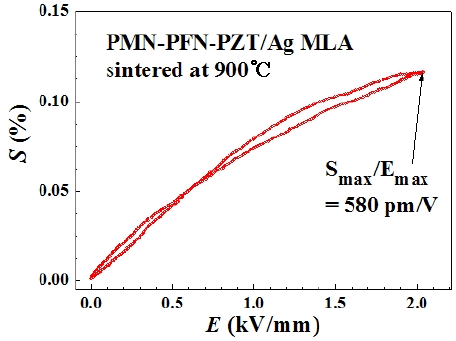
EFIS was measured for a MLA sintered at 900℃, and the result is displayed in Fig. 6. The normalized strain
The effects of Li2CO3 on the sintering behavior and piezoelectric properties of PMN-PFN-PZT ceramics have been investigated.It was found that Li2CO3 is quite effective in lowering
an unwanted Li2PbO3 secondary phase and subsequent degradation in the piezoelectric properties. Using 0.1 wt% Li2CO3 added PMN-PFN-PZT ceramics, MLAs using Ag inner electrodes were successfully fabricated, resulting in a normalized strain of 580 pm/V at a dc field of 1.5 kV/mm. This result is very promising for low-cost MLAs.