There is considerable interest in cadmium sulphide (CdS) compound semiconductor for thin film devices in electronic and photovoltaic applications [1-6]. CdS thin films are prepared by sputtering [5,6], thermal evaporation [4,7,8], screen printing [8], spray pyrolysis [9], and chemical bath deposition (CBD) [1,2,8,10-12]. The CBD process is a simple and inexpensive technique to obtain homogeneous, adherent, and stoichiometric CdS thin films [10]. Nanocrystalline CdS thin films are grown on glass substrates by CBD, in a solution bath containing cadmium salt, thiourea, and complexing agent. Sulphur ions are supplied by the decomposition of thiourea in an alkaline solution, and cadmium ions are usually complexed by NH4OH [11].
CdS thin films by CBD occur by the ion-by-ion process (condensation of Cd2+ and S2- ions on the substrate) or by the clusterby- cluster process (adsorption of colloidal particles of CdS on the substrate) [12-14]. Lincot and Ortega-Borges reported that CdS formation reaction proceeds via two consecutive steps, the formation of a dense compact inner layer, followed by the formation of a porous less adherent layer, and the inner layer growth mechanism corresponds to the ion-by-ion process [15]. CdS can exist in two crystalline modifications : the hexagonal (wurtzite) phase and the cubic (zincblende) phase [16]. The crystal structure of CBD CdS thin films is known as hexagonal, cubic, or mixed structure of hexagonal and cubic. Cardona et al. reported that the
The optical constants are necessary to study optoelectronic devices that use semiconductor thin films. Spectroscopic ellipsometry (SE) is a useful and nondestructive tool to obtain the optical constants of thin films. The complex dielectric function, optical constants, and microstructure of thin films are studied from spectroscopic ellipsometry [6,9,10,18-21]. The optical constants of refractive index, extinction coefficient, normal-incidence reflectivity, and absorption coefficient in thin films are obtained using SE data of the complex dielectric function [18,19,22]. The dispersion of refractive index can be analyzed using the Wemple- DiDomenico single-oscillator model (W-D model) [23], in which the refractive index
using the oscillator energy, dispersion energy, and photon energy.
In this study, nanocrystalline CdS thin film was prepared on glass substrate by CBD, and dielectric function and optical constants were investigated using spectroscopic ellipsometry. The dispersion of the refractive index of CdS thin film was analyzed using the W-D model, and the dispersion parameters of oscillator energy and dispersion energy were obtained.
CdS thin film was prepared on glass substrate by chemical bath deposition in an alkaline solution. The alkaline solution bath, containing cadmium sulphate, thiourea, and ammonium hydroxide, was maintained at 80℃, and the pH value of the aqueous solution was 10 [24]. The deposited CdS thin film was rinsed in deionized water, and cleaned ultrasonically. The complex dielectric function and microstructure of CdS thin film were investigated using a spectroscopic ellipsometer (M-2000D, J. A. Woollam Co. Inc., Lincoln, NE, USA) with rotating analyzer, in the photon energy range from 1.24 to 6.48 eV at room temperature. The ellipsometric parameters ψ and Δ at an incidence angle were measured at each wavelength λ, in the range from 45° to 75°, at angle intervals of 5°.
A four-phase (ambient/roughened surface layer/thin film/ substrate) model was used to obtain the optical parameters. For the roughened surface layer of CdS thin film, the effective medium approximation (EMA) [9,10,18] was used. The CdS thin film of the four-phase model was considered as a layer of compact CdS. Then, the roughened surface layer was considered as a mixture of CdS particles and voids. The data fitting was performed to minimize the mean square error (MSE) between the measured and calculated parameters of ψ and Δ. From the fitting results of SE data, the thicknesses of the inner compact layer and the roughened surface layer of CdS thin film were 296 and 18 nm, respectively. By employing the EMA, the roughened surface layer was found to be a porous layer, with 48.7 % CdS particles and 51.3 % voids.
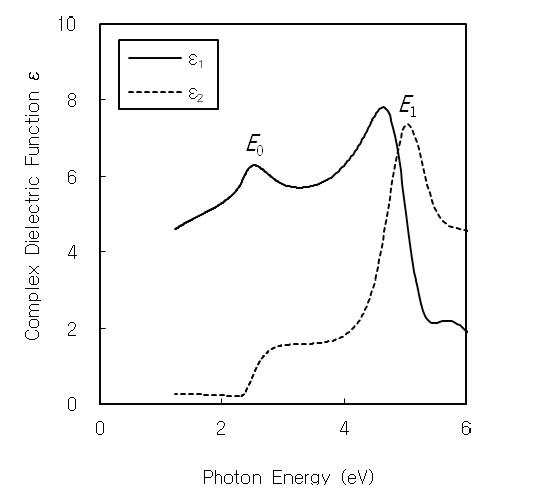
The peaks in the dielectric function spectra are useful to study the interband transitions in the Brillouin zone. Cardona et al. reported that hexagonal CdS has the
the imaginary part of the dielectric constant, and then the
The complex dielectric function ε(
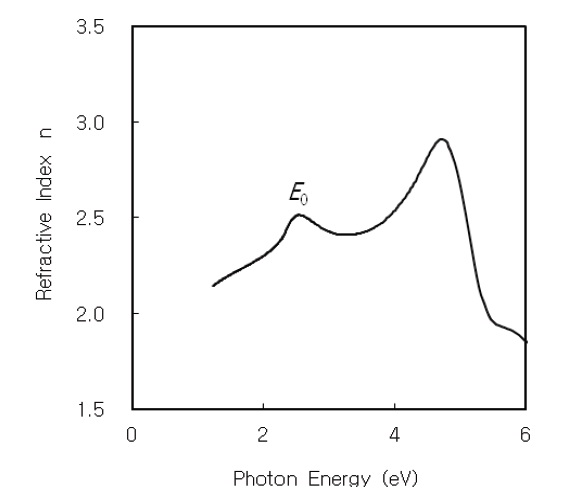
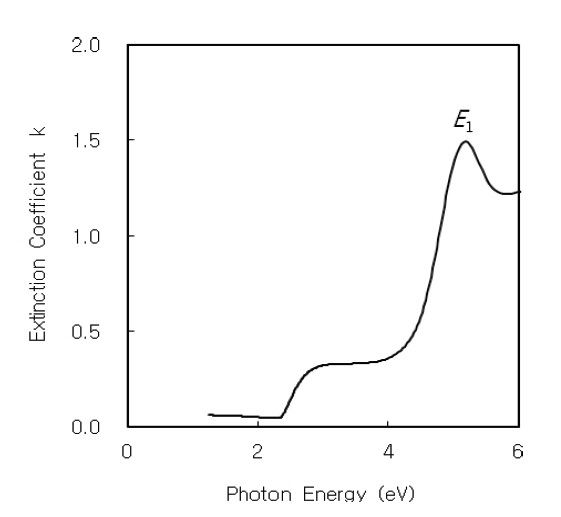
The optical constants of the refractive index
Figures 2 and 3 show the refractive index
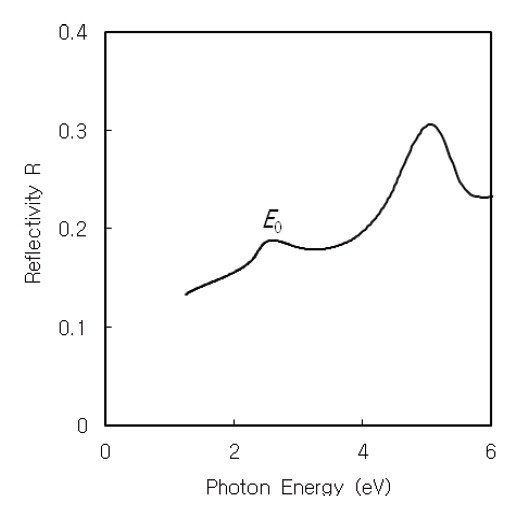
The normal-incidence reflectivity
Figures 4 and 5 show the normal-incidence reflectivity
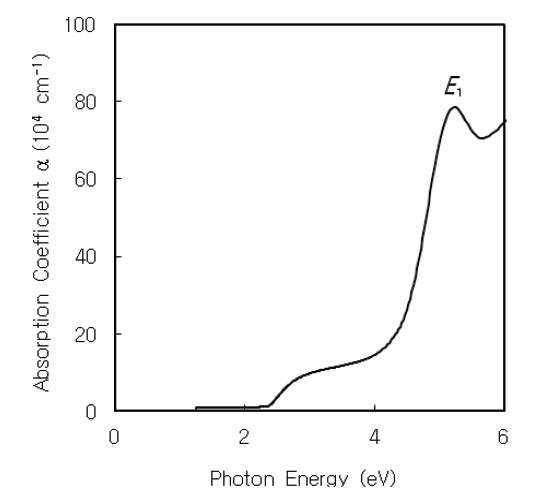
The optical absorption coefficient α(
where
The dispersion of refractive index below the interband absorption edge is analyzed using the W-D model [23]. In the W-D
model, the refractive index
where
Wemple and DiDomenico represented the dispersion energy
where
From the (
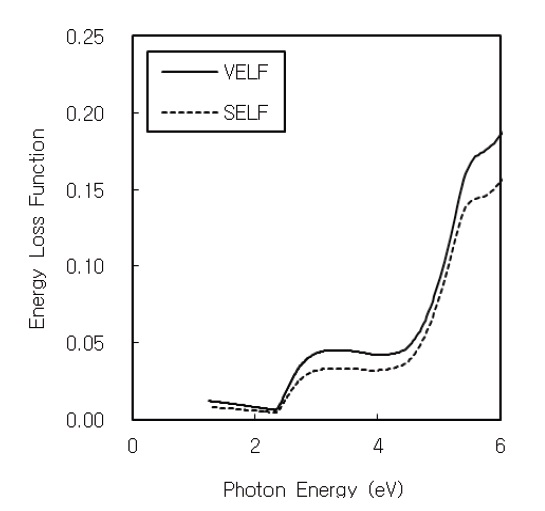
The volume and surface energy loss functions are proportional to the characteristic energy loss of fast electrons traveling the bulk and surface of the material, respectively. The volume energy loss function (VELF) and surface energy loss function (SELF) are related to the real and imaginary parts of the complex dielectric function [30].
Figure 6 shows the volume and surface energy loss functions of CdS thin film. The volume energy loss function is higher than the surface energy loss function. The energy loss functions reveal distinct
CdS thin film was prepared on glass substrate by chemical bath deposition, and characterized by spectroscopic ellipsometry. To investigate the dielectric function and optical constants of CdS thin film, a spectroscopic ellipsometer with rotating analyzer was used. The crystal structure of CBD CdS thin film was found to be cubic, by the presence of the