Aquacultured fish with gross abnormalities can be problematic because they can affect marketability(e.g., Afonso et al. 2000; Michie 2001; Verhaegen et al. 2007; Castro et al. 2008). Consequently it is important to describe deformities, determine their frequencies and determine their causes.
Abnormal fish occur in all populations. Dawson (1964, 1966, 1971) and Dawson and Heal (1976) cataloged dozens of abnormalities in several hundred species of fish in a 1,499-reference review. A detailed examination of any population will reveal deformed individuals. For example, Afonso et al. (2000) found 39 deformities in a hatchery population of 11,640 gilthead sea bream Sparus aurata and Verhaegen et al. (2007) found that 80% of intensively raised stocks of gilthead seabream had deformed individuals. Sullivan et al. (2007) surveyed six Atlantic salmon Salmo salar farms and found that 3.8-8.8% of the fish had the “stubby body” deformity.
Surveys of tilapia populations have also revealed this problem. Guilherme (1992) found 2,621Oreochromis niloticus that had one of three fin deformities in 5,456 specimen fish. Eissa et al. (2009) surveyed O. niloticus at two Egyptian farms and found that 2.7% and 1.6% of the fish at these facilities were deformed. Phelps and Karsina (Phelps RP and Karsina E, Auburn University, AL, pers. comm.) examined Auburn University-Ivory Coast strain (AU-I) O. niloticus and found 23 deformed fingerlings that had one of four deformities in a sample of 7,200 fish; they also found that 2 of 234 brood fish were deformed (a single deformity).
The occurrence of deformed fish at fish farms should not be surprising, because a major goal of fish culture is to maximize survival (Tave 1993). Eggs and fry are often artificially incubated, which enable deformed ones to survive. Ponds are fertilized and fish are fed; deformed fish that might have trouble finding food in the wild have easy access to food.Finally, efforts are taken to exclude or kill predators; abnormal individuals that would be vulnerable to predation in the wild can survive at a fish farm.
It is often assumed that all abnormalities have a genetic basis, but most are non-heritable (Tave, 1993). Non-heritable abnormalities can be caused by disease (e.g., Hoffman et al., 1962; Hoffman, 1984; Pasnik et al. 2007); injury (e.g., Breeder, 1953; Gunter and Ward, 1961); environmental disturbances (e.g., Rogers 1956; Garside, 1959; Yamamoto et al. 1963; Schroder, 1969; Mayer et al., 1978; Couch et al., 1979; Backiel et al., 1984; Grady et al., 1992; Wargelius et al., 2005; Sun et al., 2009); nutritional deficiencies (e.g., Rucker at al., 1970; Andrews and Murai, 1975; Lim and Lovell, 1978; Murai and Andrews, 1978), and culture conditions (e.g., Romanov,1984; Wiegand et al., 1989; Leary et al., 1991; Krise and Smith, 1993; Sawada et al., 2006). They can also be the result of developmental errors (e.g., Tave et al.,1982; Dunham et al., 1991; Handwerker and Tave, 1994; Tave and Handwerker, 1994).
When abnormalities are produced genetically, they are usually produced by recessive alleles (e.g., Aida, 1930; Matsui, 1934a; Rosenthal and Rosenthal, 1950; Schultz, 1963; Thomerson, 1966; Tukeuchi, 1966; Mair, 1992). One reason why genetically-produced abnormalities occur at hatcheries is that most hatchery populations have small effective breeding numbers (Tave, 1993, 1999). Inbreeding is inversely related to effective breeding number, and one aspect of inbreeding depression is an increase in the production of abnormal fish--these abnormalities are produced by recessive alleles.
Inbreeding studies in fish have produced abnormalities in rainbow trout Oncorhynchus mykiss (Aulstad and Kittelson, 1971; Kincaid, 1976), zebra fish Danio rerio (Piron, 1978; Mrakov?i? and Haley,1979; and convict cichlid Cichlasoma nigrofasciatum (Winemiller and Taylor, 1982). For example, one generation of brother-sister matings (inbreeding=25%), increased the percentage of deformed rainbow trout by 3%, and two generations (inbreeding=37%) increased it by 10% (Kincaid,1976).
Even though most heritable abnormalities are produced by recessive alleles, they can also be produced by dominant alleles (Matsui, 1934b; Lodi, 1978; Tave et al., 1983).
Deformed fish can be produced when species are hybridized (e.g., Ihssen, 1978; Chappell, 1979; Beck and Biggers, 1983; Blanc and Poisson, 1983; Blanc and Chevassus, 1986; Kurokura et al., 1986; McKay et al., 1992). In some cases the incidence is so high that it makes the hybrid unsuitable for commercial development. For example, Chappell (1979) found that the hybrids between white catfish, Ameiurus catus and both channel catfish Ictalurus punctatus and blue catfish I. furcatus were unsuitable for commercial purposes because many were deformed. These abnormal hybrids were produced because white catfish have 48 chromosomes, while channel catfish and blue catfish have 58 chromosomes (LeGrande et al. 1984).
Deformed fish can be produced during breeding programs that involve chromosomal manipulation (e.g., Chourrout, 1980 1982; Thorgaard et al., 1981; Cassani and Caton, 1986; Chao et al., 1986; Chen et al., 1986; Myers, 1986; Cherfas et al., 1990, 1993; Myers and Hershberger, 1991; Gray et al., 1993; Yamazaki and Goodier, 1993). These deformities can be either genetic or envronmental.
Because all hatchery populations contain deformed individuals, farmers and hatchery managers should routinely monitor their fish and catalog abnormalities (written description and photograph) and calculate the frequencies. This is critical, because it is the only way to determine if certain abnormalities recur annually and if the frequencies are increasing.
If the frequency of an abnormality increases, the effects that it has on production traits and marketability should be studied. Some deformities have been shown to adversely affect growth, survival, hatchability, disease resistance, or other traits (Tave et al. 1983; Koumoundouros et al., 1997). On the other hand, some can improve market value. For example, bizarre phenotypes such as telescope eyes in goldfish Carassius auratus are desired by tropical fish fanciers, and common carp Cyprinus carpio, with abnormal scale patterns command higher market prices in some locales.
This paper describes 31 gross deformities that have been observed in tilapia and provides some information on their frequencies and causes. Twentyone were described in published reports; the others were obtained from a survey.
This abnormality was discovered in the Auburn University population of O. aureus (Tave et al., 1983). Bondari (1984) observed it in a population of O.aureus at the University of Georgia, Tifton, GA; that population was founded by fish that came from Auburn University. It was observed in O. aureus in the St. John River, Florida (Zale A, University of Florida, Gainesville, FL, USA, pers. comm.); he estimated that the frequency was 0.05%.
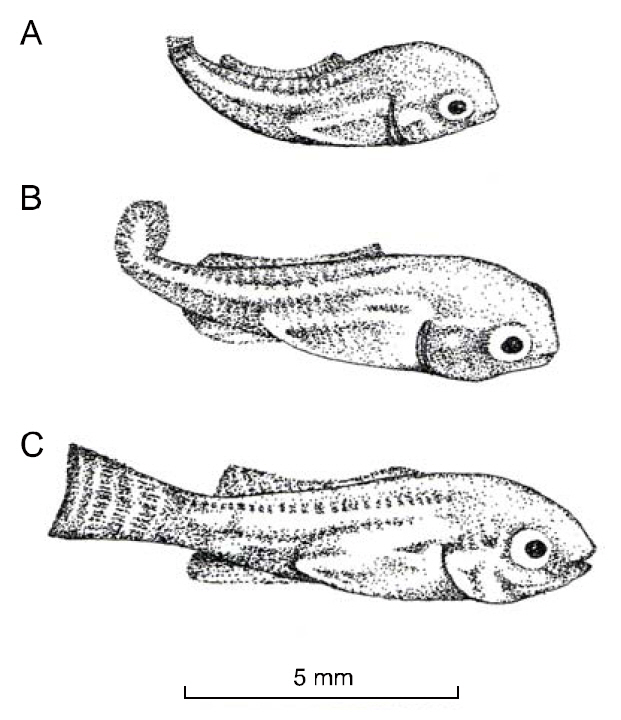
Saddlebacks lack parts of or all of the dorsal fin.Fin ray loss was variable; some individuals were missing only the first 1 or 2 hard spines, while others had no dorsal fin (Fig. 1). In some saddlebacks, the pectoral, pelvis and/or anal fins were either deformed or missing. Vertebrae 1-3 were always deformed, and this resulted in curvature of the spine and affected body length. Missing spines and rays in the dorsal fin were accompanied by missing pterygiophores; this produced the depression of the dorsal margin of the
body. Deformed or missing paired fins were accompanied by abnormal or missing bones in the pectoral and/or pelvic girdles.
Saddleback is produced by an autosomal dominant lethal allele that exhibits incomplete dominance.Homozygous dominant individuals (SS) are aborted; heterozygotes (S+) are saddlebacks; homozygous recessive (++) individuals are normal.
The S+ genotype (the genotype that produces saddleback) had adverse pleiotropic effects on viability and disease resistance: 67% of saddlebacks died during the first 2-3 months. When challenged by Saprolegnia sp., 33% of saddlebacks died, while only 5% of normal fish died.
Because this abnormality is produced by a dominant allele, a single generation of selection where all saddlebacks are culled will eliminate the deformity.
Abella (Abella TO, University College of Swansea,Wales, UK, pers. comm.) observed a similar phenotype in O. niloticus. In a sample of 1,000 fish, 10 had no dorsal fin.
CDS was discovered in the Lake Manzala strain of O. niloticus maintained at University College of Swansea, Wales (Mair 1992). The deformity was characterized at the first feeding stage by the reduction of the caudal fin; additionally, there was an upward curvature of the caudal region of the spine (Fig. 2). Fish with CDS also had deformed opercula; they were not fully developed, and some gill lamellae were exposed. Fish with this deformity were unable to swim efficiently. No fish with CDS lived more than 7 days after yolk sac absorption.
Breeding experiments revealed that this deformity is produced by an autosomal recessive allele; CDS is produced by the recessive allele: ++ and +m fish are normal, while mm fish have CDS.
If this phenotype exists in a population of O.niloticus, it can adversely affect fingerling production, since 25% of the offspring produced by the mating of two heterozygotes will die. Because this deformity is produced by a recessive allele, selection cannot eliminate the allele. Progeny testing must be used to eliminate the allele.
This deformity was observed in O. niloticus during a sex reversal study; 1,114 of 5,416 had this deformity. Survival of the fish with no dorsal fish was similar to that of normal fish (Guilherme 1992). Pasnik et al. (2007) found that Steptococcus agalactinae could produce this deformity in O.niloticus.
This deformity was observed in O. niloticus during a sex reversal study (Fig. 3). A subsequent breeding study revealed that the deformity was heritable(Guilherme, 1994).
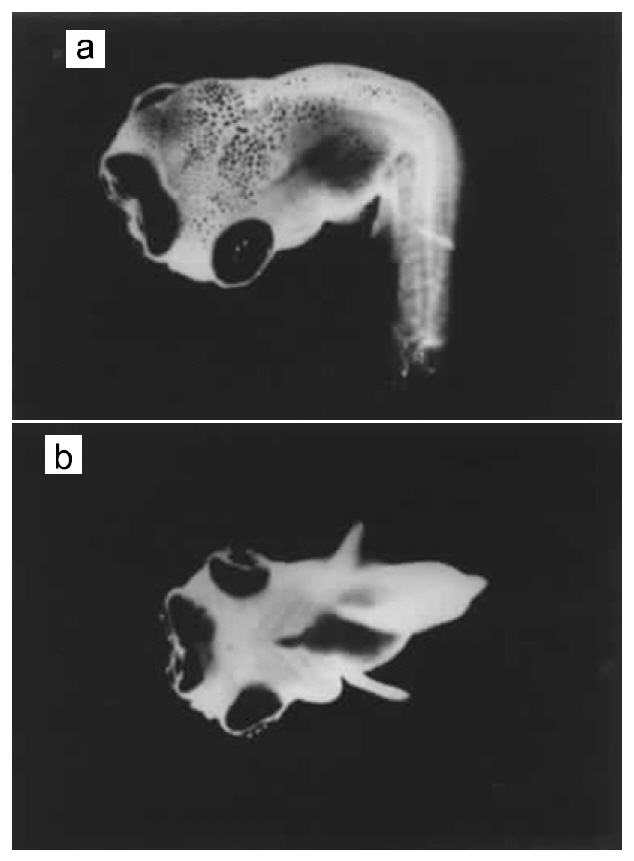
This phenotype has been observed in several stocks of O. niloticus. It was first described in AU-I O.niloticus (Tave et al., 1982). Tailless individuals were missing the caudal peduncle and the caudal fin (Fig.4b). Tailless fish had only 26 vertebrae, 3 fewer than normal fish. Vertebrae 20-26 were deformed, causing curvature of the spine; this demonstrated that the deformity was not due to injury. Only two tailless individuals were found, and they died before they could be spawned. Phelps and Karsina (pers. comm.) surveyed a population of 7,200 AU-I O. niloticus fingerlings, and observed one tailless individual.
A tailless individual was observed in a population in Saudi Arabia (George, 2006; George TT, pers. comm., Global Aquaculture Consultants, Toronto, ON, Canada).
A tailless individual was observed in the population maintained at the Pukyong National University, Busan, Korea (Jo and Kim, unpublished data.). Abella (Abella TO, pers. comm.) found 10 tailless fish in a survey of 1,000 O. niloticus at the University College of Swansea, Wales.
Pasnik et al. (2007) found that S. agalactinae could produce this deformity in O. niloticus.
Guilherme (1992) found 863 O. niloticus with no anal fin in a population of 5,456 fish during a sex reversal study. Survival of the deformed fish was 3.4%.
Phelps and Karsina (pers. comm.) surveyed a population of 234 AU-I O. niloticus brood fish and observed two that had no anal fin. Tave et al. (1983) found one O. aureus saddleback with no anal fin, but that individual had no fins except for the caudal fin(Fig. 1n).
Pasnik et al. (2007) found that S. agalactinae could produce this deformity in O. niloticus.
Guilherme (1992) found 644 O. niloticus with this deformity in a population of 5,456 fish during a sex reversal study. Survival of the deformed fish was 46.9%. Tave et al. (1983) found some saddleback O.aureus that were missing pelvic fins.
Pasnik et al. (2007) found that S. agalactinae could produce this deformity in O. niloticus.
Eissa et al. (2009) found this deformity in O.niloticus in Egypt. The deformity constricted and twisted the body (Fig. 5).
This deformity was observed in the Auburn University population of O. mossambicus (Tave, unpublished data). Swaybacks were missing the middle portion of the dorsal fin. Twenty-four individuals were observed when fish were 2-4 cm. All died within a week of discovery.
Oreochromis spp. with split fins were observed in polluted rivers in Taiwan; 1-87.5% of the fish observed had this deformity (Sun et al. 2009).
Stumpbody was discovered in the Auburn University population of O. aureus (Tave et al., 1982). Stumpbodies were dwarfs, whose body was shortened along the anterior-posterior axis (Fig. 4c and 4d). The posterior portion of the trunk appeared to be compressed. The trunk was much thicker than that of normal fish, and the relative body depth was also greater.
Stumpbodies had gross skeletal deformities. They had 15 vertebrae, which was approximately half the
number found in normal fish (29). Many vertebrae were grossly deformed, which resulted in marked curvature of the spine. Vertebral deformities varied considerably among stumpbodies; no two individuals were identical. Breeding experiments revealed that it was not heritable.
Eissa et al. (2009) observed this phenotype in farmed populations of O. niloticus in Egypt. Eissa (Eissa AE, Faculty of Veterinary Medicine, Cairo University, Giza, Egypt pers. comm.) was able to produce stumpbodies by experimental infection with Aspergillus ochraceus.
A similar phenotype was observed in Lake Manzala strain O. niloticus at University College of Swansea, Wales, (Mair GC, Central Luzon State University, Nueva Ecija, Philippines, pers. comm.) and in a strain of “Florida red tilapia,” (a strain derived from O. urolepis hornorum ♀ × O.mossambica ♂ matings) (Watanabe WO, Caribbean Marine Research Center, Vero Beach, FL, USA, pers. comm.); he estimated that the frequency of the deformity in the hybrid population was 0.014-0.02%.
Phelps and Karsina (pers. comm.) found 19 runts in a survey of 7,200 AU-I O. niloticus fingerlings. These fish were one-half to one-quarter the size of their sibs and did not grow well. It is not known if this was a morphological deformity or a physiological defect, such as a malfunctioning pituitary.
Ra’anan and Lahav (Ra’anan Z and Lahav E,Aquaculture Production Technologies, Ltd., Jerusalem, Israel, pers. comm.) found runts in the ND5 female line of O. niloticus. They described the fish as exceptionally short and flat, and the deformity appears at a constant frequency of 2%.
This deformity was produced in F2 O.mossambicus × O. niloticus hybrids at the University of Wales, Swansea (Shah 1984). Sperm from F1 hybrid males had been treated with 0.005-M of methyl methane sulphonate (a mutagen). A single fish with a flattened body was produced by this treatment.
This deformity was produced in O. niloticus from Stirling University, Stirling Scotland (Shah 1984). It was produced from matings where males had been injected with 50 mg methyl methane sulphonate/kg body weight IP prior to spawning. Individuals with this deformity has marked curvature of the body; the caudal area and tail was bent laterally at a right angle.
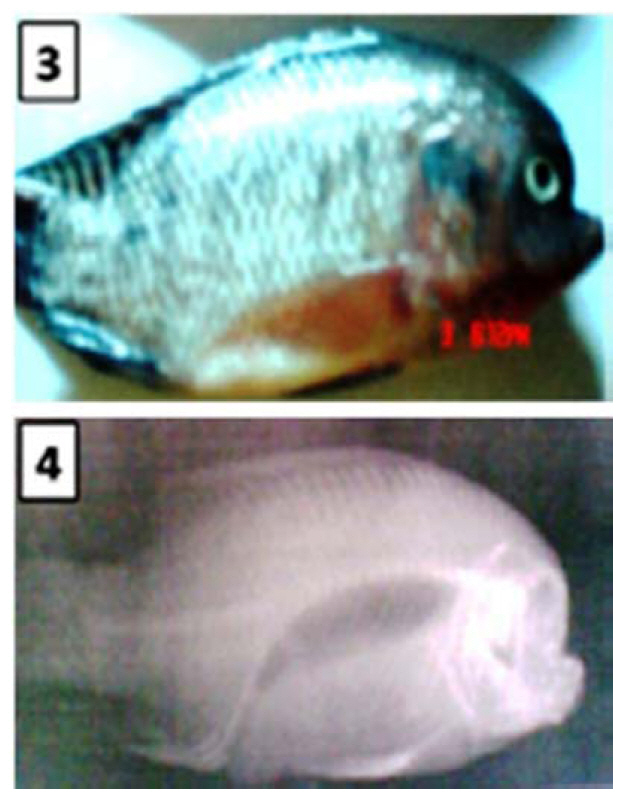
Operculum Deformities Deformed opercula, described as “semi-operculum,” were observed in both Auburn University-Egypt strain O. niloticus and Auburn University population of O. mossambicus (Handwerker and Tave, 1994; Tave and Handwerker, 1994). Fish with this deformity had a shortened operculum that exposed part of the gills (Fig. 6). All fish had one normal and one deformed operculum. The deformity was variable,in that some fish with a semi-operculum had a deformed operculum that was minimally shortened, while others had an operculum that so deformed that most of the gill lamellae were exposed. Breeding studies with both species revealed than the deformity was not genetic.
Phelps and Karsina (pers. comm.) found 2 fish with this deformity in their survey of 7,200 AU-I O.niloticus.
An incompletely developed operculum in tilapia can be produced by environmental contaminants and by infection. Clemens and Inslee (1968) observed fish with what they described as a “poorly formed opercle” in O. mossambicus that had consumed methyl testosterone-treated feed during a sex-reversal study; the deformity was not observed in the control.Pasnik et al. (2007) found that S. agalactiae could produce this deformity in O. niloticus. Sun et al.(1998, 2009) found that this deformity in O. niloticus and O. mossambicus in rivers in Taiwan. Sun et al. (2009) found that it could be produced in O. niloticus by low levels of dissolved oxygen, high levels of ammonia, and by heavy metals.
Anopthalmia (the congenital absence of one or both eyes) was observed in the Auburn University population of O. mossambicus (Tave and Handwerker,1998). Seven anopthalmics were observed; five were missing the left eye, and two were missing the right eye. The area over the missing eye was covered with scales and the skull was slightly depressed. When viewed dorsally, the skull appeared to be slightly curved (Fig. 7). A breeding study revealed that the deformity was not heritable.
In a survey of 1,000 fish in the O. niloticus population at the University College of Swansea, Abella (Abella TO, pers. comm.) observed 3 anopthalmics.
A single four-eyed fish (Fig. 8) was observed in O.niloticus at the Tennessee Valley Authority Hatchery, Muscle Shoals, AL (Jo, unpublished data). The fish died shortly after it was observed.
This deformity was observed in a O. niloticus population at the University College of Swansea, (Abella TO, pers. comm.); the position of the eyes was described as similar to that of most flounders.
This deformity was produced in O. niloticus from Stirling University (Shah, 1984). It was produced from matings where males had been injected with 50 mg methyl methane sulphonate/kg body weight IP prior to spawning. Eyes in fish with this deformity were significantly smaller than those of normal fish.
This deformity was observed in O. aureus maintained at the University of Wales (Mair GC, pers. comm.). The anterior-most portion of the head in these fish was flat and the eyes were fused at the
flattened front. These fish also had the deformity called “protruding jaw.” Three fish with fused eyes were observed. A breeding experiment revealed that the deformity was not genetic.
This deformity was produced in F2 O.mossambicus × O. niloticus hybrids at the University of Wales (Shah, 1984). Sperm from F1 hybrid males had been treated with 0.05 M of methyl methane sulphonate. Three bullheads were produced. The lower jaw of these fish was much larger than that of normal fish. A breeding study showed that the deformity was not genetic.
This deformity was produced in F2 O. niloticus × O.mossambicus hybrids at the University of Wales(Shah, 1984). Sperm from F1 hybrid males were treated with 0.05 M of methyl methane sulphonate. A single fish with upturned mouth was produced. The mouth in the deformed individual terminated in an upward direction.
Clemens and Inslee (1968) reported that some O.mossambicus that had consumed methyl testosteronetreated feed during a sex-reversal breeding program had a deformed lower jaw, but the deformity was not described. No fish in the control population had this deformity.
This deformity was observed in O. aureus maintained at the University of Wales (Mair GC, pers. comm.). Fish with this deformity had a jaw that protruded in a tube-like fashion, and the mouth was at the end of the jaw. These fish also had the fused eye deformity. Three fish with protruding jaw were observed. A breeding experiment revealed that the deformity was not genetic.
A similar deformity named “lower lip extension” was observed in O. niloticus found in polluted rivers in Taiwan (Sun et al., 1998, 2009).
This deformity was observed in farmed populations of O. niloticus in Egypt (Eissa et al. 2009). The deformity is characterized by a lateral bowing of the mandibles with outward thickening of the lateral sides of the mandibles (Eissa AE, pers. comm.)
Clemens and Inslee (1968) observed this deformity in O. mossambicus that had consumed methyl testosterone-treated feed during a sex reversal study; the deformity was not observed in the control population.
This deformity was observed in a farmed population of O. niloticus in Egypt (Eissa et al., 2009). The anterior portion of the head above the mouth is stunted, given the fish a protruding mouth (Fig. 9).
Microcephaly, an abnormal reduction in head size,was observed in O. niloticus from Stirling University (Shah, 1984). It was produced from matings where males had been injected with 50 mg methyl methane sulphonate/kg body weight IP prior to spawning.
Rothbard et al. (1980) described an O. urolepis
hornorum fry with a double yolk sac. The yolk sac was large and partially constricted in the middle.
This deformity was produced in O. niloticus from Stirling University (Shah, 1984). It was produced from matings where males had been injected with 50 mg methyl methane sulphonate/kg body weight prior to spawning. Individuals with this deformity had yolk sacs that were significantly larger than those possessed by normal individuals.
Hulata and Rothbard (1978) described two sets of conjoined twins that they found in O. mossambicus and one from an O. urolepis ♀ × O. niloticus ♂ hybrid mating. Rothbard et al. (1980) described one set of conjoined twins from an O. niloticus ♀ × O. aureus ♂ hybrid mating. Owusu-Frimpong and Hargreaves (2000) found conjoined O. aureus following heat shock of fertilized eggs. Four sets of conjoined twins were observed from a total of 100 spawns (70,000-100,000 eggs) that were produced by 14O. niloticus females at the Pukyong National University, Busan (Fig. 10) (Jo and Kim, unpublished data). Abella(Abella TO, pers. comm.) observed conjoined O.
niloticus. Abiado (Abiado MA, Central Luzon State University, Nueva Ecija, Philippines, pers. comm.) observed 4 sets of conjoined twins in a population of 2,500 O. niloticus × O. aureus hybrid swim-up fry.Mair (Mair G, pers. comm.) observed conjoined twins in both O. niloticus and O. aureus stocks maintained at the University of Wales. Tave (unpublished data)observed a set of AU-I O. niloticus conjoined twins.
All fish populations contain abnormal individuals. Thirty-one gross abnormalities in tilapia were described in this review. Fin abnormalities accounted for almost a third of those that were observed (10), followed by abnormalities of the eye (five), jaw (five), body shape (four), head (three), yolk sac (two), operculum (one), and conjoined twins. It is often assumed that abnormalities have a genetic basis and that they are a clinical sign of inbreeding depression,but this survey shows that only three of nine abnormalities that were subjected to genetic studies were heriatable, and one was caused by a dominant allele which means that it was not a side effect of inbreeding. Most abnormalities are non-heritable, and this survey shows that that abnormailites in tilapia can be produced by pathogens, by pollutants and other chemicals, by heat shock, and by sex reversal.Because abnormalities can decrease yields and profits, it is important to census a managed population to catalog the abnormalities. A yearly census will let managers know how many abnormalities are present and whether the percent is increasing over time. An increase in percent deformities or of a particular deformity can be an indication that a problem exits. If a deformity becomes problematic, a study should be conducted to determine the cause and possible solutions.