Dyes are extensively used in various industries, particularly in the textile, paper, rubber, plastic, leather, cosmetics, food, and drug industries. Effluents from the dyeing and finishing processes in these industries are highly colored, and their discharge into rivers makes the water unfit for domestic, agricultural and industrial purposes. Dye wastewater discharged from various industries creates certain hazards and environmental problems. Recently, all governments have been under severe pressure by their people to stop this type of effluent into the public water courses unless it is treated properly. Some dyes are toxic, and the biodegradation of others yields carcinogenic products such as aromatic amines [1,2].
Activated carbons pollutants are commonly used for the removal of dyes from potable water and wastewater due to their high sorption capacities for organic and inorganic pollutants despite their high cost. Agricultural byproducts such as fruit peel have been used [3-6]. Extensive studies have been run to improve the sorption capacities of agricultural byproducts for dye removal via physical or chemical modification [7-10]. Reactive dyes are typically izo-based chromospheres combined with different types of reactive groups. They differ from other classes of dyes in that they bind to textile fibers such as cotton via the formation of covalent bonds. Thus, they are widely used compared to other dyes.
Various techniques have been employed for the removal of dyes from wastewater, including adsorption [11-14], photocatalytic degradation [15], electrokinetic coagulation [16], advanced chemical oxidation [17,18], ozonation [19], liquidliquid extraction [20] and biological processes [21,22]. However, adsorption proved itself as the most feasible and efficient technique for the removal of dyes and other organic and inorganic pollutants from wastewater. Among the different sorbents, activated carbons have been reported to be the most efficient. Unfortunately, activated carbons are now expensive such that the trend is now to use agricultural byproducts as sorbents for dyes and other pollutants in potable and wastewater. To enhance the sorption capacities of agricultural byproducts, physical and/ or chemical modifications are made [23,24]. Grafting of the sorbent surface with functional groups or surfactant molecules has been done [25,26]. However, less attention has been devoted to the use of surfactants to modify the surfaces of agricultural byproducts for dye adsorption purposes.
The aim of this work was to modify orange peel (OP) with a cationic surfactant (cetyltrimethylammonium bromide, CTAB) before or after the pretreatment of raw OP with sodium hydroxide (NaOH) to enhance the adsorption capacity of the by-product and to investigate the adsorption of reactive blue 19 (RB19) dye on it. The study includes a characterization of the adsorbents and the determination of the factors affecting the sorption, including the contact time, the temperature, and the pH. The adsorption data were analyzed using the Langmuir, Freundlich and Tempkin equations. The kinetic characteristics of adsorption were studied with pseudo first- and second-order equations, the Elovich equation, and the intraparticle diffusion model.
2.1.1. Raw OP
The OP was obtained from a local fruit field. It was cut into small pieces, washed several times with distilled water and dried at 70℃ for 24 h. The product was then crushed and sieved through a screen to an average particle size of less than 0.5 mm and was designated as OP.
2.1.2. Chemical modification with sodium hydroxide
One hundred grams of dried OP was immersed in 1 L of NaOH for 24 h under occasional shaking. After decantation and filtration, this saponified product (NaOH-treated OP, SOP) was washed with distilled water until the pH of the filtrate reached 7.0.
2.1.3. Modification with cationic surfactant
Twenty grams of each sample (OP and SOP) was treated with 100 mL of CTAB, amounting to 1.24 g, approximately equivalent to a CMC value of 2.6 considering the reported CMC value (1.30 × 10-3 mol/L) [27]. Preliminary experiments showed that the amount of CTAB that had been sorbed remained unchanged even after the sample had been shaken for more than 2 h. The sorbent-CTAB mixture was stirred for 30 min. using a mechanical stirrer. The CTAB-OP and CTABSOP were washed several times with distilled water until they were Br?free, as indicated by an AgNO3 test. The samples were then dried in an oven at 80℃ .
2.1.4. Preparation of RB19 dye
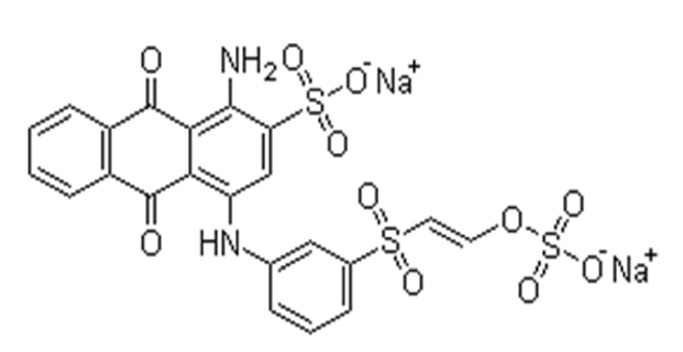
RB19 dye was used as an adsorbate in the form of a commercial salt, which is widely used in the textile industry. Its molecular formula is written as C22H16O11N2S3Na2 (mol.wt. 626.59 g/mol), the corresponding structure is illustrated in Fig. 1.
2.2.1. Instruments used for adsorbent characterization
Nitrogen adsorption isotherms at 77 K were constructed using a conventional volumetric apparatus. The sample was degassed at 70℃ for 6 h under a reduced pressure of 10-5 torr. The different types of functional groups were identified by Fourier transform infrared (FT-IR) spectroscopy in the range of 4000-400 cm-1 using a Perkin Elmer Paragon 1000 FT-IR spectrometer (USA), and the KBr disc technique. Surface morphologies of the adsorbents were identified using a JOEL JSM device.
2.2.2. Adsorption of RB19
The batch sorption experiments were conducted by mixing adsorbent (0.2 g) with 50 mL of dye solutions at the desired concentrations (20-300 mg/L) in a 100 mL sealed conical flask using a shaking thermostat machine at a speed of 120 r/min for an equilibrium time of 24 h. The effect of the solution pH value on the equilibrium adsorption of the dye was investigated by mixing 0.2 g of sorbent with 50 mL of dye solution between pH values of 2.0-8.0. In the kinetic experiments, a sample of dry adsorbent (~0.5 g) was added to 300 mL of the aqueous adsorbate solution at an initial pH of 4.0 and a temperature of 30℃ . The uptake of the adsorbate per unit mass of the sorbent was followed over time by withdrawing 2 mL of sample at fixed time intervals to determine the concentration at each time. Co, Ce and Ct were measured spectrophotometrically at a wavelength of 590 nm with a Shimadzu model UV-Vis spectrophotometer.
The amount adsorbed at equilibrium qe or at time (t) qt was determined from the corresponding initial concentration using Eq. (1).
Where, Co, Ce and Ct are the initial, equilibrium and concentration at time (t) (mg/L), respectively, V is the volume of the adsorption solution (L), and m is the mass of the sorbent (g).
3.1. The chemistry of the surface of OP, SOP, CTAB-OP and CTAB-SOP
The chemistry of the adsorbent is equally important to its textural properties in determining the adsorption capacity of this adsorbent, particularly in the case of adsorption from a solution.
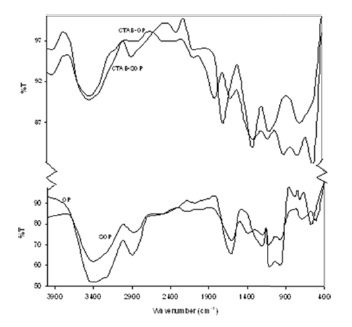
Infrared techniques have been used for the identification of adsorbents. Usually, the band at 3640-3510 cm-1 denotes the OH stretching of polymeric compounds. The band at 3000-2800 cm-1 is the stretching vibration of C-H. The band at 1740-1725 cm-1 is the stretching vibration of COO and C= O. The 1130-1000 cm-1 band is the vibration of C-O-C, C-O-P and O-H of the polysaccharides.
The FT-IR curves of OP, SOP, CTAB-OP and CTAB-SOP are shown in Fig. 2. This figure shows that some peaks disappeared after OP was pretreated with NaOH and that new peaks are observed after further cationic surfactant (CTAB) modification.
NaOH is good saponifying agent for the conversion of ester groups to carboxyl groups. It indicated the weakening intensity of the peak at 1744 cm-1 after the NaOH pretreatment, which can enhance the binding ability of the biomass. Fig. 2 also shows that the intensity of peaks 2926 cm-1 increased from 66 to 95.3 (44%) after modification of OP with CTAB and increased from 75.6 to 97.2 (29%) after modification of SOP with CTAB, indicating that the presence of the surfactant (CTAB) increases the number of ? CH2- groups.
The scanning electron microscope (SEM) micrographs clearly reveal the surface texture. SEM micrographs of the OP, SOP, CTAB-OP and CTAB-SOP samples are shown in Fig. 3. The surface morphology of OP (Fig. 3a) is different from that of SOP (Fig. 3b). After being treated with NaOH, SOP has a more irregular and more porous structure than OP and therefore a larger surface area. The surface areas of OP and SOP were determined to be 4.28 and 6.82 m2/g by the BET method. The surface morphology of the CTAB-OP and CTAB-SOP samples are rougher compared with the CTABfree samples. Also, the surface areas of OP and SOP are smaller than those of CTAB-OP (5.15 m2/g) and CTAB-SOP (8.25 m2/g). The existence of the CTAB layer on the surface
of the biomass leads to these morphological changes. It can be seen that the surfaces on the surfactant-modified OP and SOP samples are more uneven than those of the OP and SOP samples, which indicates that CTAB is chemically bonded and/or physically adheres to the surface of OP. A similar result was reported with copolymer-grafted OP [28].
3.2. Effect of solution pH on dye uptake
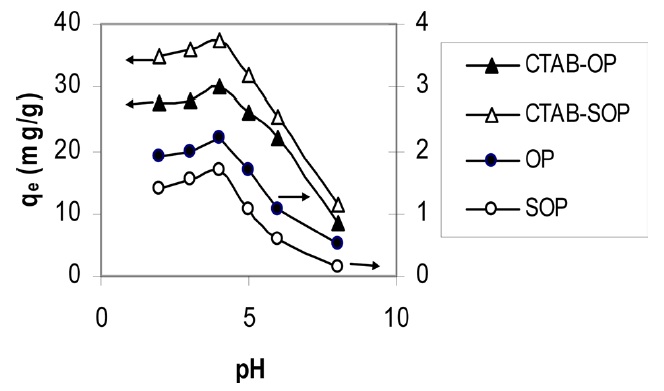
Fig. 4 shows the effect of the pH on the removal of RB19 from an aqueous solution. The figure shows that the maximum removal of RB19 occurs at pH = 4. A trend similar to that of the pH effect was observed for the adsorption of DR28 and acid violet on activated carbon prepared from coir pith [29,30]
as well as for the adsorption of direct blue 2B and direct green B on activated carbon prepared from mahogany sawdust [31]. As the surfaces of OP and CTAB- OP are both positively charged at a low pH value, a significantly strong electrostatic attraction is expected with the negatively changed anion RB19. On the other hand, an increase of the pH value led to an increase in the number of negatively change sites and a decrease in the number of positively charged sites. A negatively charged surface site on the OP or modified OP does not favor the adsorption of anion RB19 molecules due to electrostatic repulsion. Moreover, the decrease in the adsorption of anion RB19 with an increase in of the pH value is also due to the competition between the anionic dye and the excess OH ions in the solution. A pH of 4 was selected for the additional experiments.
3.3. Effect of the contact time and adsorption kinetics
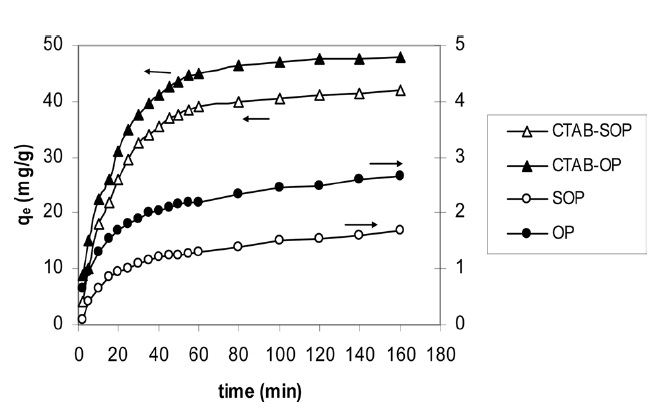
Fig. 5 depicts the amount of RB19 removed by OP and its modified forms at 30℃ and pH 4.0 as a function of the contact time. The figure shows that dye adsorption at the initial stages is quite rapid and then deceases with time, approaching a plateau. Evidently, equilibrium was attained after 30 min in the case of RB19 adsorption onto OP and SOP but after 60 min in case of the adsorption of the same dye onto CTAB OP and CTAB-SOP,
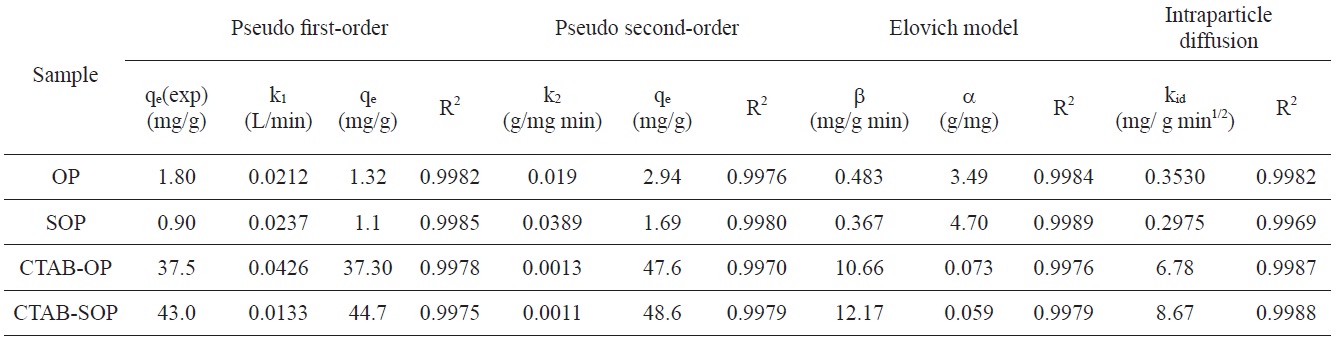
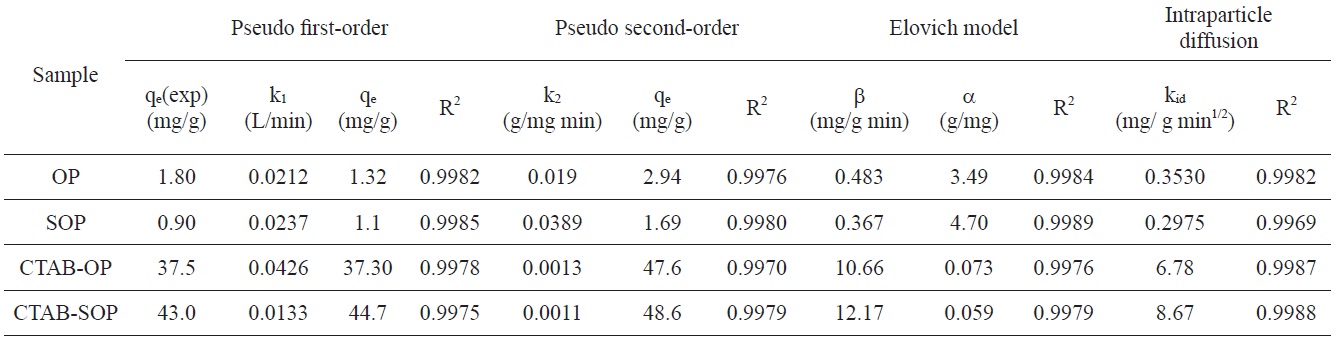
Kinetic constants of adsorption RB19 onto OP, SOP, CTAB-OP and CTAB-SOP with the pseudo first-order and second-order kinetics, the Elovich equation and intraparticle diffusion models
indicating completely adsorbed monolayers.
Determination of order of the dye adsorption process and the rate constant were obtained by applying Lagergren’s pseudo first-order kinetic model [32], a pseudo second-order kinetic model [33], the Elovich equation [34] and the intraparticle diffusion model [35]. The kinetic rate constants and the corresponding correlation coefficients R2 are listed in Table 1.
The Lagergren equation reads
where qe and qt (mg/g) are the amounts of the dye adsorbed at equilibrium and at time t, respectively, and where
The pseudo second-order equation is expressed as
where qe and qt (mg/g) are the amounts adsorbed at equilibrium and at time t, respectively, and where k2 (g mg-1 min -1) is the pseudo second-order rate constant. The calculated values are listed in Table 1.
Inspection of Table 1 reveals that the values of R2 obtained by applying the pseudo first- and pseudo second-order kinetic equations are comparable. However, the authors are more inclined to apply the pseudo first-order model because it gives calculated qe values of the same order of magnitude as those determined experimentally.
The Elovich equation is another rate equation. It is expressed as
where β is the initial adsorption rate mg (gmin)-1 and α is the desorption constant (gmin-1). Elovich plots of RB19 adsorption on the investigated adsorbents are shown in Fig. 6b. The figure shows that the qt versus lnt plots are linear, indicating the ap-
plicability of the Elovich equation to the present adsorption system. Table 1 lists the values of α and β for each dye-adsorbent experiment, showing that the β values of the dye-CTAB-OP and dye-CTAB-SOP are higher than the values of β obtained from the OP and SOP sorbents. On the other hand, the desorption constants α of the CTAB-modified sorbent are lower than those of CTAB-free sorbents.
The mechanism of the diffusion of dye molecules during adsorption was studied considering the intraparticle diffusion model, which is expressed as
where
Fig. 6c shows the initial curved portions followed by the linear portions. The initial curved portions of the plots refer to boundary layer diffusion, whereas the linear portions correspond to intraparticle diffusion. However, the linear portion does not pass through the origin, indicating that intraparticle diffusion is not the only process controlling dye sorption.
The values of kid listed in Table 1 indicate that modification of OP with CTAB to give CTAB-OP resulted in a considerable increase in
3.4. Equilibrium adsorption modeling
We have already determined the appropriate pH value for the sorption of RB19 for the sorbents investigated here (pH = 4). It was also shown from the kinetic curves in Fig. 6 that beyond 100-120 min of contact, no significant increase in the sorption over time is observed. This equilibrium time is much shorter than that reported for the adsorption of dyes by activated carbons, as the latter contain a large fraction of the surface located on microspores [1].
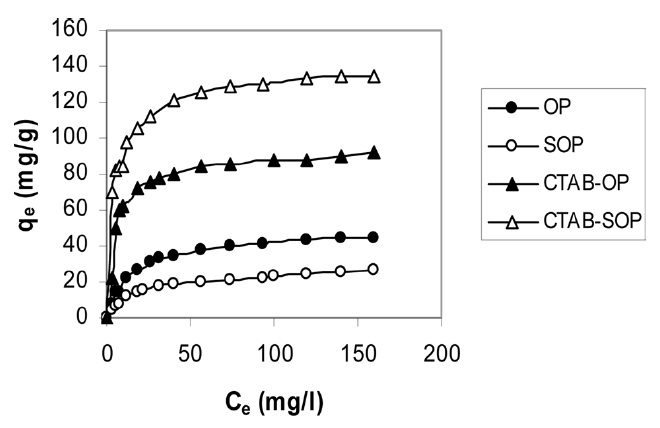
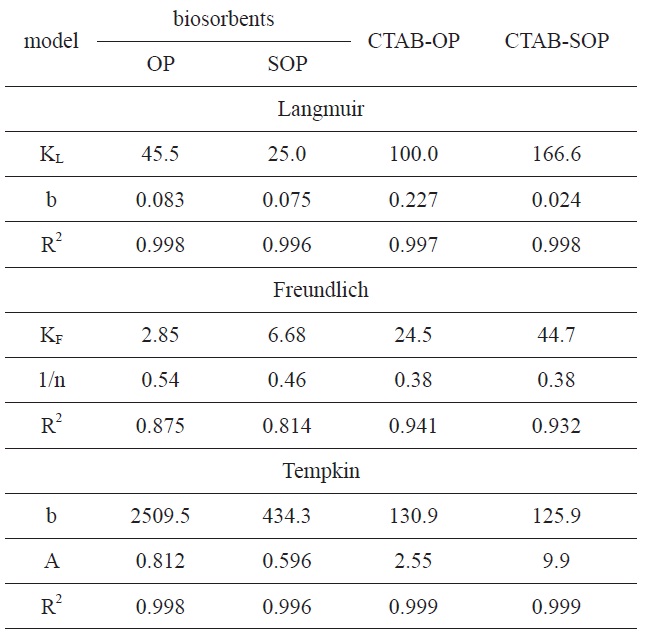
Typical sorption isotherms of RB19 dye on the investigated sorbents at 313 K are shown in Fig. 7. These isotherms are typical type L isotherms according to the classification reported by Zheng et al. [2] for adsorption from dilute solutions. Three models were applied for the equilibrium data of RB19: the Langmuir, Freundlich and Tempkin models.
The Langmuir adsorption model [36] assumes that adsorption occurs at specific homogeneous adsorption sites within the adsorbent and that intermolecular forces decrease rapidly with the distance from the adsorption surface. The model is based on the assumption that all adsorption sites are energetically identical and that adsorption occurs on a structur-
ally homogenous adsorbent. The linear form of the Langmuir equation is written as
where kL is a parameter related to the maximum adsorption capacity (mg/g), qe is the amount of dye sorbed at equilibrium per unit mass of sorbent, Ce is the equilibrium concentration of the dye in the solution (mg/L) and b is the equilibrium adsorption
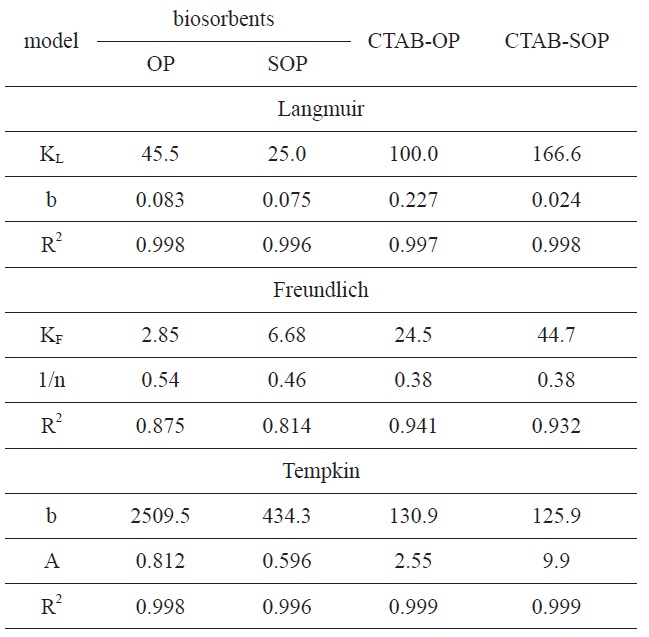
Langmuir, Freundlich and Tempkin model parameters for RB19 biosorption equilibrium on surfactant-modified OPs
constant related to the energy of the adsorption (L/mg) Fig. 8a.
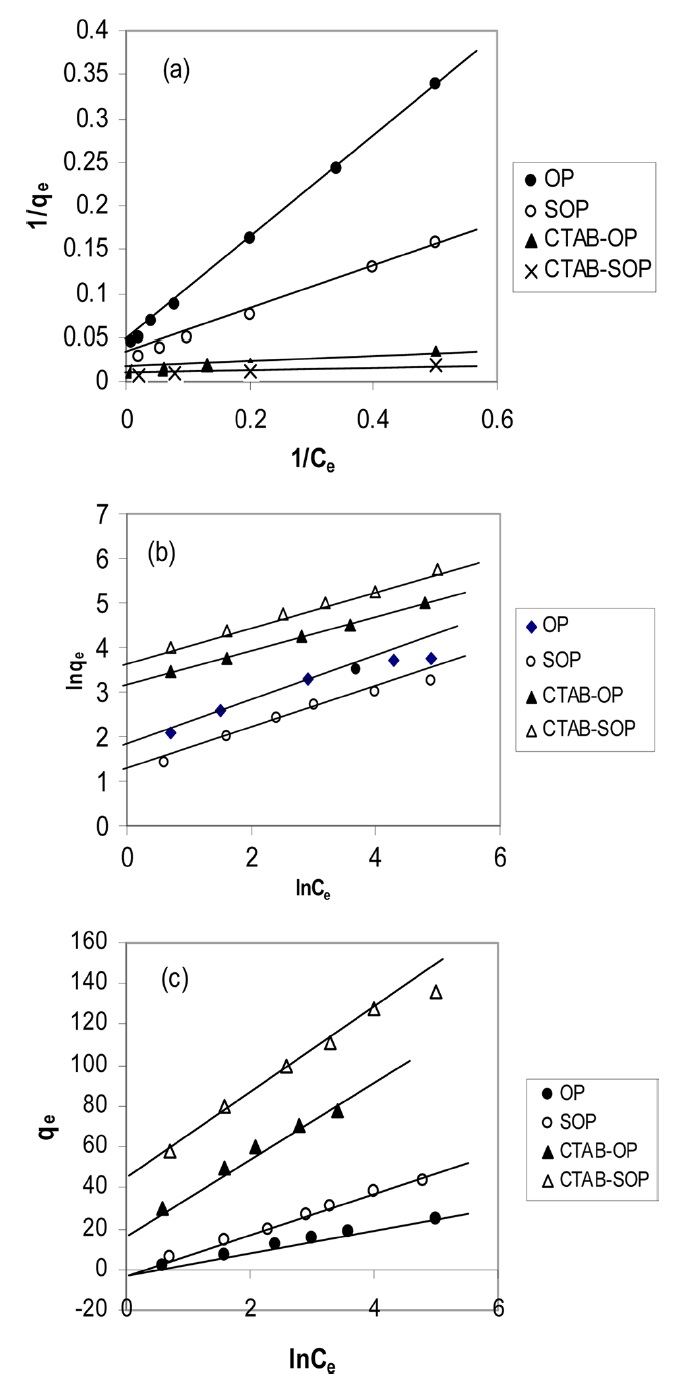
The Langmuir parameters and correlation coefficients of the experimental data are shown in Table 2. The values of R2 are ≥0.997, clearly indicating the excellent fitting of the adsorption data to Langmuir’s model. The maximum uptake of RB19 by SOP is lower than that taken by OP. The alkaline saponification decreased the RB19 uptake, which may be ascribed to the conversion of the ester groups to carboxylic groups on the surface of OP.
The modification of OP and SOP by CTAB brought about a 2.2-fold increase in the dye uptake amount by OP, whereas a 6.66-fold increase occurred by SOP. The chemical modification mechanism of OP and SOP can be explained as follows. The cationic surfactant (CTAB) combined with cellulosic hydroxyl groups of OP to form an ester linkage and introduce -CH2- groups to the fiber. As expected, the cationized OP was effective in removing anionic dyes from an aqueous solution. This behavior may be attributed to coulombic attraction between the cationized cellulose and the anionic dyes, whereas coulombic repulsion could occur in the case of cellulose as a result of the partial negative charge developed on the surface in the aqueous solution [37].
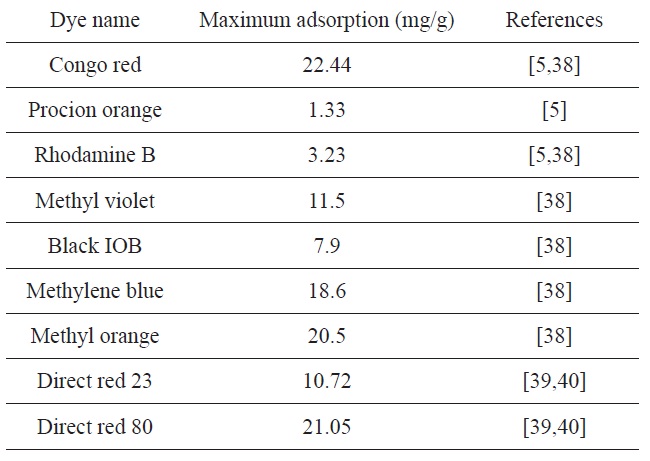
Unmodified OP was previously used to remove different dyes from aqueous solutions [5,38-40]. The maximum capacity of dye removal (mg/g) is listed in Table 3. The maximum capacity (166.6 mg/g) obtained for CTAB-modified OP is higher than that obtained for unmodified OP.
The adsorption data was also analyzed by the Freundlich model. The Freundlich model [41] is given by the equation
[Table 3.] Reported maximum dye adsorption amounts obtained for orange peel
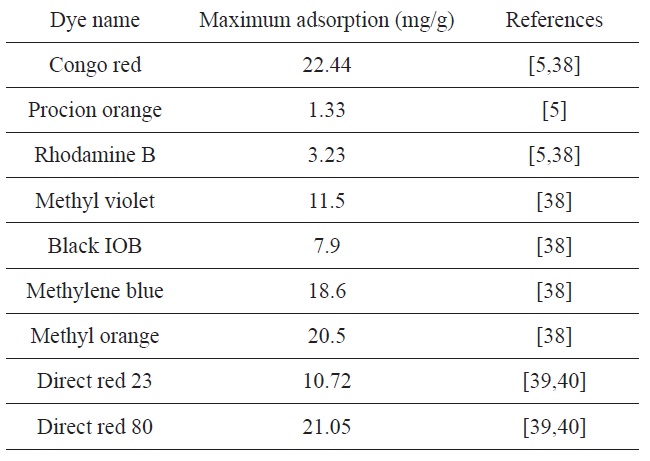
Reported maximum dye adsorption amounts obtained for orange peel
where qe is the amount adsorbed (mg/g), Ce is the equilibrium concentration of the adsorbate (mg/L), KF is a constant related to the maximum amount adsorbed and
is a measure of the binding energy between the sorbate molecule and the sorbent surface.
The applicability of the Freundlich sorption isotherm was also analyzed using the same set of experimental data by plotting lnqe versus lnCe (Fig. 8b). The data obtained from a linear Freundlich isotherm plot for the adsorption of the RB19 onto the investigated samples are given in Table 2. The correlation coefficients R2 are less than 0.886, indicating less fitting of the adsorption data to the Freundlich model. This can be taken as evidence that the adsorption of RB19 by the investigated biosorbent is chemical adsorption.
Tempkin’s adsorption model [42] was used to evaluate the adsorption potentials of the OP and its modified forms for RB19. Tempkin's equation suggests a linear decrease of the sorption energy as the degree of completion of the sorptional centers of an adsorbent is increased. The heat of adsorption of all the molecules in the layer would decrease linearly with coverage due to adsorbent-adsorbate interactions. The adsorption is characterized by a uniform distribution of the binding energies up to some maximum. The Tempkin isotherm is generally applied in the following form:
where β =
The correlation coefficients R2 are clearly higher than 0.995, indicating excellent fitting of the dye adsorption data to the Tempkin model. The heat of RB19 adsorption onto OP and its modified forms was found to decrease from 2509.5 to 131 J/mole to result in 434 to 126 J/mol for the CTAB-modified SOP. The R2 values obtained are comparable to those calculated from the Langmuir isotherm. Regarding values of the heat of adsorption as determined from the Langmuir and Tempkin models, the values are considerably higher than 45 J/mole, suggesting that dye adsorption by the investigated sorbents is of a chemical nature.
The adsorption of RB19 by CTAB-OP and CTAB-SOP was assessed at 303, 313 and 333 K. The adsorption increased with the increase in the temperature from 303 to 313 K and then decreased with a further rise of the adsorption temperature from 313 to 333 K. This may be explained as follows. The increased adsorption by the CTAB-modified sorbents with the increased temperature from 303 to 313 K may be ascribed to the enhancement of the rate of diffusion of the dye molecules to the adsorption sites. A further increase of the temperature from 313 to 333 K may be sufficient to weaken the binding energy between the sorbent sites and sorbate molecules, thus resulting in a considerable decrease in the amount adsorbed, i.e., desorption.
OP, SOP, and their CTAB-modified forms were prepared and characterized for their physical and chemical properties using nitrogen adsorption at 77 K, SEM, and FT-IR. The adsorption of the anionic dye RB19 was highly adsorbed on CTAB-modified forms, particularly at pH 4. The adsorption was of the chemisorption type and the adsorption data followed the Langmuir and Tempkin models and followed pseudo first-order kinetics.