The genus Ceramium is one of the largest genera in the rhodophytes, and monographic studies have added new members to the genus (South and Skelton 2000, Cho et al. 2003a, Cho and Fredericq 2006). Ceramium is charac-terized by cylindrical axial cells that are incompletely to completely covered by cortical cells, alternate to pseu-do-dichotomous branching, straight to inrolled apices, tetrasporangia produced from periaxial to cortical cells, spermatangia occurring on cortical nodes, and spherical cystocarps surrounded with involucral branches (Dixon 1960, Hommersand 1963, Womersley 1978, Cho et al. 2001).
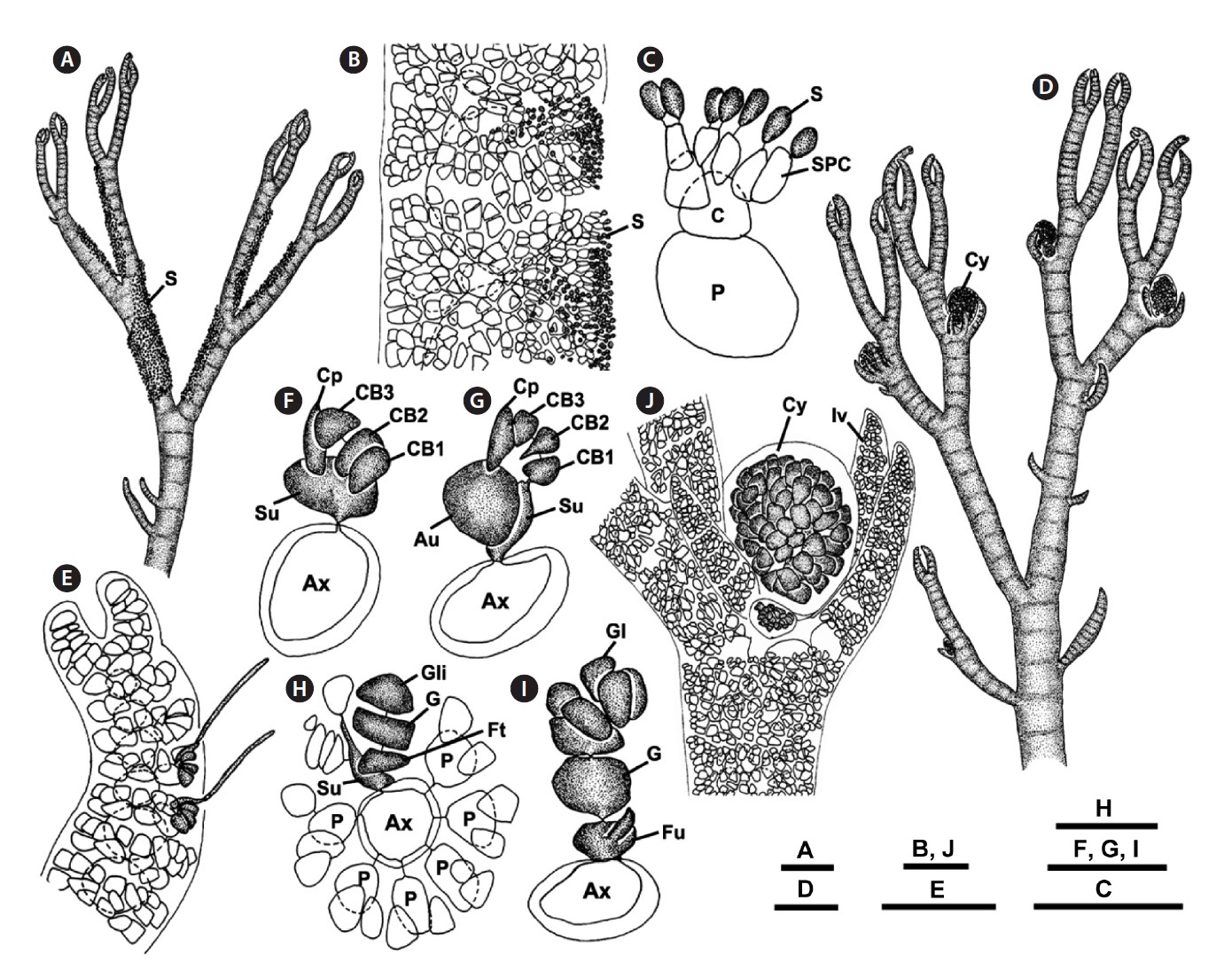
Approximately 60 Ceramium species are currently rec-ognized from the North Pacific Ocean (Cho et al. 2003b, Cho and Riosmena-Rodriguez 2008). Fifteen of them are known as complete corticated species. Of these species, C. washingtoniense Kylin is conspecific and synonymous with C. pacificum (Collins) Kylin (Carlile et al. 2010) and C. johnstonii Setchell & Gardner forms a series within a single species with C. sinicola Setchell et Gardner (Daw-son 1944, 1950). Cho et al. (2003b) examined the pattern of cortical cell formation and phylogenetic relationship of the C. sinicola complex (C. sinicola, C. interruptum, C. codicola). Although C. sinicola, C. interruptum, and C. codicola J. Agardh are known as a similar group in size, habit, and habitat, they were distinguished morphologi-cally based on the number of corticating filaments, de-velopment of cortical cells at the branching point, shape of the rhizoidal tip, and by molecular data based on rbcL, small subunit ribosomal RNA (SSU rDNA), and the RUBISCO spacer (Cho et al. 2003b).
We collected a complete corticated Ceramium species on Gracilaria from the subtidal zone in Bahia Magdalena, B. C. S., Mexico and observed details of the morphology and anatomy of the reproductive organs. We describe this species as Ceramium riosmenae sp. nov. based on its de-tailed morphology and rbcL gene molecular data.
Samples of unidentified Ceramium species were col-lected from Estero Santo Domingo, Lopez Nateos, Bahia Magdalena, B. C. S., and sorted with hosts under a ste-reomicroscope. Each identified sample was preserved in 4% formaldehyde / seawater for morphological observa-tions. Microscopic observations were made from materi-als stained with 1% aqueous aniline blue acidified with 0.1% diluted HCl. Voucher specimens used in this study were deposited in the herbarium of Chosun University (CUK), Gwangju, Korea, and Chungnam National Univer-sity (CNUK), Daejeon, Korea.
Genomic DNA was extracted from silica-gel samples using the G-spinTM Iip Genomic DNA Extraction kit (iN-tRON Biotechnology, Inc, Seongnam, Korea). The rbcL gene was amplified using the primer combinations F7-R753 and F6450-Rrbcst, as listed in Lin et al. (2001) and sequenced with the F7, F645, F993, R376, R753, R1150, and RrbcStart primers (Freshwater and Rueness 1994, Lin et al. 2001, Gavio and Fredericq 2002). Polymerase chain reaction (PCR) and sequencing protocols are as described in Cho et al. (2003b). Sequences were determined for both forward and reverse strands using the ABI Prism 3100 Ge-netic Analyzer (Applied Biosystems, Foster City, CA, USA). A new rbcL sequence was obtained for Ceramium riosme-nae and was deposited in the EMBL / GenBank under ac-cession number JN712693.
All rbcL sequence data were compiled, and the se-quences were aligned in the Genetic Data Environment (GDE 2.2) program (Smith et al. 1994). Maximum likeli-hood (ML) analyses were performed using PAUP*4.0b10 for Macintosh and UNIX (Swofford 2003). We performed a likelihood ratio test using MODELTEST 3.06 (Posada and Crandall 1998) to determine the best available model for the rbcL data. ML analyses were performed using the GTR + G model. A bootstrap analysis was conducted by performing replicate ML searches, with random stepwise addition of sequences, using the same search condition as described above. We succeeded in performing 500 bootstrap replicates with the rbcL data.
Diagnosis. Thalli epiphytici, erecti, 0.7-2.0 cm alti, hostem per rhizoidea curta, penetrantibus, pseudodichotome ra-mosi; intervallum ramificans 8-16 ad lateralem axes 7-15; corticatio completa; periaxiales cellulae 7-8, omnes forma-tae initia corticalia 5; rhizoidea formata cellulis periaxialus, elongate, multicellularia; tetrasporangiis cruciata, immers-es; spermatangia ad apices cellulis parentalibus spermatan-giorum in cellulis corticalibus extimis exorientibus; cysto-carpia prope apices, circumcincta 5-6 ramis involucralibus.
Thalli epiphytic, erect, 0.7-2.0 cm high, attached to the host by short, branched, penetrating rhizoids, dichoto-mously branched; branching interval 8-16 nodes for main axes, 7-15 nodes for lateral axes; cortication complete; 7-8 periaxial cells each producing five cortical initials; rhizoids produced from periaxial cells, elongate, multicellular; tet-rasporangia cruciately divided, completely immersed in the cortex; spermatangia in clusters near the apices; the spermatangial parent cells borne on outermost cortical cells; cystocarps produced near the apices, surrounded by 5-6 involucral branchlets.
Holotype: CUK245, 1 m subtidal zone in Estero Santo Domingo, Lopez Nateos, Bahia Magdalena, B. C. S., Mex-ico, collected by T. O. Cho and R. Riosmena-Rodriguez (1 June 2000, TC245).
Additional isotypes: Estero Santo Domingo, Lopez Na-teos, Bahia Magdalena, B. C. S., Mexico (T. O. Cho and R. R. Rodriguez, 1 June 2000, CNUK C005121, tetrasporangial; C005110, female; C005111, male and female; C005114, tetrasporangial and female).
Etymology: The name Ceramium riosmenae is chosen to honor Dr. Rafael Riosmena-Rodriguez, UABCS, Mexi-co, for his significant contributions to the understanding of phycology.
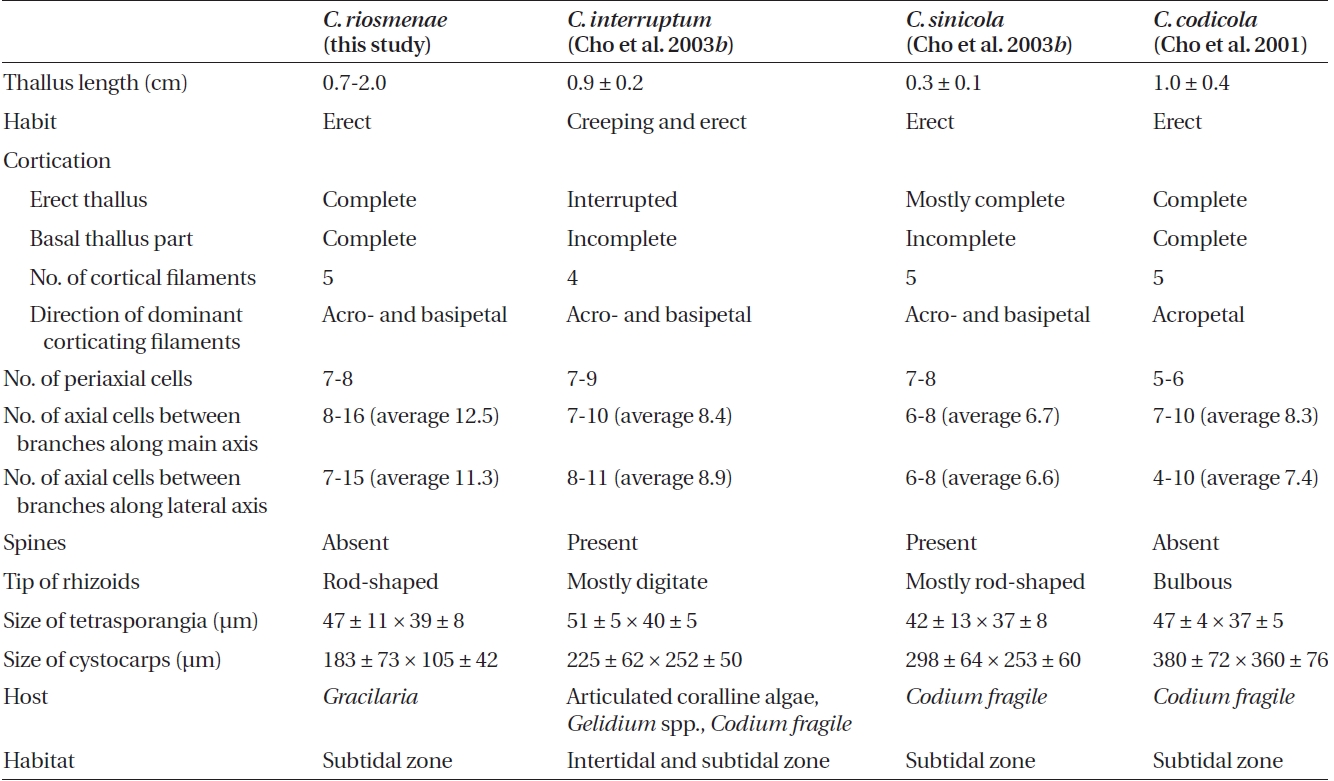
[Fig. 1.] Ceramium riosmenae sp. nov. (A) Vegetative thallus. (B) Apical regions. (C) Upper thallus part with complete cortication. (D) Lower thallus part with incomplete cortication. (E-G) Cross sectional views through the cortical node of an axis showing the alternate sequence of the eight periaxial cells. (H-K) Sequence information for the five cortical initials from a periaxial cell. (L) Rhizoids showing slender, rod-shaped and non-bulbous rhizoids. (M) Tetrasporangial thallus. (N & O) Detail of cortex showing tetrasporangia completely immersed in upper (N) and middle (O) thallus. (P & Q) Cross sectional views showing alternate sequence of tetrasporangia at the cortical node. (R) A cortical filament showing tetrasporangia produced from periaxial and cortical cells. Ax, axial cell; C1-5, sequence of cortical cell formation; P, periaxial cell; P1-7, sequence of periaxial cell formation; R, rhizoid; T, tetrasporangium. Scale bars represents: A-C, K, M & P-R, 50 μm; E-J, 25 μm; D, L ,N & O, 100 μm.
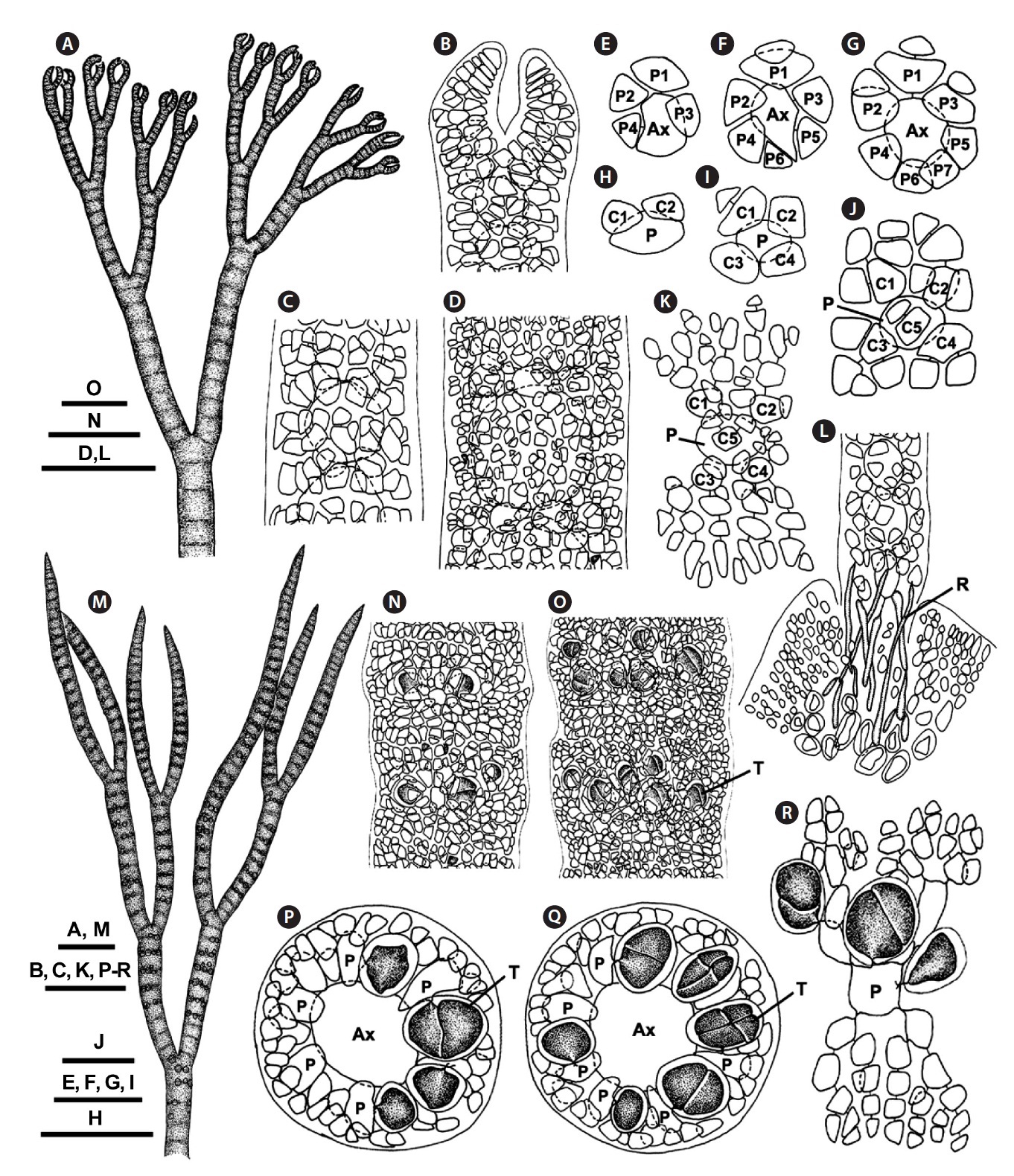
[Fig. 2.] Ceramium riosmenae sp. nov. (A) Male thallus. (B) Surface of cortex bearing spermatangia on adaxial side. (C) Cross section showing spermatangia developed from cortical cells. (D) Female thallus bearing cystocarps. (E) Apical region bearing procarps on abaxial side. (F) Four-celled carpogonial branch. (G) Formation of auxiliary cell from supporting cell. (H) Formation of gonimoblast initial and foot cell. (I) Formation of first gonimolobe. (J) Mature cystocarp surrounded by involucral branches. Au, auxiliary cell; Ax, axial cell; C, cortical cell; CB1-3, carpogonial branch cells; Cp, carpogonium; Cy, cystocarp; Fu, fusion cell; Ft, foot cell; G, gonimoblast; Gl, gonimolobe; Gli, gonimolobe initial; Iv, involucral branches; P, periaxial cell; S, spermatangium; SPC, spermatangial parent cell; Su, supporting cell. Scale bars represent: A, D, 200 μm; B, E, J, 50 μm; C & F-I, 25 μm.
Morphological observations
Vegetative plants: Thalli are rose-pink in color, 0.7-2.0 cm high, epiphytic, attached to Gracilaria by short, branched, penetrating rhizoids. They mostly consist of erect pseudodichotomous axes on host (Fig. 1A & 3A). Apexes are blunt, forcipulate, and incurved (Fig. 1B & 3B). The axial cells are spherical to cylindrical. Seven to eight periaxial cells are cut off obliquely from the upper part of each parent axial cell and remain at the nodes after axial cell elongation (Fig. 1E-G). All periaxial cells produce corticating filaments that form the cortex. Five primary cortical cells are produced from each periaxial cell in an alternate sequence (Fig. 1H-K, 3D & E). The first two primary cortical cells are cut off obliquely from the anterior end of periaxial cells and grow acropetally (Fig. 1H); the second two are produced obliquely from the posterior end and grow basipetally (Fig. 1I); the fifth one is produced on the outside face of periaxial cells (Fig. 1J). The acropetal and basipetal corticating filaments are both 5-7 cells long (Fig. 1K). The mature cortex is com-plete through all thallus (Fig. 1C, D & 3C).
Branches are pseudodichotomous. Branching takes place at intervals of 8-16 (average, 12.5 cells) axial cells in the main axes and at intervals of 7-15 (average, 11.3 cells)
[Fig. 3.] Ceramium riosmenae sp. nov. (A) Vegetative thallus. (B) Apical region. (C) Cortical node in the middle. (D & E) Cortical filaments showing five cortical initials from periaxial cell. (F) Basal part of erect axis on Gracilaria. (G) Rizoids produced from periaxial and cortical cells. (H) Tetrasporangial thallus. (I & J) Cross sectional views showing tetrasporangial arrangement. (K & L) Cortex showing completely immersed tetrasporangia. (M) Male thallus. (N) Cross section view through axis of male thallus. (O) Surface of cortex bearing spermatangia. (P) Female thallus. (Q) Cross sectional view of female axis with early stage cystocarp. (R) Mature cystocarp surrounded by involucral branchlets. Ax, axial cell; C1-4, sequence of cortical cell formation; Cy, cystocarp; Ft, foot cell; Gl, gonimolobe; Iv, involucral branches; P, periaxial cell; R, rhizoid; S, spermatangium; Su, supporting cell; T, tetrasporangium. Scale bars represent: A, P & Q, 500 μm; B, K & L-O, 100 μm; C, F, G, I, J & N, 50 μm; D & E, 25 μm; H, 700 μm; R, 200 μm.
axial cells on lateral axes. Adventitious branchlets are very rare.
Rhizoids are multicellular, uniseriate, and rod-shaped (Fig. 1L). They are slender, branched, and non-bulbous; produce extensions in bundles on Gracilaria sp. and are of various lengths depending upon the texture of the host (Fig. 3F & G). Rhizoids are produced from periaxial and cortical cells located on the basal region of erect axes.
Reproductive plants: In tetrasporic plants, tetraspo-rangia are completely immersed in the cortex (Fig. 1M & 3H). They are whorled at the nodes of the upper part and scattered irregularly in the middle thallus region (Fig. 1N, O, 3K & L). They are produced initially from periaxial cells in the upper part (Fig. 1P, Q, 3I & J) and then from inner cortical cells in the middle part (Fig. 1R). One tetraspo-rangium develops from each tetrasporangial parental cell. Tetrasporangia are cruciately divided, spherical to ellipsoidal, and average 47 ± 11 × 39 ± 8 μm excluding the sheath.
In male plants, spermatangia are first produced adaxi-ally and later make a whorl around the cortical axis in the typical pattern described for the genus (Fig. 2A, B & 3M-O). Spermatangial parent cells develop from cortical cells and produce 1-2 spermatangia terminally (Fig. 2C). Sper-matangia are colorless and elliptical to spherical.
In female plants, cystocarps are borne on the abaxial face of the upper thallus and are surrounded by 5-6 fin-ger-like branchlets with incomplete cortication in the typical pattern described for the genus (Fig. 2D, E & 3P). After presumed fertilization, the supporting cell enlarges, cutting off an auxiliary cell (Fig. 2F & G). The auxiliary cell then divides into a foot cell and a gonimoblast initial (Fig. 3Q). The gonimoblast initial cuts off the first gonimolobe terminally (Fig. 2H & I). Mature cystocarps are spherical, 183 ± 73 μm long, and 105 ± 42 μm in diameter and com-posed of carposporangia transformed from gonimolobe cells (Fig. 2J & 3R).
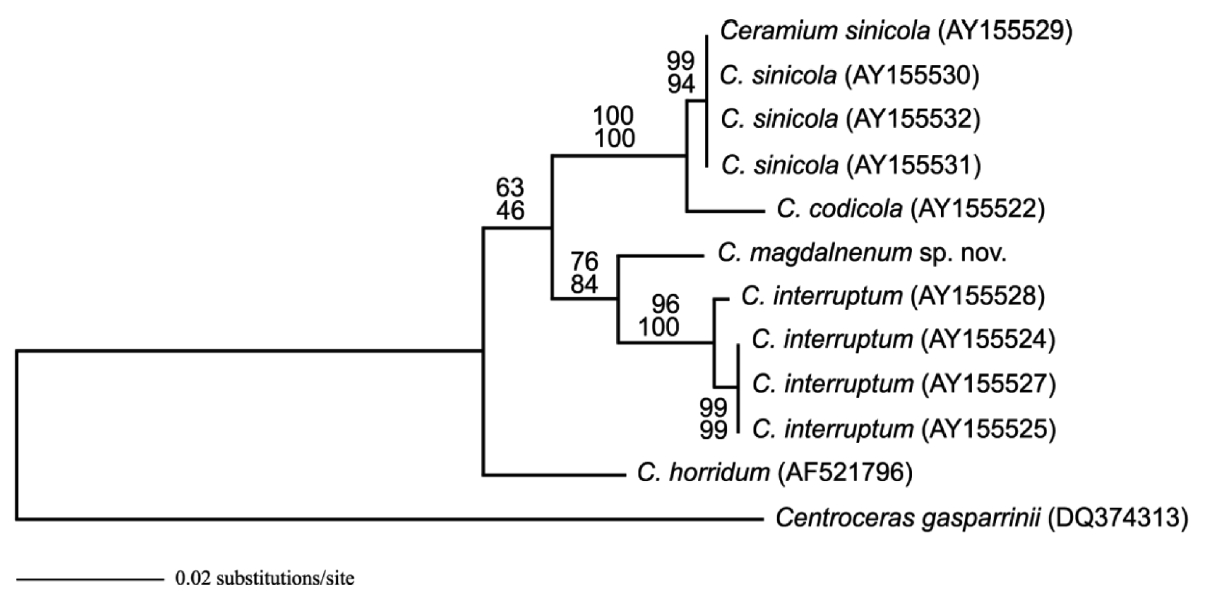
Molecular analyses: The 1,431 rbcL bp portion of the 1,467-bp rbcL gene (97.5% sequenced) analyzed included 67 parsimony informative sites. The phylogenetic trees were obtained from alignment of the rbcL sequences newly generated and downloaded from GenBank. Cen-troceras gasparrinii was selected as the outgroup. C. rios-menae differed from C. interruptum by 2.4-2.5% sequence divergence, from C. sinicola by 3.7-3.9% sequence diver-gence, and from C. codicola by 4.2% sequence divergence. Topologies of ML and maximum parsimony trees were congruent in the phylogenetic position C. riosmenae, C. horridum, C. interruptum, C. sinicola, and C. codicola (Fig. 4).
Ceramium riosmenae sp. nov. is described from Baja California Sur, Mexico based on morphological and mo-lecular data. C. riosmenae is recognized by its incurved apices, thallus with penetrating rhizoids on Gracilaria, rod-shaped rhizoidal tips, 7-8 periaxial cells, five cortical initials per periaxial cell, complete cortication through-out, an average of 11-12 segments between branching points, erect, rare adventitious branchlets, and cruciate tetrasporangia. The entity of C. riosmenae is strongly sup-ported by the phylogenetic analyses inferred from the rbcL sequences.
Nine Ceramium species with complete cortication have been reported from Baja California and the Gulf of California, Mexico: C. bicorne Setchell et Gardner, C. codicola, C. codiophila Setchell & Gardner, C. horridum, C. interruptum, C. obesum Dawson, C. pacificum, C. sini-cola, and C. viscainoense Dawson. Except C. pacificum, most are tiny and some are closely related by similar mor-phology in habit and cortication. Although these species are similar to C. riosmenae sp. nov. in size, they are dis-tinguished by their specific characters: C. bicorne by the development of two sharp points after the last division of the apical cell on the forcipate branches (Setchell and Gardner 1924); C. codiophila by mostly simple thalli and rhizoids that are more or less bulbose at the tip (Setch-
ell and Gardner 1937); C. horridum by the acute, spine-like growing points at their apices (Setchell and Gardner 1924); C. obesum by its creeping habit morphology and rhizoidal attachment among mat-forming small algae (Dawson 1950); and C. viscainoense by the divergent api-ces and the dense massing of short, secondary branches in the upper parts of the plant (Dawson 1950).
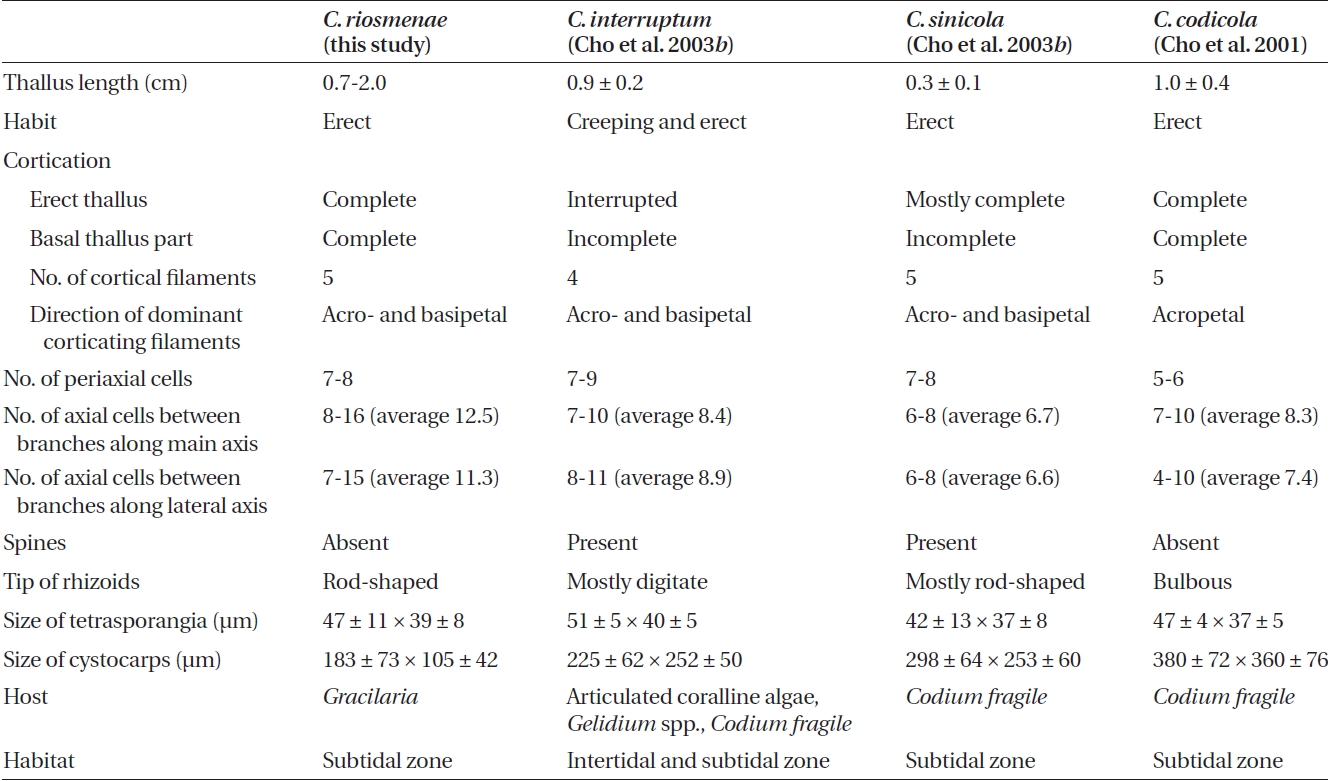
Our observations indicate that the degree of cortica-tion and rhizoid shape are important features for distin-guishing C. riosmenae from three similar-looking species such as C. interruptum, C. sinicola, and C. codicola (Table 1). Cho et al. (2003b) collected C. interruptum, C. sinicola, and C. codicola from the northeast Pacific and observed the detailed developmental pattern of the cortical cells and the rhizoids related to host texture. Although Dawson (1944) compared the degree of cortication in these three species and was of the opinion that they formed a series within a single species, Cho et al. (2003b) distinguished them clearly after detailed observations of their morphol-ogy. Our C. riosmenae is also separated from C. interrup-tum with interrupted cortication at branching points and on the lower thallus part and from C. sinicola with incom-plete cortication only in the lower thallus. Although C. riosmenae has an erect thallus, five cortical initials, and complete cortication throughout as with C. codicola, C. riosmenae with penetrating rhizoids on Gracilaria is dif-ferent from C. codicola with bulbous rhizoids on Codium. Some Ceramium species have distinct rhizoids that are closely related to host texture (Cho et al. 2003b).
Our rbcL sequences revealed sufficient sequence di-vergence among C. riosmenae, C. interruptum, C. sini-cola, and C. codicola to warrant species recognition for C. riosmenae. The rbcL sequence divergences (2.4-3.9%) between C. riosmenae and these three species are similar to other Ceramium species (2.5-3.8%) (Cho et al. 2003b) and within the range of other red algae, e.g., between spe-cies of Porphyra (1.3%) (Brodie et al. 1996) or Grateloupia (2-10%) (Gavio and Fredericq 2002). Our rbcL sequence ML tree also distinguished C. riosmenae as a distinct spe-cies with robust bootstrap support from other complete corticated Ceramium species.