Recent climate changes have been affecting various aspects of the lives of many different plant and animal taxa (Walther et al. 2002, Parmesan 2006, Willis and Bhagwat 2009, Gilman et al. 2010). Should this trend continue,many species are expected to become extinct in the near future (Brook et al. 2008). Thus, many countries are monitoring various field populations and evaluating the direct and indirect effects of climate changes on these populations (Araujo et al. 2006, Whitfield et al. 2007, Laurance 2008, D’Amen and Bombi 2009). In particular, amphibians and reptiles are employed widely as model taxa for studies of how climate changes affect animal ecology,as the small, poikilothermal bodies of amphibians and reptiles render them quite sensitive to environmental changes (Wake 2007, Whitfield et al. 2007).
It is difficult to detect declines or changes in field populations or communities following environmental changes without instituting long-term monitoring of the populations and communities both prior to and after the environmental changes occur (Blaustein et al. 2003).Therefore, developed countries have been conducting long-term monitoring of various plant and animal taxa and have begun to use the data obtained from the monitoring to deal with recent climate changes (Gibbs and Breisch 2001, Reading 2007, Whitfield et al. 2007, Hartel 2008, McMenamin et al. 2008, Olson 2009).
In this study, we monitored the herpetofauna in Woraksan National Park from March 2005 to September 2009, and analyzed the data in an effort to determine how climate changes affected the amphibian and reptile communities in the park.
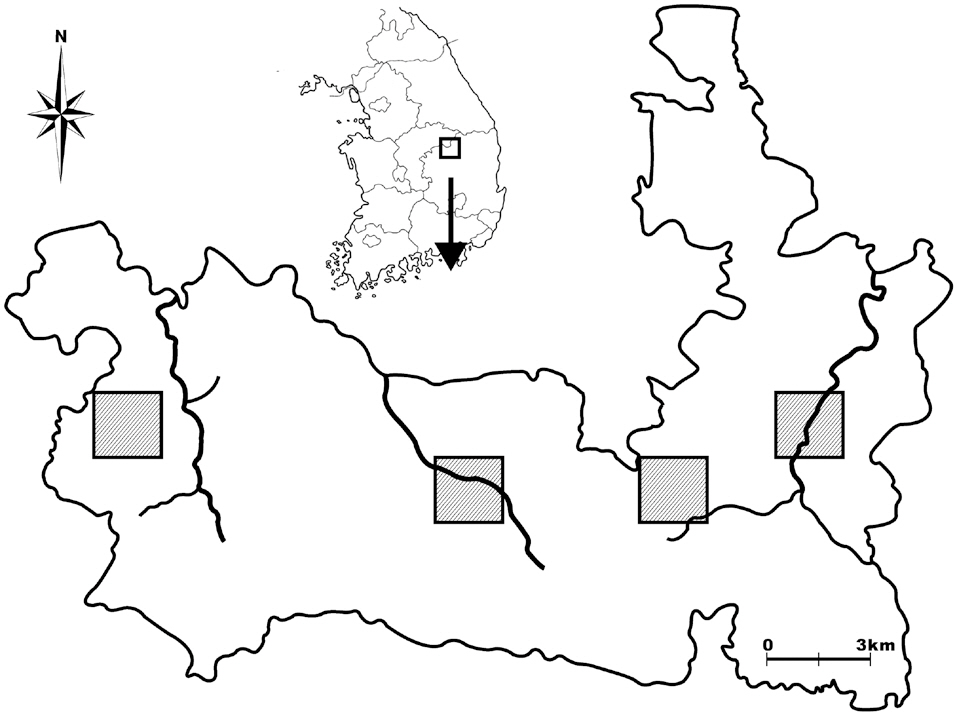
Woraksan National Park covers a total area of 288 km2, including parts of Jecheon-si, Moonkyeong-si, Danyang-gun, and Chungcheongbuk-do; the park contains more than five mountains, the altitudes of each over 1,000 m. There are three main mountain valleys in the site: the Songgae, Yongha, and Sunam valleys. To select amphibian and reptile survey sites, we first imposed 2 km × 2 km quadrates on a map of Woraksan national park, and from these quadrates, we selected two quadrates in the east side of the park, one quadrate in the middle, and one quadrate on the west side, taking into consideration the topographic characteristics of the sites, such as the existence of rocky areas and valleys (Fig. 1). We then selected one or two detailed survey sites in each quadrate by visiting the selected quadrate sites.
For the amphibian surveys, we selected both valley sites and mountain edge sites to represent both the center and the edge areas of amphibian communities. For the valley sites, we selected parts of mountain streams (100-300 m in length, 3-5 m in width) in the valleys for detailed surveys. For the mountain edge sites, we select-
ed small ponds, wetlands, or agricultural lands located at the edges of mountains. For the reptile surveys, we selected approximately 50 m × 50 m areas, which included dry lands, rocky sites, and abandoned crop fields. Of the nine reptile survey sites, six were in the amphibian survey quadrates, and three sites were near the quadrates, thus ostensibly increasing the likelihood of finding reptiles during surveys.
We conducted amphibian and reptile surveys once per month from March to September, from 2005 to 2009 (total 35 times). Between March and May, our surveys focused on amphibians, but between June and September, they focused on reptiles. For the amphibian surveys, we scrutinized entire survey sites, found and identified individuals based on the methods described by Kang and Yoon (1975), and recorded all the individuals detected. In the valley sites, two persons slowly walked along the stream line, finding and recording all individuals. Additionally, we counted and recorded the numbers of egg clumps of the Korean salamander (
We divided the raw data into annual data: the number of individuals of each amphibian and reptile species found in each year and the number of egg clumps of the Korean salamander, Dybowskii's brown frog, and Korean stream brown frog found in each year. Using these data, we first calculated the diversity, evenness, and richness of amphibian species at the valley sites, at the mountain edge sites, and with combined data from the two sites (Margalef 1958, Pielou 1966, McNaughton 1967). We also calculated the species diversity, evenness, and richness of reptile species, in a similar fashion. We assessed the dominance of both amphibians and reptiles.
Climate data were obtained from the Chungju metrological center (http://chungju.kma.go.kr), which is located approximately 20 km from the survey sites. From the archived climate data, we extracted climate factors between 2005 and 2009 including annual minimum temperature, annual maximum temperature, monthly mean temperature, monthly mean precipitation, monthly mean temperature during winter, and monthly mean precipitation during winter (from December to March). We used the winter temperature and precipitation data, because those would be the most relevant to spring-breeding amphibians and reptiles. We included the weather conditions in March because the conditions in the March could be parts of winter season in some areas in Korea.
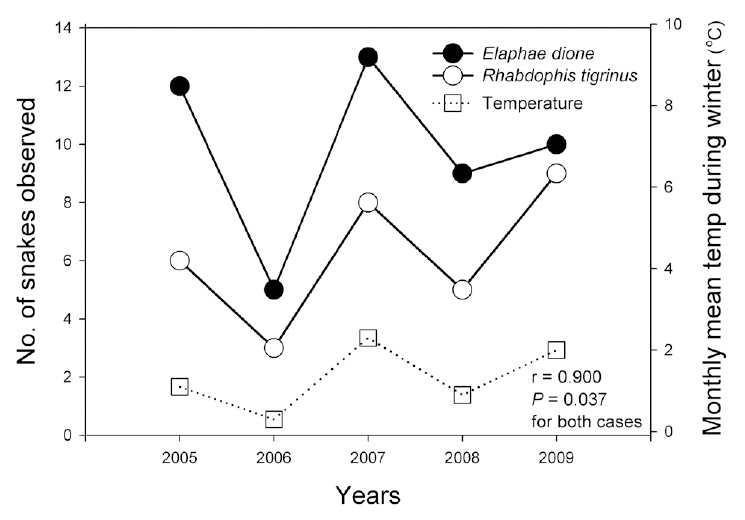
Because the year-based sample size was frequently as small as five, we used Spearman’s correlation test to analyze relationships among the six climate factors and the survey data, including amphibian species indices; the numbers of egg clumps of the Korean salamander, Dybowskii's brown frog, and Korean stream brown frog; reptile species indices; and the numbers of the Dione's ratsnake (
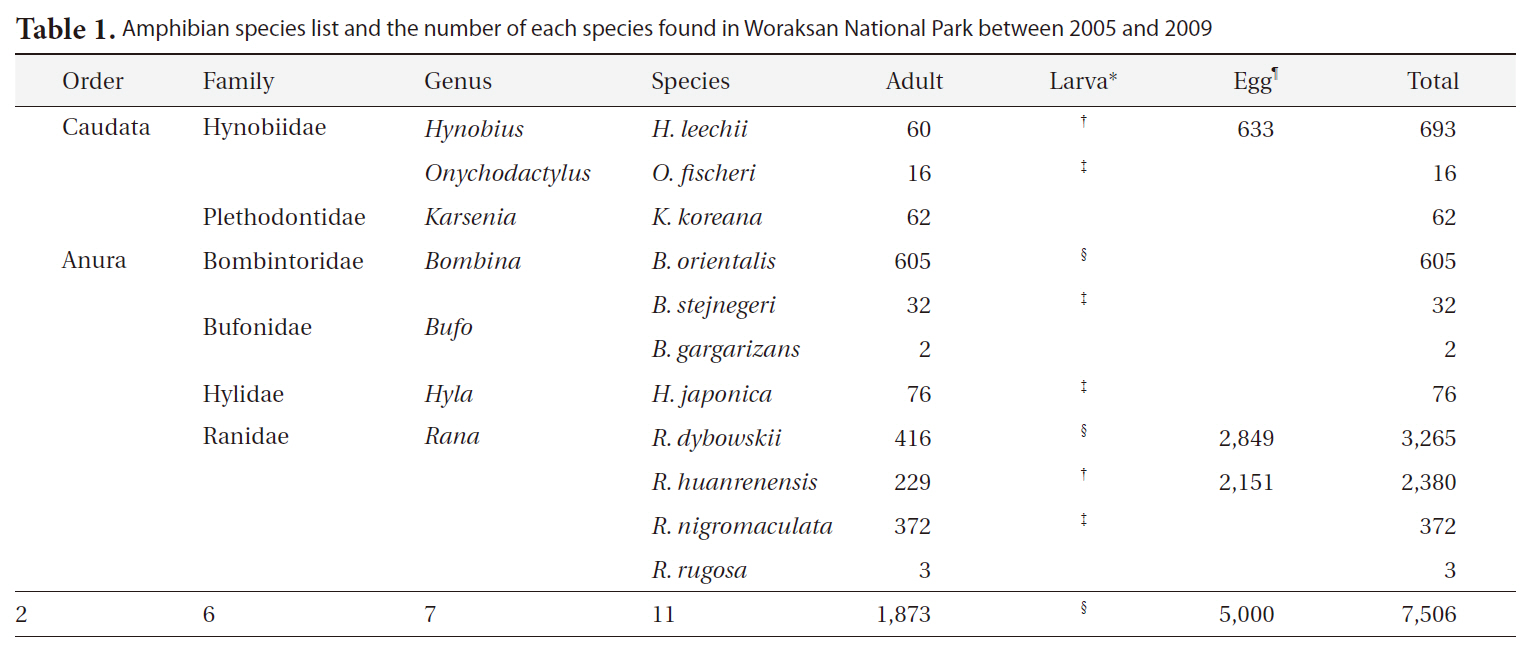
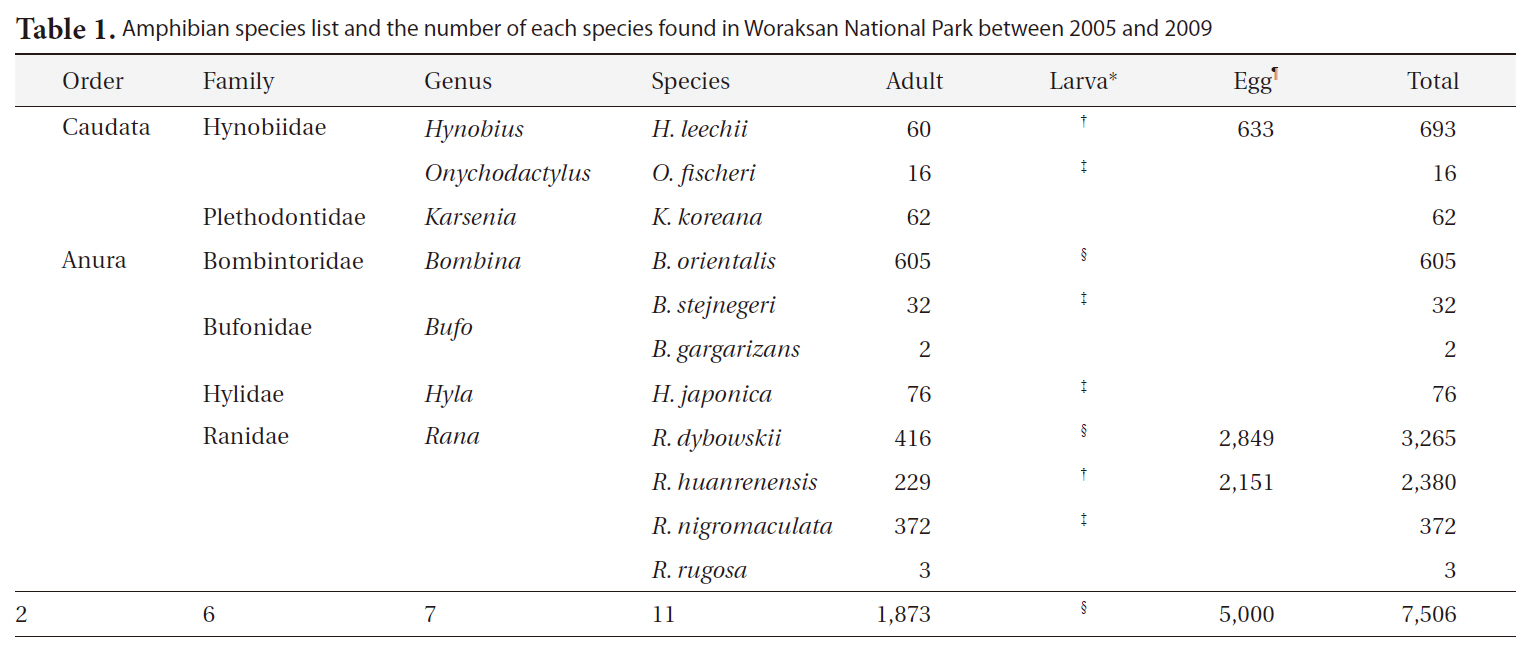
Amphibian species list and the number of each species found in Woraksan National Park between 2005 and 2009
During the monitoring, we detected a total of 11 amphibian species (1,873 individuals) in seven genera, six families, and two orders (Table 1). We found 5,000 egg clumps of the Korean salamander, Dybowskii's brown frog, and Korean stream brown frog. The most dominant species was the Korean fire-bellied toad (
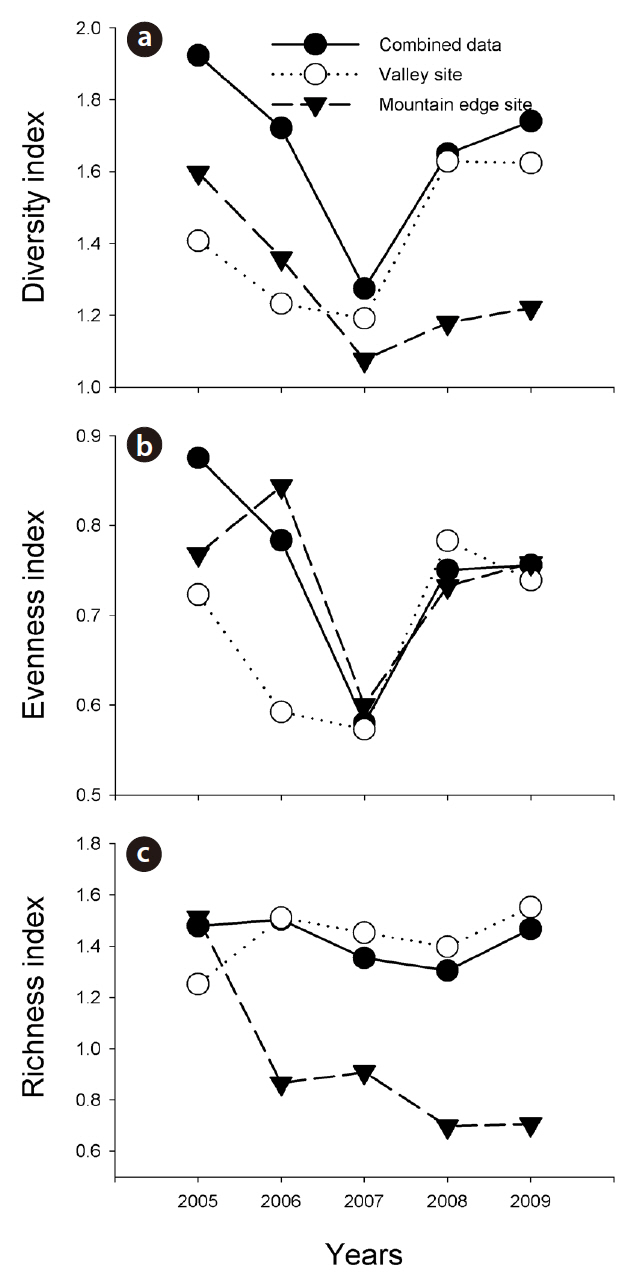
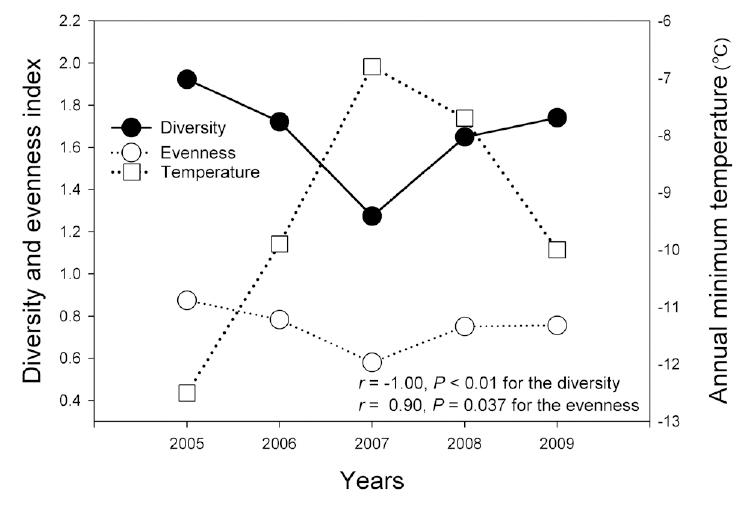
Combined amphibian species diversity and evenness was negatively correlated with the annual minimum temperature (
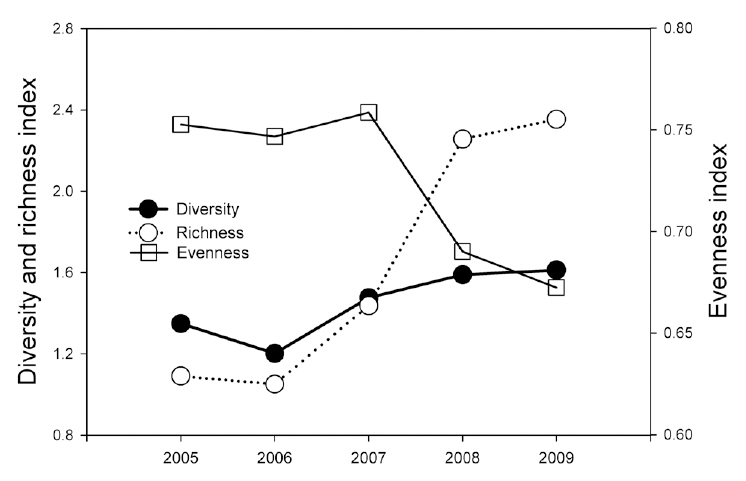
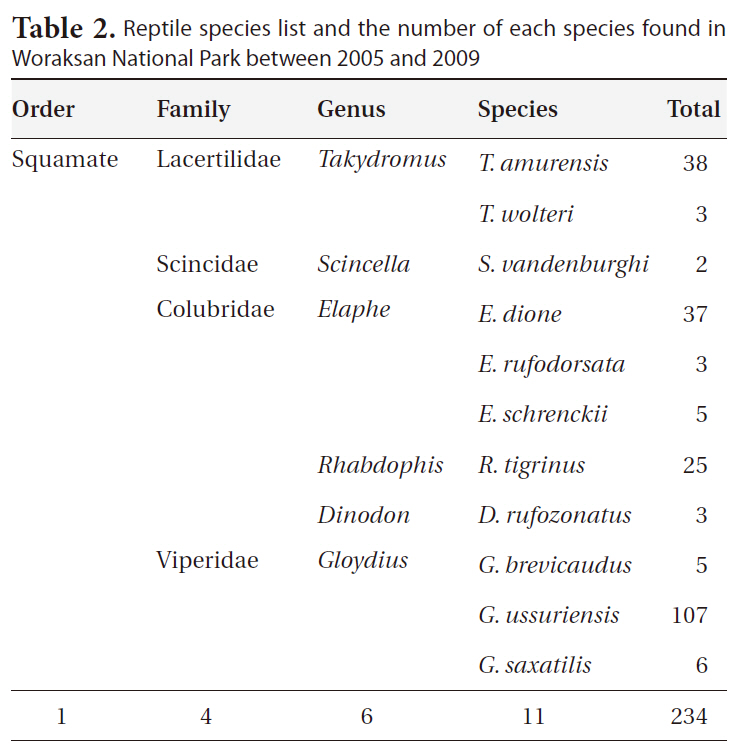
During the monitoring, we identified 11 reptile species (234 individuals) in six genera, four families, and one order (Table 2). The most dominant species was the Red-tongue pit viper, followed by the Long-tailed lizard (
Reptile species evenness was positively correlated with monthly mean precipitation (r = 0.900, N = 5, P = 0.037), and both reptile species diversity and evenness evidenced a somewhat positive relationship with monthly
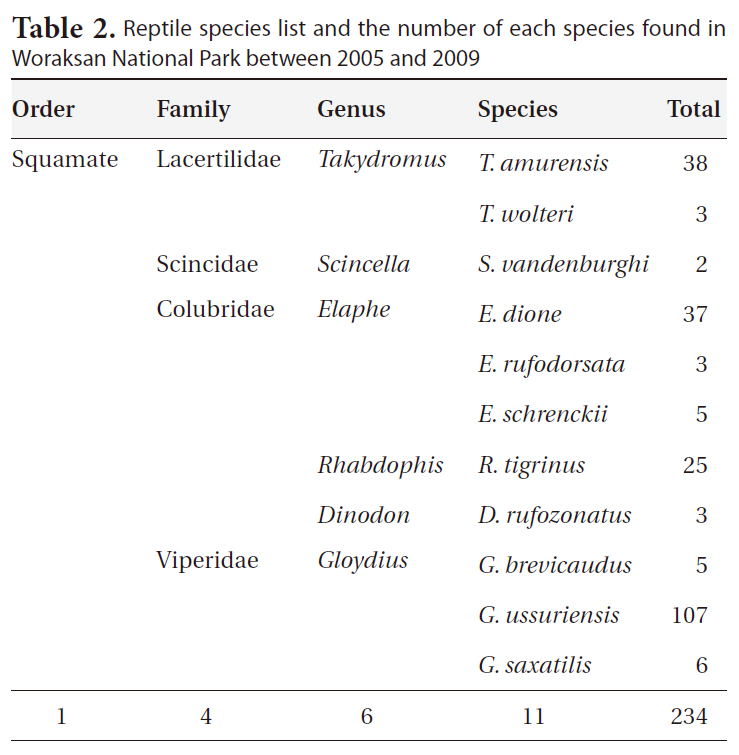
Reptile species list and the number of each species found in Woraksan National Park between 2005 and 2009
mean temperature (
Our results demonstrated that amphibian and reptile communities responded sensitively to climate changes, and in particular, to the sudden climate changes that occurred in 2007 (Sin et al. 2008). Sensitivity to and effects of climate changes varied depending on species and habitat, thereby underlining the need for studies of individual species and their response patterns in the near future.
Our results imply that the effects of climate changes at the valley sites and mountain edge sites can differ substantially. In this study, increased monthly mean precipitation negatively affected amphibian species diversity and evenness in the valley sites, but positively affected amphibian species richness in the mountain edge sites. Increased precipitation might result in a reduction in potential amphibian breeding sites in valleys, such as small ponds along stream lines, and might remove oviposited amphibian egg clumps from those areas. By way of contrast, in the mountain edge sites, increased precipitation could provide more breeding sites for the Korean salamander, Dybowskii's brown frog, and Black-spotted pond frog, and might also extend their breeding period. orgOur results are consistent with previous results showing that climate changes affect amphibian populations differently based on their habitats and locations (Buckley and Jetz 2007, Soares and Brito 2007), and our results indicate that efforts to conserve mountain frogs in Korea would be best served by knowledge regarding how amphibian individuals at valley and mountain-edge sites interact in detail.
In this study, the annual minimum temperature was shown to negatively affect the diversity and evenness of amphibian species, thereby indicating that increased annual minimum temperature induced a reduction in these indices. Among a variety of factors, winter temperature in the temperate climate zone is an important factor in the winter mortality of amphibians (Anholt et al. 2003, Reading 2007). Temperatures that are too low could result in death during hibernation, whereas overly high temperatures could also increase mortality due to excessive energy losses (Costanzo et al. 1997). Several previous studies addressing the manner in which climate changes affect hibernation patterns have shown that increased winter temperatures negatively affect amphibian populations (Anholt et al. 2003, Reading 2007). In particular, the fact that the annual minimum temperature affected species evenness demonstrates that different species might be differently affected, and also suggests the necessity of further studies to elucidate more clearly how different species respond to annual minimum temperature fluctuations.
Monthly mean precipitation affected reptile species evenness, whereas monthly mean temperature tended to affect reptile species diversity and richness; these results indicate that both temperature and precipitation are important factors for reptiles. In particular, the numbers of the Dione's ratsnake and Korean tiger keelback snake were correlated with the monthly mean temperature and precipitation during the winter season. Increased winter temperatures positively affected the number of individuals of the species, as opposed to the negative effects of increased winter temperatures on amphibians. Considering that temperature fluctuations during hibernation are related to the mortality of snakes, which breed in the spring (Bauwens 1981, Gregory 1982), the increased winter temperatures in this study may have reduced the mortality rates of the Dione's ratsnake and Korean tiger keelback snake. By way of contrast, winter temperatures do not appear to affect significantly the snakes that breed in summer, such as the Red-tongue pit-viper and Long-tailed lizard.
Considering the long-term ecological monitoring efforts thus far conducted in developed countries (Gibbs and Breisch 2001, Whitfield et al. 2007, Olson 2009), the five-year monitoring duration of this study is relatively short. Nevertheless, our results amply demonstrate the sensitivity of amphibians and reptiles to climate changes, and also the variance in their responses depending on species and habitat. In order to gain more detailed insights into the effects of recent climate changes and to better prepare for such changes, continuous long-term monitoring programs should be implemented, coupled with studies of phenological changes, physical and behavioral responses, and adaptation responses. Additionally, ecological modeling on a national scale to determine the expected changes in amphibian and reptile distributions in response to climate changes should prove helpful in the development of long-term conservation plans for amphibians and reptiles on the Korean peninsula.