Gastrointestinal cancer refers to malignant conditions of the gastrointestinal tract, including the esophagus, stomach, biliary system, pancreas, bowels, and anus. The symptoms relate to the organ affected and can include obstruction (leading to difficulty swallowing or defecating), abnormal bleeding, or other associated problems. Among gastrointestinal cancer, stomach and colorectal cancer are the most common cancer in South Korea [1] and the second cause of cancer mortality worldwide [1]. The diagnosis often requires endoscopy, followed by biopsy of suspicious tissue. The treatment depends on the location of the tumor, as well as the type of cancer cell and whether it has invaded other tissues or spread elsewhere in the body [2]. In the middle of treatment, several nonoperative treatment modalities, such as chemotherapy and radiotherapy, are tried, but frequently survival is not prolonged, and side effects are severe [3]. As side effects of chemotherapeutic agents without survival advantage are not trivial, development of new agents exerting anticancer activity through novel mechanisms of action is needed to reduce side effects and increase efficacy [3].
Multiple organ failure caused by cancer progression is the major cause of death in cancer patients. Cancer progression is a multistep process and involves a series of processes including deregulation of cell growth and resistance to apoptosis, cellular adhesion and invasion through the basement membrane, transfer via circulation, extravasation, and proliferation at distant sites [4,5]. Therefore, prevention of tumor progression is one of the primary goals of cancer prevention and control. However, agents that are currently being used for cancer therapy are generally cytotoxic, with serious side effects that can diminish the quality of life of cancer patients [6]. Recently, many efforts have been made to find non- or low-cytotoxic bioactive compounds that are present in the diet and that have potential anticancer effects [7]. One focus is on compounds found in medicinal plants, which are able to inhibit one or more steps of cancer cell progression [8- 10]. For example, the intestinal metabolite of ginseng saponin, compound K (CK), has various chemopreventive and chemotherapeutic activities, including anti-tumor activity by targeting the NF-
The roots of
In the present study, the effect of SR on the growth of human gastric (MKN-45 cell line) and colorectal adenocarcinoma cells (WIDR cell line) and the combined effects of doxorubicin (DOX) and SR were examined. A cell cycle analysis examined the mechanisms of the inhibitory effects of SR on the proliferation of MKN-45 and WIDR cells.
2.1. Preparation of the extract from sophorae radix
Dried root of
Human gastric (MKN-45) and colorectal (WIDR) adenocarcinoma cell lines were used. All cell lines were established at the Cancer Research Center, College of Medicine, Seoul National University, Korea. All cell lines were propagated in RPMI-1640 medium (Gibco-BRL) supplemented with 10% heat-inactivated fetal bovine serum and 20 μg/ml of penicillin and streptomycin in an atmosphere of 5% CO2 at 37℃.
2.3. MTT (3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl- tetrazolium bromide) assay
Cell viability was assessed by using a MTT (3-[4, 5- dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide) assay. The MKN-45 and WIDR cells were seeded into each well of 12- well culture plates and then cultured in RPMI-1640 supplemented with other reagents for 72 hrs. After incubation, 100 μL of MTT solution (5 mg/mL in PBS) was added to each well, and the plates were then incubated for 4 hrs at 37℃. After removing the supernatant and shaking with 200 μLof dimethyl sulfoxide (Jersey Lab Supply, Livingston, NJ, USA) for 30 mins, we measured the absorbance at 570 nm. All experiments were repeated at least three times.
Caspase-3 assay kits (Cellular Activity Assay Kit Plus) were purchased from BioMol (Plymouth, PA, USA). After experimental treatment, cells were centrifuged (1,000 g, 4℃, 10 minutes) and washed with PBS (phosphate buffered saline). Cells were resuspended in ice-cold cell lysis buffer and incubated on ice for 10 mins. Samples were centrifuged at 1,000 g (4℃, 10 mins), and the supernatant was removed. Supernatant samples (10 μL) were incubated with 50 μL of substrate (400 μ M Ac-DEVD-pNA) in 40 μL of assay buffer at 37℃. The absorbance at 405 nm was read at several time-points. The pNA concentration in samples was extrapolated from a standard created with absorbances of sequential pNA concentrations.
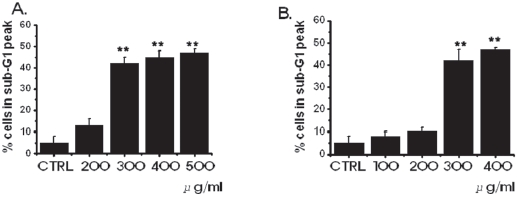
In order to investigate whether the cell cycles of the MKN-45 and the WIDR cells were redistributed, we used a flow cytometric analysis with propidium iodine (PI) stain [25,26]. Cells, 1 × 106, were placed in an e-tube. An ice-cold fixation buffer (ethyl alcohol), 700 μL, was slowly added with vortexing. Tubes were sealed with parafilm and incubated at 4℃ overnight. Samples were spun for 3 mins at 106 g at 4℃, and the supernatant was aspirated and discarded. The cell pellet was resuspended by using 200 μL of PI staining solution (PI [5 mg/mL], 2 μL, and RNase, 2 μL, in PBS, 196 μL) at 20,817 g for 5 secs. After 30 mins in the dark at room temperature, sam-ples were analyzed in a fluorescence-activated cell sorter (FACScan; Becton-Dickinson, Mountain View, CA, USA) at λ= 488 nm by using Cell-Quest software (Becton-Dickinson). The DNA content distribution of normal growing cells was characterized by using two peaks, those of the G1/G0 and the G2/M phases. The G1/G0 phase comprises the normal functioning and resting state of the cell cycle with the most diploid DNA content while the DNA content in the G2/M phase is more than diploid. Cells in the sub- G1 phase have the least DNA content in the cell cycle distribution; this is termed hypodiploid. The hypoploid DNA contents represent DNA fragmentation [26].
Data are expressed as mean ± SEM (Standard Error of the Mean). Differences between the data were evaluated by using the Student’s
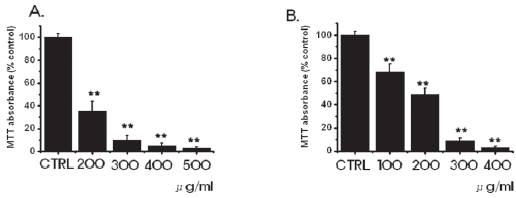
3.1. Inhibition of cell survival by SR
We investigated whether SR influenced the survival of MKN-45 and WIDR cells. SR inhibited the growth of MKN-45 and the WIDR cells (Fig. 1). As the concentrations were increased, the growth of the MKN-45 and WIDR cells was increasingly retarded in a dose-dependent manner. In the case of MKN-45 cells, following treatment with a 200 μg/ml concentration of SR for 3 days, survival of MKN-45 cells was reduced by 35.1 ± 9%, compared with controls. Treatment with 400-and 500-μg/ml concentrations of SR exhibited considerable antiproliferative effects on the MKN-45 cells. In case of WIDR cells, following treatment with a 100 μg/ml concentration of SR for 3 days, survival of WIDR cells was reduced by 68.2 ± 7%, compared with controls. Treatment with 300-and 400-μg/ml concentrations of SR exhibited considerable antiproliferative effects on the WIDR cells.
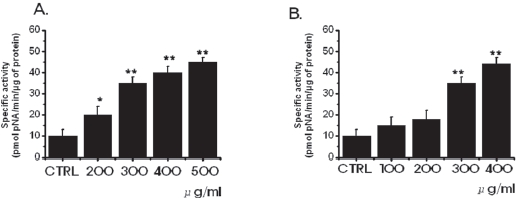
3.2. Inhibition of cell survival by SR leads to increased apoptosis
in MKN-45 and WIDR cells to be 10.3 ± 1.7 nmol pNA/min/μg protein and 11.2 ± 0.9 nmol pNA/min/μg protein, respectively. In MKN-45 cells, after 72 hours of being treated with 500 μg/ml of SR, elevated caspase- 3 activity was 45.3 ± 2.1 nmol pNA/min/μg protein (P < 0.01; Fig. 3A). In WIDR cells, after 72 hrs of being treated with 400 μ g/ml of SR, elevated caspase-3 activity was 44.2 ± 3.2 nmol pNA/min/μg protein (
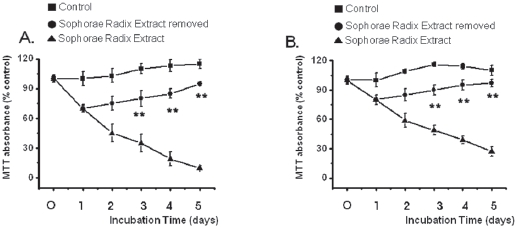
3.3. Reversibility of inhibition
The growth of the SR-treated MKN-45 and WIDR cells remained inhibited with exposure to SR for 1, 2, 3, 4, or 5 days (Fig. 4). After 4 days of incubation, the MKN-45 cells in the SRremoved group exhibited about 15% growth inhibition whereas about 81% inhibition was observed for continuous treatment with SR. After 4 days of incubation, the WIDR cells in the SRremoved group exhibited about 5% growth inhibition whereas about 61% inhibition was observed for continuous treatment with SR. Also, survival of MKN-45 and WIDR cells in the SRremoved group was increased to about 95% and 97% compared with about 115% and 110%, respectively, for the control group at 5 days.
3.4. Synergistic effects of SR and DOX
Combined treatment with SR and DOX significantly increased the growth inhibition of MKN-45 and WIDR cells compared with SR or DOX alone (P < 0.05, Tables 1 and 2). After 3 days of treatment, the inhibition rates for MKN-45 cell proliferation for 100 μg/ml of SR and 500 nM of DOX were about 42% and 32%, respectively. However, combined treatment with 100 μg/ml of SR and 500 nM of DOX exhibited about 87% inhibition against MKN-45 cells. As the concentration of SR combined with DOX was increased, the growth of the MKN-45 cells was retarded in a dose-dependent manner. In particular, 100 μg/ml of SR, combined with 1 μM of DOX, markedly inhibited the growth of MKN-45 cells (Table 1). After 3 days of treatment, the inhibition rates for WIDR cell proliferation for 100 μg/ml of SR and 500 nM of DOX were about 31% and 5%, respectively. However, combined treatment with 100 μg/ml of SR and 500 nM of DOX exhibited about 57% inhibition against WIDR cells. As the concentration of SR combined with DOX was increased, the growth of the WIDR cells was retarded in a dose-dependent manner. In particular, 100 μg/ml of SR, combined with 1 μM of DOX, markedly inhibited the growth of WIDR cells (Table 2).

Effects of Soph orae Radix extract (SR) and/or Doxorubicin (DOX) at varied concentrations on growth of MKN-45 human gastric cancer cell after 3 days of treatment
In this study, SR significantly inhibited the growth of MKN-45 and WIDR cells compared with controls (P < 0 .01). The inhibitory effects of SR on the growth of MKN-45 and WIDR cells were first observed at a 200-μg/ml and a 100-μg/ml concentration, respectively and this inhibitory effect was dose-dependent up to 400 μg/ml. These data suggest that SR has a significant antiproliferative effect in MKN-45 and WIDR cells, as has been observed in other cancer cells [30-33]. SR is the dried roots of
In the present study, inhibition was lost when SR was removed by media change after 24 hrs of SR treatment and was not different from the control group thereafter, suggesting reversibility of the effect of SR against MKN-45 and WIDR cells. The reversibility of growth inhibition suggests that SR is not a nonspecific cytotoxin but has a specific mechanism for inhibiting proliferation of MKN-45 and WIDR cells.
Apoptotic cell death pathways are induced by a variety of signals. One mechanism which is consistently implicated in apoptosis is the activation of a series of cytosolic proteases, the caspases [34]. Caspases are synthesized as inactive proenzymes that are processed in cells undergoing apoptosis by self-proteolysis and/or cleavage by another protein. In MKN-45 and WIDR cells, after 72 hrs of being treated with SR, caspase-3 activity is increased (Fig. 3).
In conclusion, SR significantly inhibited the growth of MKN-45 and WIDR cells in a dose-dependent manner. The inhibitory effect of SR on the growth of MKN-45 and WIDR cells was reversible, for the growth of MKN-45 and WIDR cells after removal of SR by media change after 24 hrs of treatment was not different from that of the control group. The reversibility of growth inhibition suggests that SR is not a nonspecfic cytotoxin for MKN-45 and WIDR cells. This is the first report that SR may be very useful for the treatment of gastric and colorectal adenocarcinomas, especially in conjunction with DOX. More studies employing animal experiments and clinical trials are needed to establish the efficacy of SR as an anticancer drug for treating human gastric and colorectal adenocarcinomas.