A paddy field is a flooded parcel of arable land used for growing rice and other semi-aquatic crops. Paddy fields account for approximately 15% of the global arable land area (Huke and Huke 1997, Maclean et al. 2002). In South Korea, paddy fields are the most common type of agricultural land, covering approximately 60% of all agricultural land. Additionally, rice cultivation is the most important agricultural industry and greatly influences the regional economy (Korea Rural Economic Institute 2005). However,land-use patterns of agricultural landscapes have changed due to physical and economic urbanization(e.g., buildings and pavement, less competitive pressure,and compensation systems for abandoning agricultural land). In particular, in addition to paddy fields in the lowland areas, a number of paddy fields in hilly or mountainous areas are no longer cultivated because of a rapid decrease in the rural population (Chae et al. 2007).
Recently, interest in abandoned paddy fields has increased with respect to ecological restoration and succession.Abandoned paddy fields act as a bridge between paddy fields and secondary forests and decrease grassland-specific plant diversity within several years.Therefore, many studies have focused on evaluating vegetation in abandoned paddy fields (Lee et al. 2002) and factors causing changes in the composition of plant species,such as soil moisture (Hakoyama et al. 1977), light conditions (Kang et al. 2004), and microclimate (Lee et al. 2002), under non-management and wet conditions during secondary succession. Some of these studies have examined the effectiveness of restoring abandoned paddy fields with respect to improved agricultural landscape biodiversity (Comin et al. 2001, Yamada et al. 2007, Uematsu et al. 2010). Paddy fields and abandoned paddy fields provide forage, refuge, and a place for overwintering or estivation for many species, including soil macrofauna and microfauna, insects, and birds (Kato 2001, Thomas et al. 2004, Katoh et al. 2009, Paik et al. 2009). Among these, carabid beetles constitute an important component of the beneficial polyphagous predator fauna of cultivated land in temperate regions and are used as environmental change and habitat quality indicators (reviewed by Kromp 1999, Griffiths et al. 2007, Niemela and Kotze 2009). More specifically, in wet habitats such as paddy fields, abandoned paddy fields, wet grasslands, riversides, and lowlands with different vegetation, lower soil pH, and higher soil moisture than surrounding areas, carabid beetles can be characterized by species composition, food preference, and habitat selection (Hengeveld 1987, Luff et al. 1989, Eyre et al. 1990, Do and Moon 2002, Do et al. 2007).
We monitored carabid beetles in a spatially connected area (paddy fields, abandoned paddy fields, and mountains) to investigate differences in species diversity and composition for each habitat. The purposes of this study were to (1) investigate differences in diversity and species composition of carabid beetles in paddy fields, abandoned fields, and mountains, and (2) to confirm changes in carabid beetle composition patterns that follow cultivation of abandoned paddy fields, thereby considering species composition, species characters, and appearance patterns using a self-organizing map (SOM). Confirmation of the diversity and composition patterns of carabid beetles will provide information for effectively managing abandoned paddy fields.
The study was conducted in three habitat types: a paddy field, abandoned paddy fields, and mountain sites near Yangsan City, Republic of Korea. We selected four paddy field sites (cultivating
>
Data collection and analysis
To study diversity and structure of adult carabid beetle communities, we utilized pitfall trapping during 1-week trapping periods between mid-September 2003 and October 2005. In paddy fields, pitfall traps were installed on the levee between rice fields, because the inner areas of the paddy fields in which rice was growing were filled with water. However, in abandoned paddy fields, pitfall trapping was conducted inside the sites. Eight pitfall traps (7-cm diameter plastic cups), partially filled with propylene glycol, were installed at 4 m intervals at each site. Carabid beetle diversity was calculated using the Shannon-Weaver diversity index (
An SOM was used to cluster the study sites and identify the carabid beetle distribution pattern. This algorithm is also called a Kohonen network and is a non-supervised artificial neural network used to reduce data dimensionality during non-linear data processing (Kohonen 1984, Chon et al. 1996, Kohonen 1998). In addition, several review articles have emphasized the efficiency and performance of neural networks and SOMs for ecological patterning compared to conventional statistical methods (Gevrey et al. 2003, Kalteh et al. 2008). The detailed approach and methodological aspects can be found in other computational studies (Kohonen 1998, Kohonen and Hauta-Kasari 2008).
Determining map size is an important feature when applying a SOM (Jeong et al. 2010). In this study, map quality was evaluated using two types of error values: a quantification error and a topographic error. The former is the most common method used to determine the topological representation of the SOM. It uses the average distance between each data vector and the weight vector of the best-matching unit (BMU, the node that has the smallest Euclidean distance) and is usually used to measure map resolution (Kohonen1998). The latter is an alternative, extended index used to measure topological preservation, which uses input samples to determine how continuous the mapping is from the input space to the clusters. This error measures the proportion of data vectors for which the first and second BMUs are not adjacent units (Kohonen 2000).
The SOM algorithm weight values were determined by linear initialization, which is more effective for accelerating the learning phase as well as a more appropriate method for recognizing patterns in small data sets, such as ecological data collected and measured over a short period. In contrast, the other common method, random initialization, sometimes causes a “boundary effect” near the edges of networks (Kiang et al. 2001, Su et al. 2002). However, linear initialization is constrained for selecting the best topological network due to the same weight value being used at the beginning of training. Thus, we attempted to vary the size of the SOM according to the possible range using the data number to mitigate this drawback of the model.
Because there is no strict rule for determining SOM size (Park et al. 2007), the optimal size of the SOM was determined by trial and error (from 2 × 2 to 10 × 10). We trained SOM models with different map sizes and selected the model that exhibited the smallest topographic and quantification errors. After training, the SOM model nodes were clustered according to a calculated U-matrix, which discriminated one SOM node from another according to the non-linearly calculated dissimilarity. SOM modeling was conducted using Matlab 6.1, and the SOM coding solution was based on the SOM Toolbox in Matlab (Alhoniemi et al. 2000). Further information on this modeling approach can be found in Kohonen (2000). We used Matlab 5.3 and the SOM Toolbox in Matlab to develop the SOM model.
Experience in the use of SOM was gained using a time-series dataset for the study sites. All data points were labeled for species and sites. The data were then clustered according to the calculated U-matrix, and habitat labels were distributed on the SOM plane. Each parameter had a respective SOM plane represented by gradient coding.
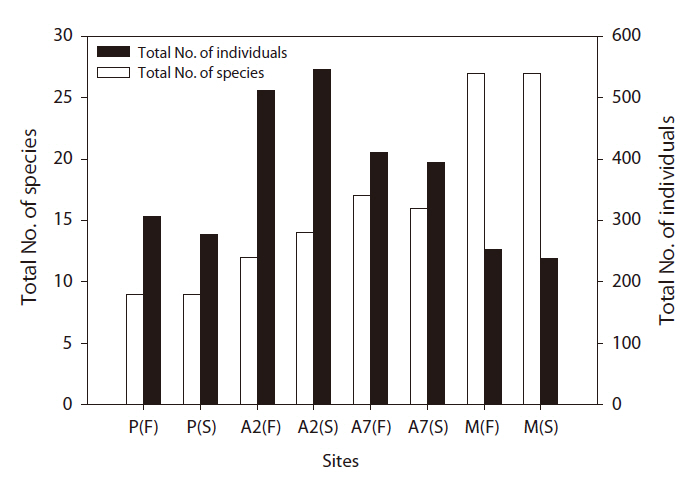
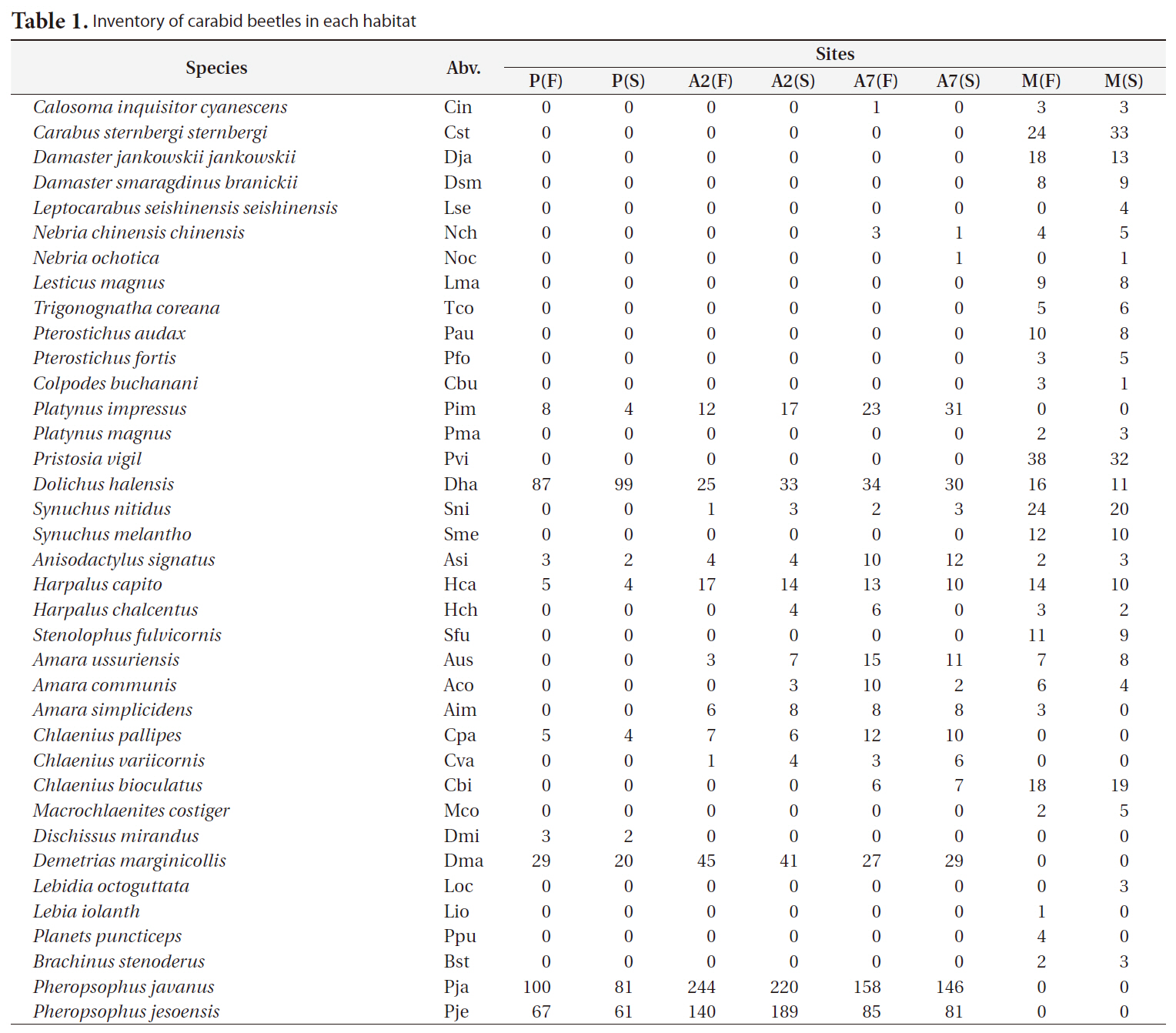
A total of 2,938 carabid beetles were collected in the paddy fields, abandoned paddy fields, and mountainous area and were classified into 37 species. In the paddy fields, 584 carabid beetles belonging to nine species were collected (Table 1, Fig. 1). The mountain site had the greatest number of species at 30, followed by the abandoned paddy field with 18 species: 18 species from the 7-year-old abandoned sites and 14 species from the 2-year-old abandoned sites. The current paddy fields had the lowest number of species at nine. Statistically significant differences in individuals of each species were observed (Kruskal-Wallis test
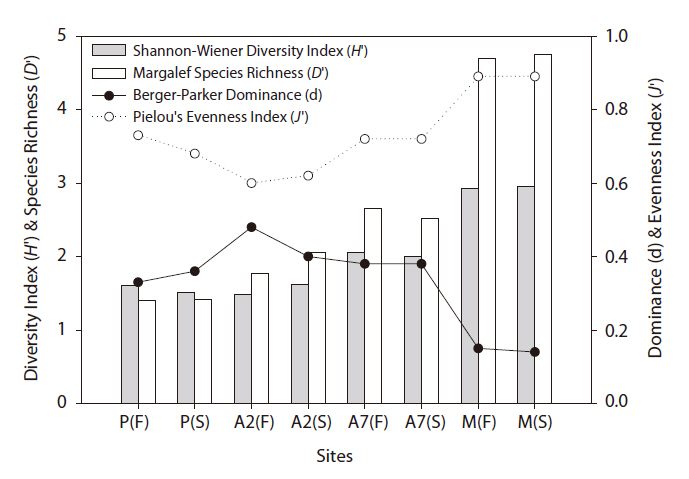
The diversity index was higher for the mountainous area than that in the other habitats. As the number of years of abandonment increased, the diversity of carabid beetles also increased (y = 0.02201x + 1.0257,
[Table 1.] Inventory of carabid beetles in each habitat
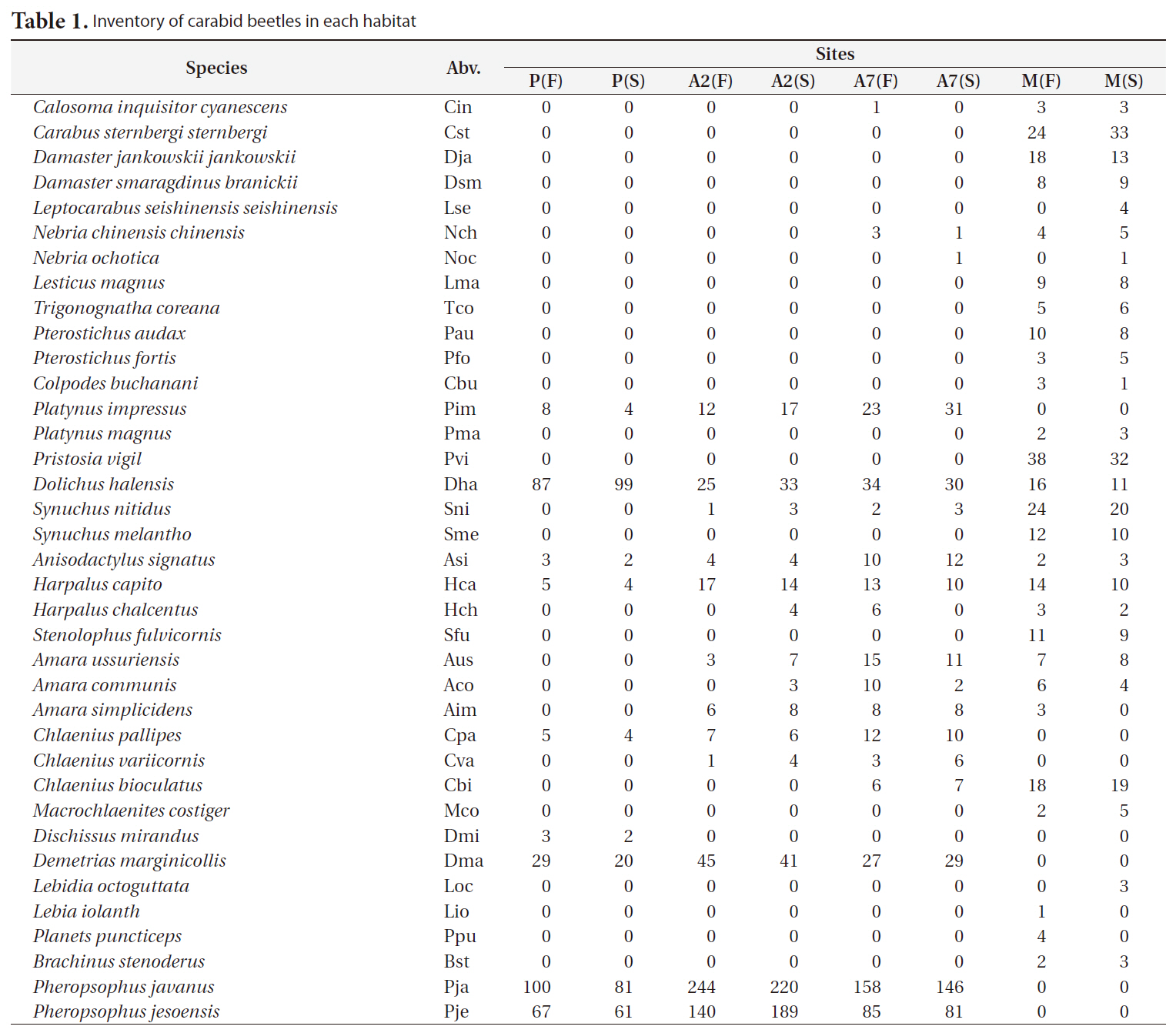
Inventory of carabid beetles in each habitat
>
Changes in species composition in different habitats
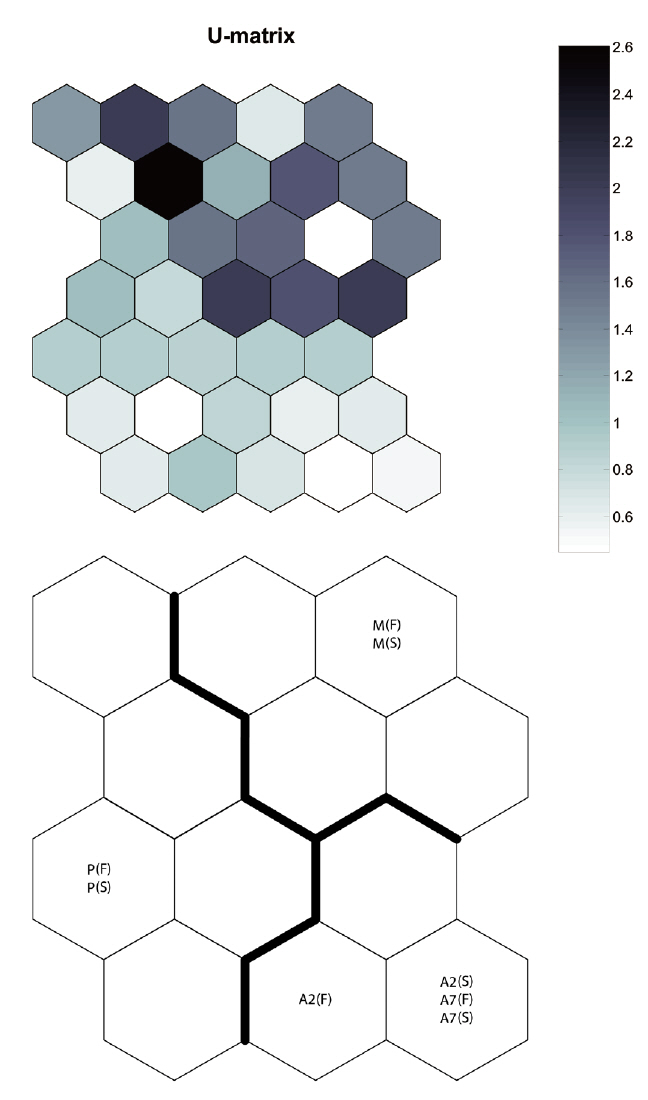
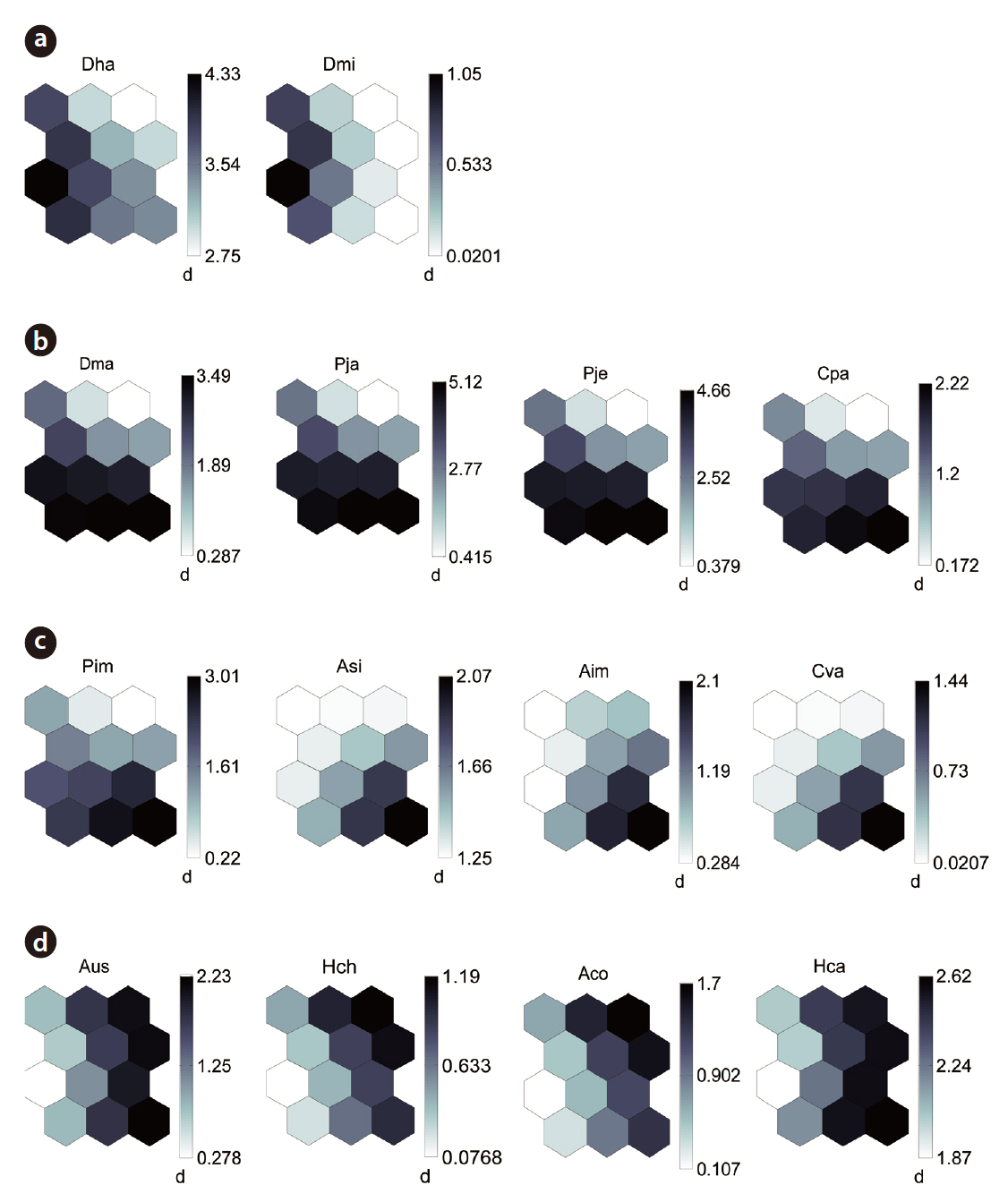
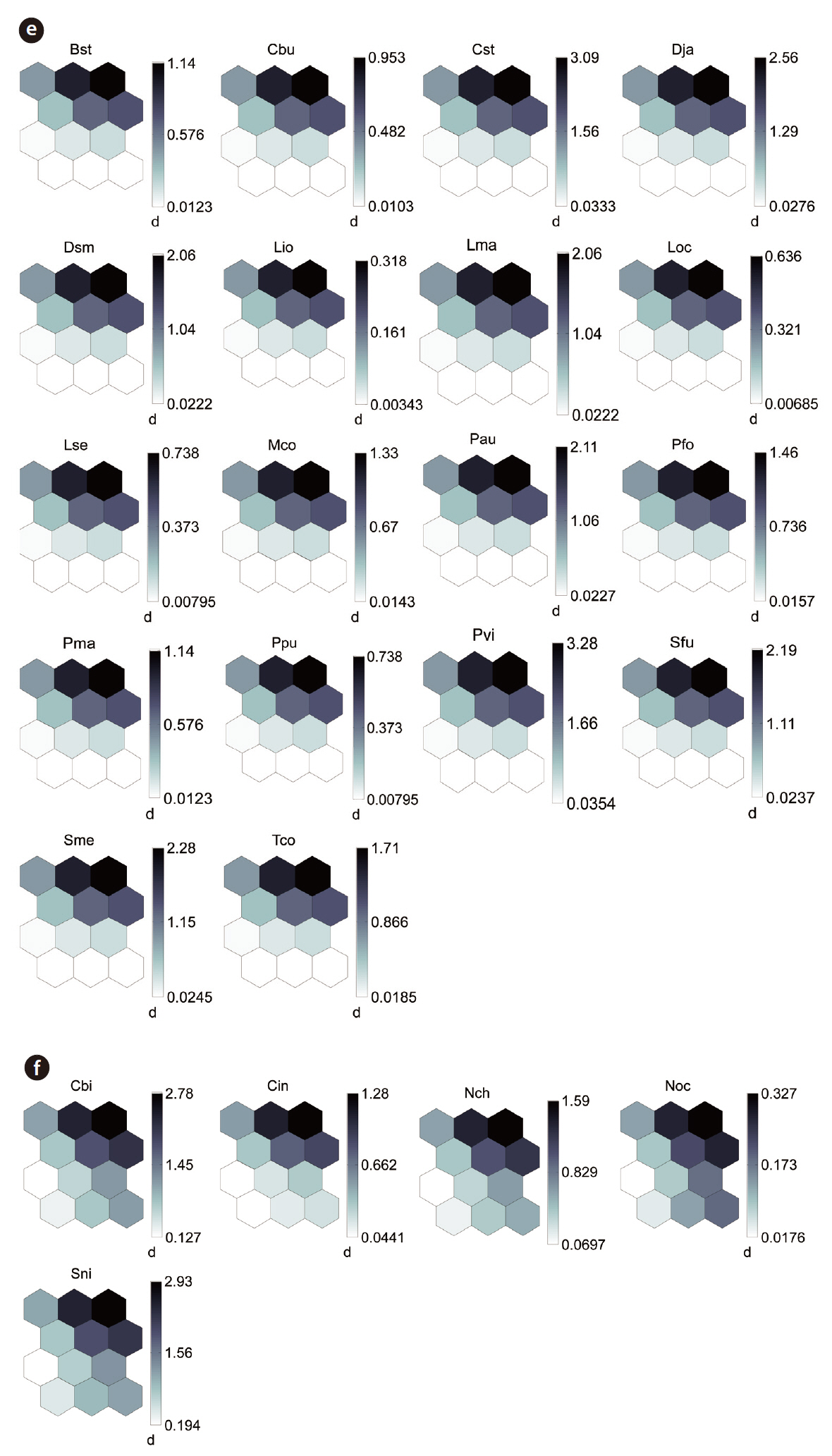
By applying the SOM, the study sites were divided into three groups based on the U-matrix (Fig. 3, quantization error, 0.880; topographic error, 0.000). This figure illustrates where each habitat was arranged on the SOM plane, and clusters are shown by thick solid lines around hexagonal nodes. The paddy fields and mountain areas were distinct from the abandoned paddy fields (Fig. 3). Compared with the input variable planes, the SOM plane labeled according to the habitat enabled us to infer the parameter attributes.
The distribution patterns of the 37 species (input vari-
[Table 2.] Change in carabid beetles through the ecological succession
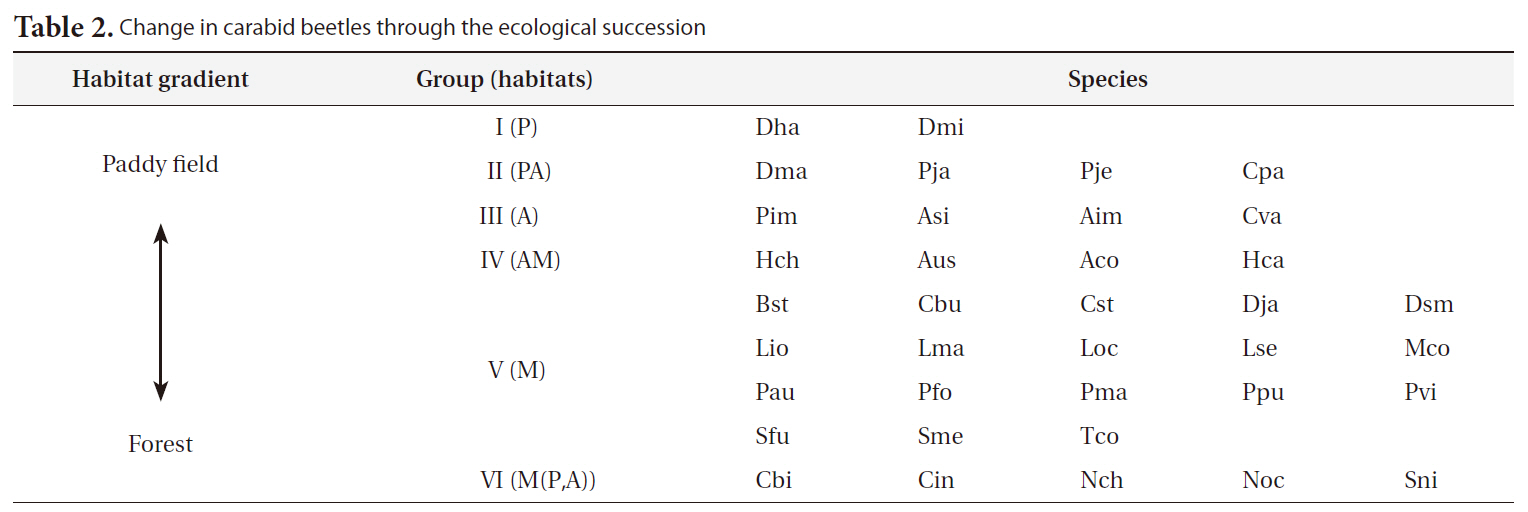
Change in carabid beetles through the ecological succession
ables) on the SOM plane allowed us to categorize six species groups (Table 2, Fig. 4). These species groups could be used to explain changes in species composition in response to paddy field abandonment. The gradients for
Carabid beetle diversity in abandoned paddy fields was higher than that in paddy fields. Carabid beetle diversity and composition were related to the cultivation system (e.g., cultivation with a fallow period, paddy field area, application of agricultural chemicals, etc.) and adjacent habitat types (e.g., forest, orchard, dry field, pond, etc.) (Kinnunen and Tiainen 1999, Huusela-Veistola 1996, Petit and Usher 1998, Varchola and Dunn 1999). In particular, artificial management had a more negative effect on larger carabid beetle species and tended to favor species preferring drier conditions (Blake et al. 1996).
The abandonment of rice paddies results in the initiation of secondary succession, which can lead to increases in biodiversity. The process of abandonment releases space previously used for cultivation, which becomes available for other plant species, typically successional species, to utilize. This opening of space allows plants previously not present in the paddy field to grow and in-
crease habitat diversity from the previous paddy field. As plant diversity increases, it is accompanied by an increase in carabid beetle diversity. As the period following abandonment increased (2-7 years), the diversity increased, reaching its greatest level at the mountain site, which had not been subject to rice paddy cultivation. While the initial stages of abandonment showed little variation from the cultivated rice paddies, they showed small changes in community assemblages. However, a larger change in the
carabid beetle assemblage was noted in the period from 2-7 years following abandonment. The greatest change in carabid beetle diversity occurred when moving from the 7-year site to the mountain site
Based on the SOM clustering, we divided the species into six groups; group II comprised species that prefer wet habitats and were adapted to water. In paddy fields, where water cultivation was practiced, group II species were more abundant and richer in diversity than at other locations. Populations of early successional species decrease according to the longevity of each species and environmental factors (Egler 1954, Southwood et al. 1979, Corbet 1995). However, the groups IV and V populations increased as the number of years of abandonment increased. These groups were found later in abandoned paddy fields because these fields are drier than paddy fields, and the species in these groups preferred a drier habitat than those in other groups. Sanderson et al. (1995) argued that the distribution and diversity of carabid beetles in wetlands preceding aquatic succession is influenced by plant height. Tall densely growing herbs and soil moisture act as a barrier to carabid beetle movement early in succession. Generalists and open-habitat species continuously increased in the paddy fields abandoned for 2 years compared to those in currently used paddy fields.
To date, the SOM technique has been rarely applied to carabid beetles. The SOM algorithm is a widely used machine learning technique, particularly for data clustering. Successful applications of the SOM can be found in a variety of ecological studies (Chon et al. 1996), such as studies of basin management and water dynamics (Garcia and Gonzalez 2004), fish distribution patterns (Kruk et al. 2007, Lasne et al. 2007), freshwater macroinvertebrates (Penczak et al. 2006, Lek-Ang et al. 2007, Park et al. 2007), trophic structures (e.g., Giraudel and Lek 2001), and habitat restoration assessment (Song et al. 2006). Using SOM for ecological patterning is superior to conventional statistical methods (Giraudel and Lek 2001). This algorithm is also used in freshwater ecosystem sciences, and our present results indicate that this study can be utilized as a good example of clustering in a terrestrial insect community according to habitat characteristics.
In the Republic of Korea, agricultural landscapes generally contain a mosaic of mixed forests, paddy fields, dry fields, grasslands, streams, ponds, and reservoirs for irrigating small agricultural landscapes (Cho et al. 2009). This mosaic landscape supports differences in carabid beetle diversity and species composition within each habitat (Fournier and Loreau 2001). We also found that individuals were habitat specific (cf. Fig. 3, SOM U-matrix), but changes in species composition created a sere from the paddy field to the mountains. These results indicate that abandoned paddy fields in small agricultural landscapes may play an important role as an aneco-tone zone, comparable to hedgerows between paddy fields and mountains. Efforts have been made by the South Korean government to enhance habitat quantity by creating small foothills, planting trees (e.g., willow, persimmon tree, chestnut), introducing no-cultivation (fallow) years, and alternating crops (practice crop rotation) (Korea Rural Economic Institute 2005). However, most abandoned paddy fields are privately owned, and many landholders who live near the city want their property status altered in the hope of a speculative investment, even though these management practices are ineffective (Hwang 2010). Of course, it is a serious problem that too many paddy fields are abandoned and become urbanized. Abandoned paddy fields in some areas and adjoining mountainous areas, provided as a precondition that they be managed and controlled, are an opportunity to improve diversity and habitat quality in small agricultural landscapes.