Monstrilloids are basically endoparasitic copepods, before free-living, non-feeding adult stage (Boxshall and Halsey, 2004). Polychaetes and prosobranch mollusks have been known as their hosts. Recently, monstrilloids were also found in sponges (Huys et al., 2007), and were reported to be highly prevalent within the mantle of brown mussels (Suarez-Mora-les et al., 2010). Although the generic nomenclature is still unsettled, 116 species of four genera are currently recognized in the single family Monstrillidae Dana, 1849 (Razouls et al., 2005-2012; Suarez-Morales, 2011). Among the four genera, the major ones are
In Korea, taxonomic study on monstrilloid copepods is still entirely lacking. As a preliminary result from nocturnal sur-veys using a light trap on the east and south coasts of South Korea, we describe a new monstrilloid species belonging to the genus
Materials examined in the present study were collected using a light trap. The light trap is made with a PVC pipe (10 cm in diameter, 42 cm long) and a flashlight for SCUBA divers, placed on rocky or sandy bottom of 3-6m in depth at the end of piers or seawalls in small harbors after sunset. The samples caught in the light trap for overnight were filtered through a plankton net with 64 μm mesh at dawn. Monstrilloid cope-pods were fixed in 4% buffered formalin or 95% ethanol in the field and later sorted under a stereomicroscope in labora-tory. Most specimens were transferred to 80% ethanol for storage or for being deposited in the institute.
Specimens were dissected and mounted in lactophenol on the Cobb slide, after treatment in a solution of 10% glycerin-90% ethyl alcohol for 1-2 days. Dissection was performed using two needles made from a 0.3 mm-diameter tungsten wire, sharpened by electrolysis (Huys and Boxshall, 1991). Mounted specimens were observed using a differential inter-ference contrast microscope (BX-51; Olympus, Tokyo, Japan) equipped with Nomarski optics. All drawings were made with the aid of a camera lucida. Measurements were carried out with a digital camera for microscope (Cool SNAP 5.0M; Roper Scientific Co., Tucson, AZ, USA) and a calibration software QCapture Pro (ver. 5.0, Media Cybernetics Inc., Bethesda, MD, USA).
The type material of the new species is deposited in The National Institute of Biological Resources (NIBR), Korea (nos. NIBRIV0000245160 to NIBRIV0000245161). Additional materials are deposited in the Department of Biological Science, Daegu University (nos. DB20039 to DB20041), Korea.
Terminology for general copepod morphology follows Huys and Boxshall (1991). The antennular armature is des-cribed according to the nomenclature proposed by Grygier and Ohtsuka (1995).
Order Monstrilloida Sars, 1903
Family Monstrillidae Dana, 1849
Genus Cymbasoma Thompson, 1888
Cymbasoma striifrons new species (Figs. 1-3)
Material examined. Holotype: Adult female (NIBRIV0000 245160), undissected, ethanol-preserved; Korea: Gangwon-do, Samcheok-si, Gyo-dong, Hujin Harbor, 37° 27′52.79′′N, 129° 10′43.41′′E, 26 Jul 2010, Chang CY, Lee DY, Yoo JG.
Paratypes: 2 females (NIBRIV0000245161), undissected, ethanol-preserved, same collection data as in holotype. Additional material: 2 females (DB20039, 20040), dissected on slides, same collection data as in holotype; 1 female (DB20 041), Gyeongsangbuk-do, Geoje-si, Nambu-myeon, Ssang-geun dock, 34° 5′50.69′′N, 128° 35′10.71′′E, 6 Aug 2011, Chang CY, Yoo JG.
Etymology. The specific name
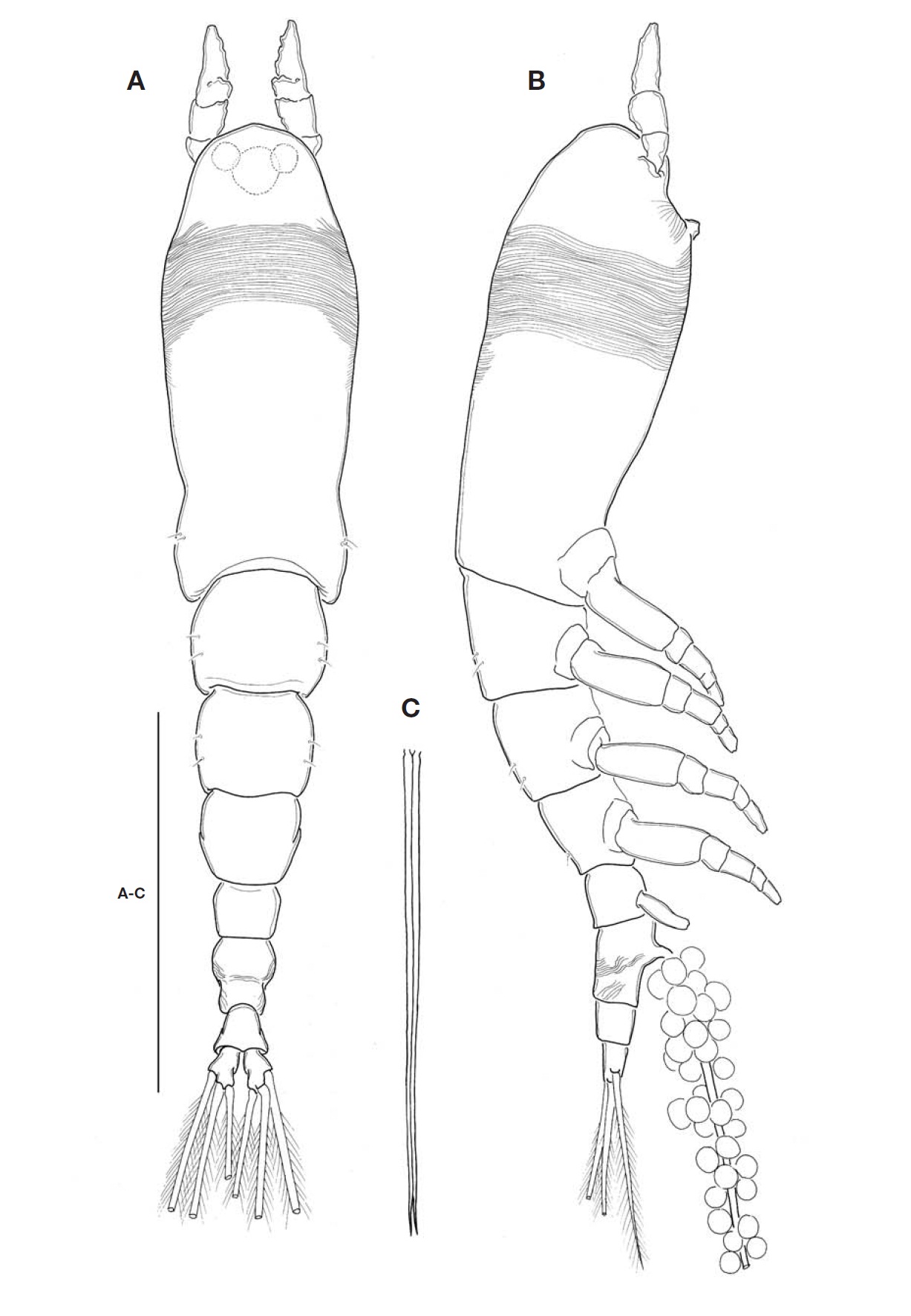
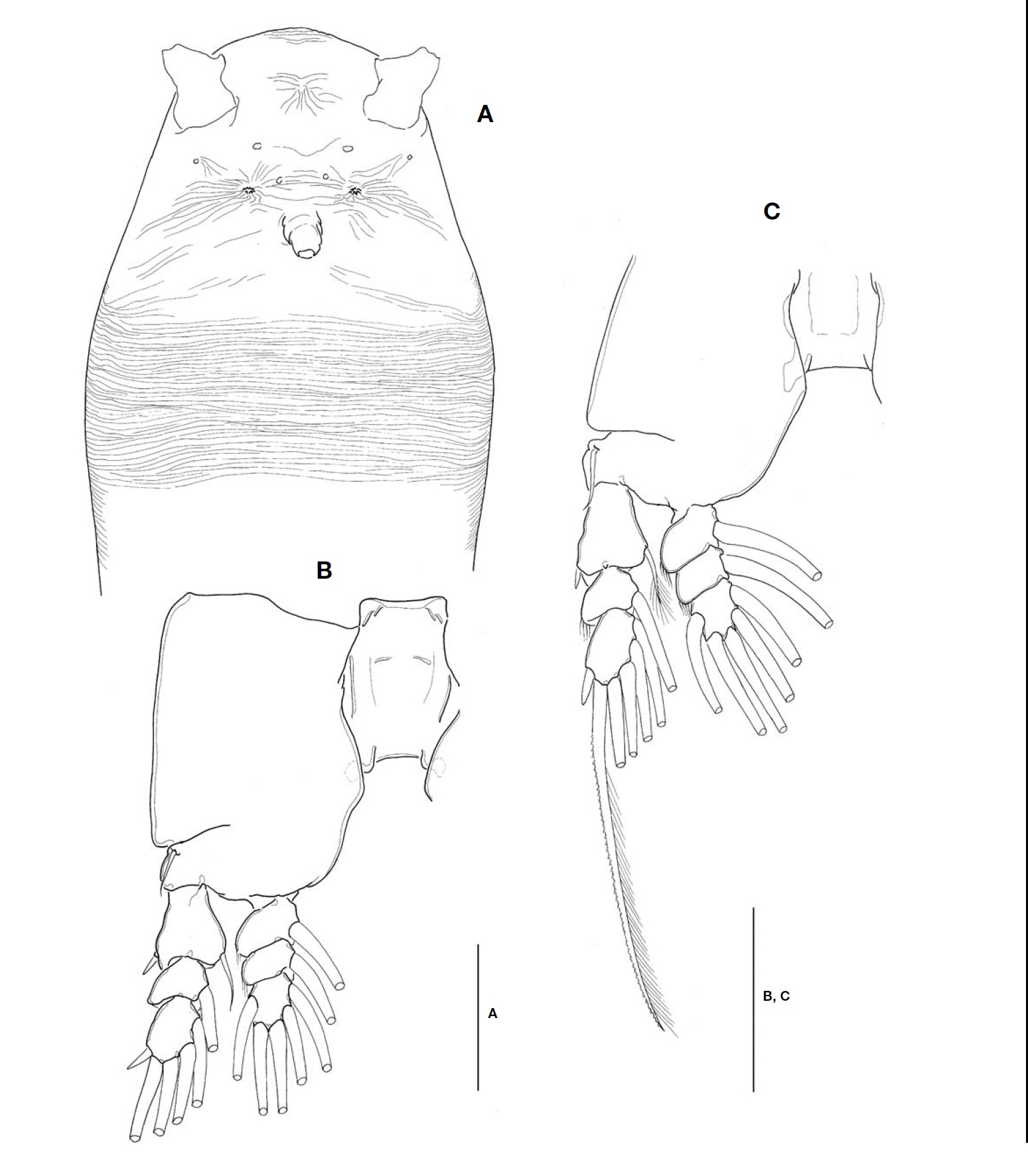
Description. Female: Body (Fig.1A, B) rather slender and relatively small, 1.1-1.4 mm long (mean 1.28 mm, standard deviation 0.12, n=6), measured from anterior end of cephalo-thorax to posterior margin of caudal rami, excluding caudal setae. Cephalothorax relatively short, accounting for 48% of total body length; anterior third swollen laterally. Anterior part of dorsal surface of cephalic region nearly smooth, ex-cept for a pair of short sensilla and fine wrinkles near anterior margin of cephalothorax. Nauplius eye well developed, round in dorsal view, about 0.06 mm in diameter. Ocelli beside nau-plius eye round, fairly apart to each other, separated by dis-tance equal to 75% of eye diameter, about 120% the diame-ter of each ocellus, in dorsal view. Paired ventral nipple-like processes situated between antennular bases and oral papilla (Fig.3A), just behind eyes; 2 small sensory pores present between the processes. Oral papilla lying somewhat anterior-ly, 0.17 of way back along cephalothorax (Figs.1B, 3A). Weak wrinkles present around oral papilla and nipple-like processes. Anterior part of cephalothorax with a wide band of fine transverse cuticular striations running around the body, covering almost a quarter (23-25%) of cephalothorax.
Antennules (Figs.1A, B, 2B) short and relatively stumpy, about 15% of total body length, and about 31% as long as cephalothorax; 4-segmented, second and third segments not well defined or partly fused; ratio of lengths of segments 13.3: 28.9 : 14.4 : 43.4=100. Armature: segment 1 with 1; segment 2 with 2d1-2, 2v1-3, IId; segment 3 with 3, IIIv, IIId; segment 4 with 4v1-3, IVv, IVd, 4d1-2, 4aes, Vm, Vd, Vv, 5, 61-2, b1-6, 6aes; distal 2 chela-like spines (61 and 62) with different lengths, length ratio 1.4-1.5.
Cephalothorax incorporating first pedigerous somite. Pro-some compring cephalothorax and 3 thoracic somites, each bearing pair of biramous swimming legs, with both endopods and exopods 3-segmented. Coxa of legs 1-4 lacking seta; in-tercoxal sclerite subrectangular, with paired lateral lobes slightly produced distally and smooth distal margin; both an-terior and posterior surfaces smooth without transverse spin-ule or setule row. Coxa and basis not fully devided. Basis lacking seta distomedially; outer proximal seta slender, naked. First and second endopodal segments of legs 1-4 swollen outer posteriorly with hairs along corner. Outer distal spines on first and third exopodal segments of legs 1-4 feeble, not ornamented with secondary spinules; inner distal seta on first exopodal segments of legs 1-4 slender and short, slightly ex-ceeding over distal margin of second exopodal segment; outer distal setae on third exopodal segments of legs 1-4 spiniform, with heterogeneous ornamentation along outer and inner margins. Seta/Spine armature on swimming legs 1-4 as fol-lows (Roman numerals indicate number of spines, and Arabic numerals indicate number of setae):
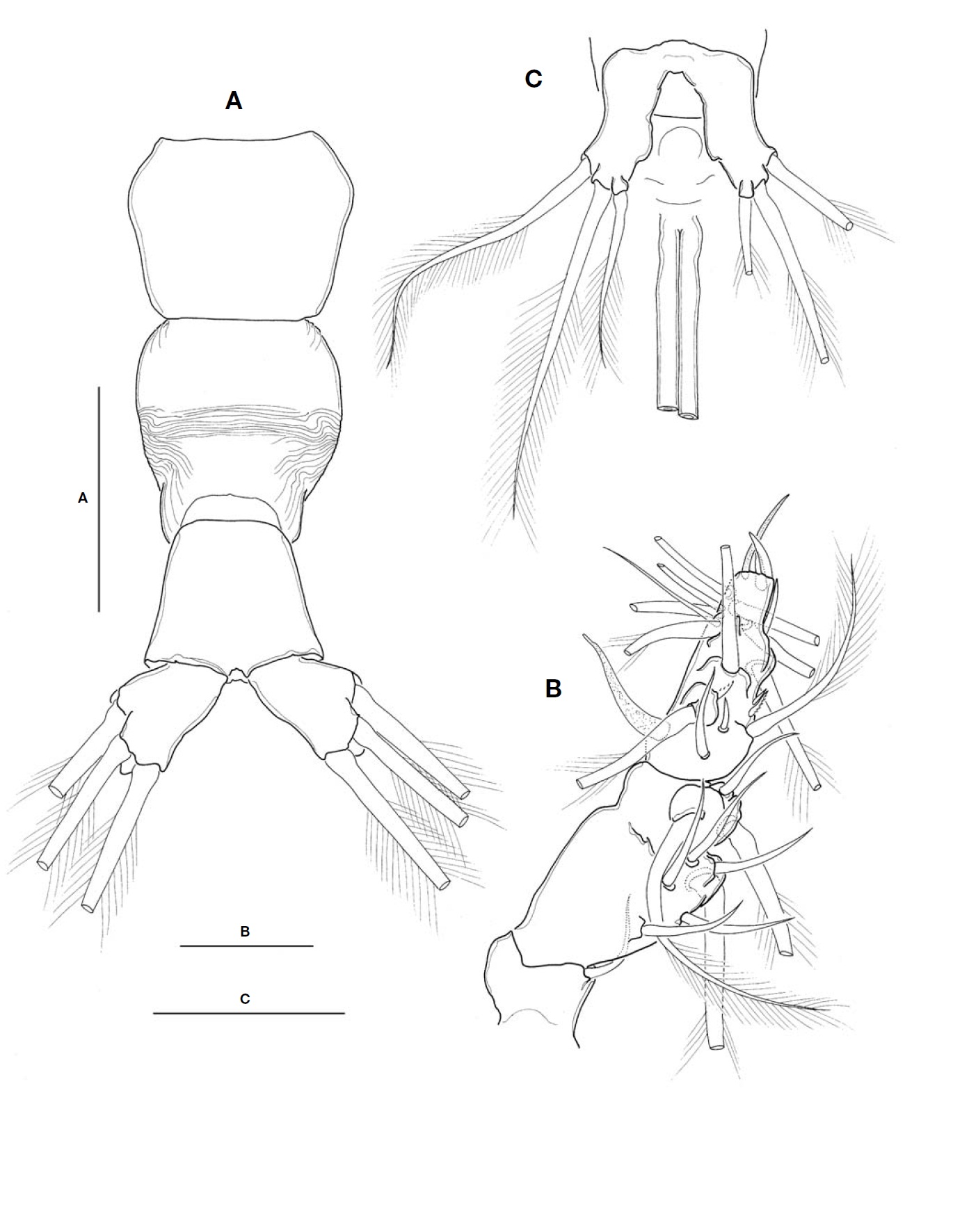
Urosome (Fig.2A) consisting of fifth pedigerous somite, genital double-somite, anal somite, with caudal rami; ratio of lengths 32.3 : 40.4 : 27.3=100. Genital double-somite strongly swollen in the anterior half, rather globose, with transverse wrinkles in the middle of dorsal surface and lateral sides of
posterior half; outer distal corner not produced (Fig.2A). Genital double-somite bearing pair of long ovigerous spines, inserted on middle of ventral surface, basally separated, re-presenting nearly half (49.4%) of total body length, about 2.7
times longer than urosome, with tips pointed, not-swollen (Figs.1C, 2C), extending slightly beyond tips of caudal setae. Anal somite trapezoid; lacking wrinkles or striae both on dor-sal and ventral surfaces; lateral margin nearly smooth, with-out apparent notch.
Leg 5 (Fig.2C) single lobed, medial margin slightly swol-len, representing vestige of endopodal lobe; armed with 3 plumose setae, comprising 2 outer subequal in length and breadth, and innermost one smallest and about 1/2 times as long as others.
Caudal rami (Fig.2A) highly divergent; ramus subquadrate, with slightly concave inner margin, 1.5 times longer than wide; furnished with 2 terminal and 1 lateral setae, subequal in length and breadth.
Male: Unknown.
Remarks. This species is assigned to the genus
Although the genuine identity of
As far as my knowledge, the distinguishable band of trans-verse striations around the body on the forehead, the decisive character of
Although the band of transverse striation on the forehead is rather faint and very narrow,
This species is the 18th monstriiloid species recorded from the Far-East Asian waters in the northwest Pacific, and ele-venth species described as new to science in the area (cf. Razouls et al., 2005-2012; Suarez-Morales, 2011).