Approximately 60% of Iran is classified as arid and semi-arid. Much of the country has a desert climate, with an average annual precipitation of less than 300 mm(Mansoory 1992). The zagros forests of Iran are classified as semi-Mediterranean forests because of their geographic location and the effects of Mediterranean climate on it. Zagros mountains are the original source of
Brant's oak only regenerates by seed. Seed quality is also critical for the early vigor of a new plant. Early vigor is a combination of the ability of the seed to germinate and emerge after planting, and the ability of the young plant to grow and develop after emergence (Jin and Wang 2002). Seed and uniformity of germination are limiting factors in forest trees (Fenner 1992). Seed germination plays an important role in the regeneration of plant species, especially under unpredictable environmental conditions like those in Mediterranean ecosystems (Gimenez-Benavides et al. 2005). Poor germination and seedling establishment are regarded as the major causes of low densities in Mediterranean forests (Close and Wilson 2002). Seed performance also depends on vigor (Copeland and McDonald 1995). Seed vigor can be affected by provenance or by environmental factors operating during development and maturation, such as seed germination, seedling establishment, and survival regulated by light, climate, edaphicity, and species-specific factors (Taghvaei 2006, Liu et al. 2008).
Environmental factors such as temperature affect germinability of seeds of many species during seed maturation (Pourrat and Jacques 1975). In different plant species, even small differences in temperature during plant development or seed maturation can influence the germinability of seeds (Gutterman 1996). Light quality affect on germinability during seed filling too. Altitude can also influence germinability of seeds; the higher the altitude of seeds, the lower their rate of germination is (Dorne 1981). Seed provenance affects the seed germination, seedling survival, and growth of
In tropical dry forests, germination, survival, and seedling growth are affected by light intensity (Khurana and Singh 2001). The reflex of germination to light in-
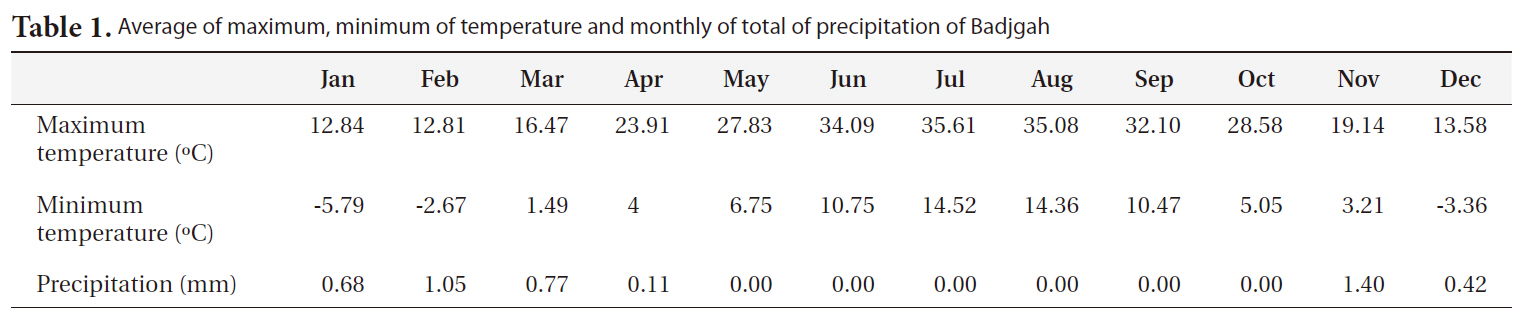
[Table1.] Average of maximum minimum of temperature and monthly of total of precipitation of Badjgah
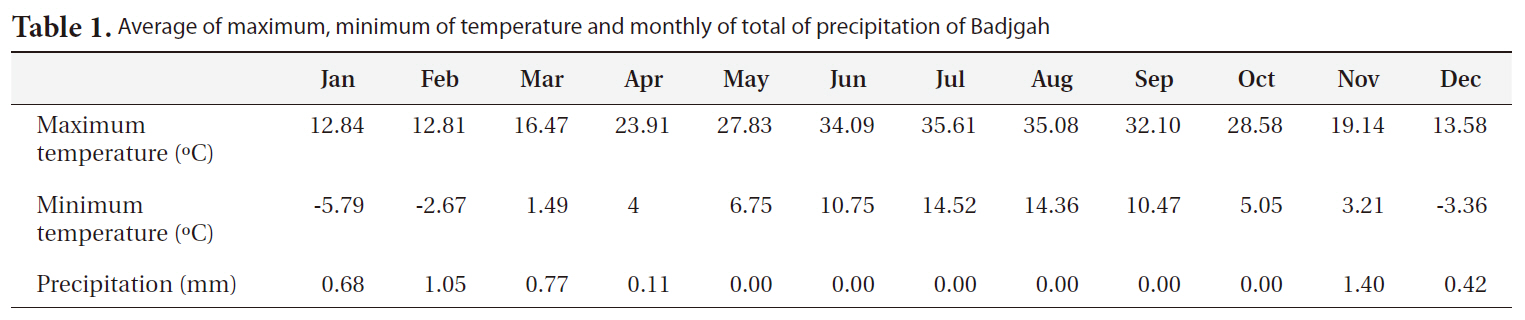
Average of maximum minimum of temperature and monthly of total of precipitation of Badjgah
intensity depends on the species and environment. Biomass production, relative growth rate, root/shoot ratio, specific leaf area (LA), net assimilation rate, and LA ratio can be limited by either poor light availability (Rincon and Huante 1993) or low soil moisture due to water use by nurse trees (Rodriguez-Calcerrada et al. 2008) in the dry forest understory. The shade protects soil moisture during seed germination in dry forests. Also it has been found that germination success was approximately equal in shaded and un-shaded sites for some species (Ray and Brown 1995). In dry forests, species germination and early establishment must occur during the first of the wet season when water is at its most available (Khurana and Singh 2001). The general assumption of this study is that provenance variables are a complex of environmental factors and shade changes in early stages of the life cycle of an acorn seed. The aim of this study was to test whether seed vigor and seedling performance are affected by altitude seed source and shade during seedling establishment.
Seed lots of Brant's oak (
Experimental design
The experiment was set up within an intact area of the College of Agriculture (Badjgah 29°50' N, 52°46' E) at Shiraz University, located at an elevation of 1,810 m a.s.l. in the southwest of Iran (Table 1). The experimental de-sign was a split plot one based on a complete randomized block with four blocks: two light levels (no shading, partial shading) were assigned to the main plots, while four seed source sites (altitudes of 850, 1,100, 1,500, 2,100 m a.s.l.) were assigned to the sub plots. There were two types of shade: no shade (full sunlight) and 50% shade by shade cloth. The shade used after seed planting to final experiment.
To determine the effects of provenance and light on the germination of seeds and the early establishment of seedlings, the seeds from each treatment were planted in a 0.5-L plastic pot filled with 500 g of sandy loam soil. The pots were watered weekly with sufficient water. Emerging seedlings were counted every three days, continuing until when no further germination was recorded, and then vigor-related traits were measured on individual plants after five months. The traits were: emergence percentage (EP), shoot length (SL), and root length (RL). Chlorophyll content (CC) was recorded using the lowest seedling leaf fully expanded from the top of the main stem for each pot at 10:00-12:00 a.m. by a SPAD chlorophyll meter (Minolta SPAD-520; Minotal, Ramsey, NJ, USA) by LA (cm2) at the end of this period. LA was measured on collected leaves of the shoot with a LA meter, model MK2 (Delta-T Co., Cambridge, UK). SL was measured by the length from the top to the shoot, and RL was measured by the length from the top of the root. Shoot dry weight (SDW) and root dry weight (RDW) were measured after drying for 24 h in an oven at 70°C (ISTA 1999). Mean time of emergence (MTE) and emergence rate (ER) were calculated for each treatment according to the equations of Ellis and Roberts (1981) and Agrawal and Dadlani (1992) as follows, respectively:
MTE = Σ (ni.ti) / Σ n
ER = Σ (ni/ti)
[Table 2.] Analysis of variance for seed and seedling parameters
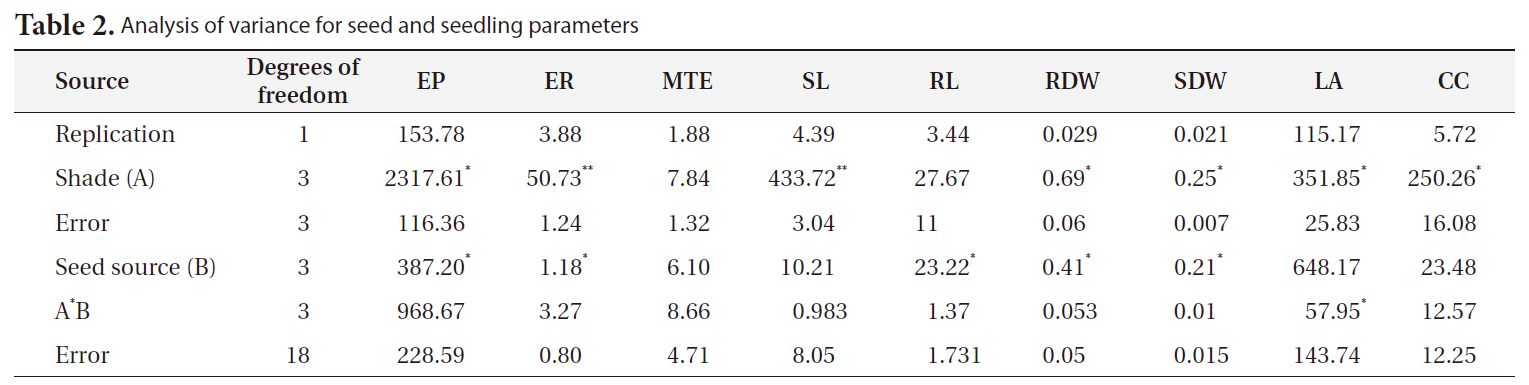
Analysis of variance for seed and seedling parameters
MTE: mean time to full emergence
ER: emergence rate
Where ni is the number of emerged seeds per day, ti is the amount of time counted from the beginning of emergence. CC was measured on the lowest seedling leaves with a Minolta chlorophyll meter.
Data were checked for normality, and then were analyzed using MSTATC statistical software (MStat Inc., East Lansing, MI, USA). Treatment means were separated by a Duncan test if the F-value of the treatment was significant at the 0.05 or 0.01 probability levels.
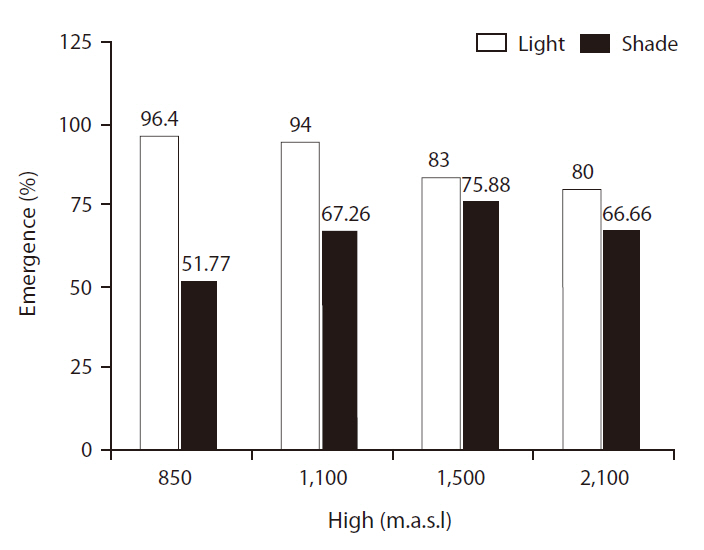
EPs were significantly different between light treatments (
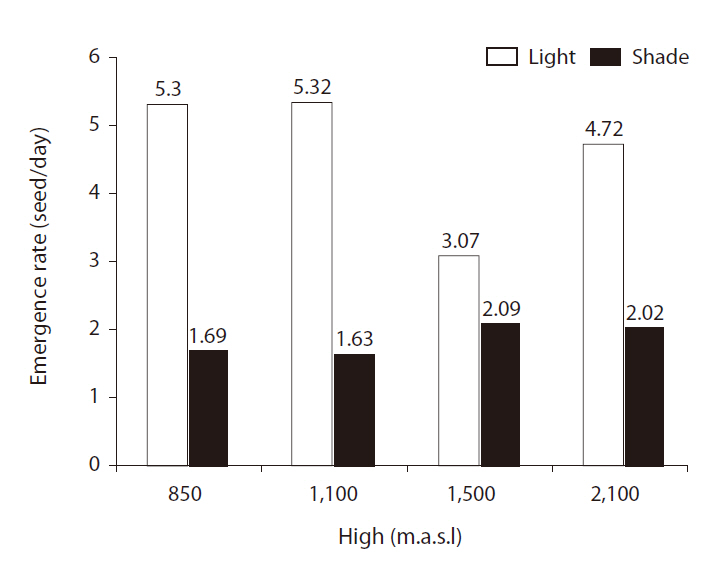
There were significant differences in ERs between light treatments (
or sites, but the highest seed ER (5.32) was obtained for altitudes of 850 and 1,100 m above sea level (Fig. 2).
>
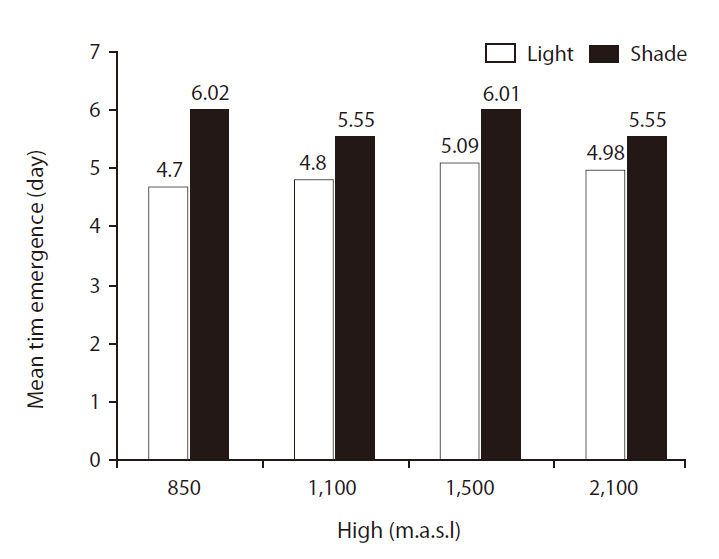
Mean time of emergence (MTE)
There were significant differences in MTE between light treatments (
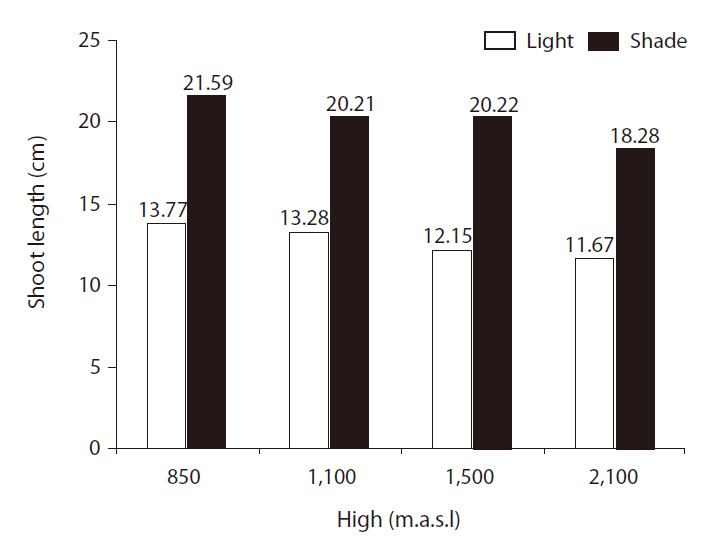
SLs were significantly affected by light treatment (
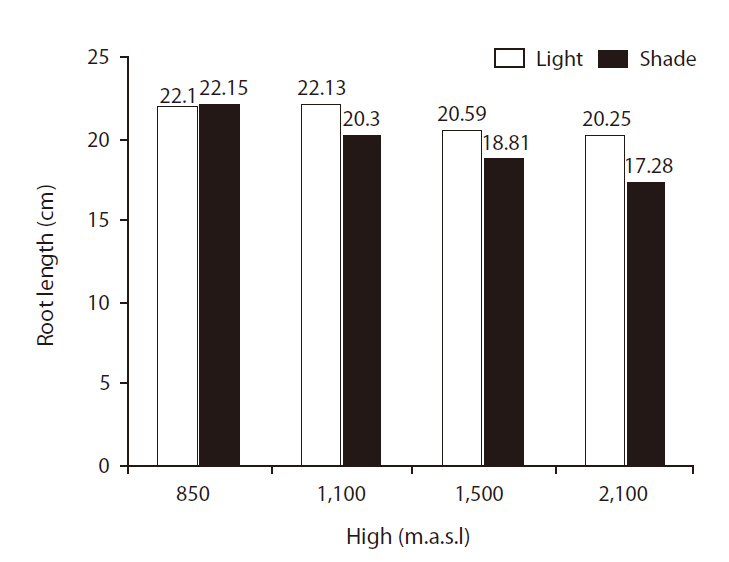
There were significant differences in RLs between site treatments (
>
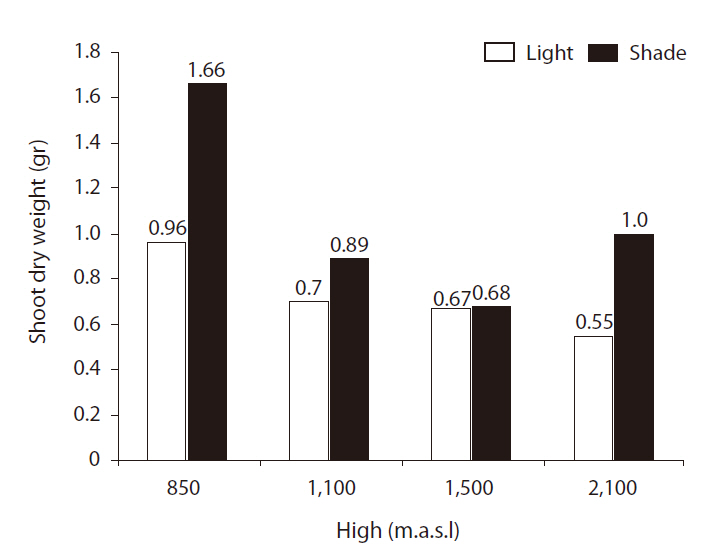
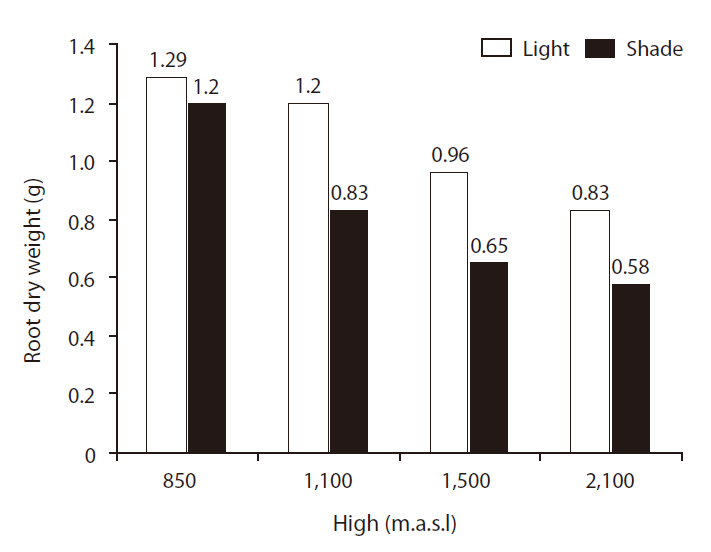
Shoot and root dry weights (SRDWs)
SRDWs were significantly affected by light (
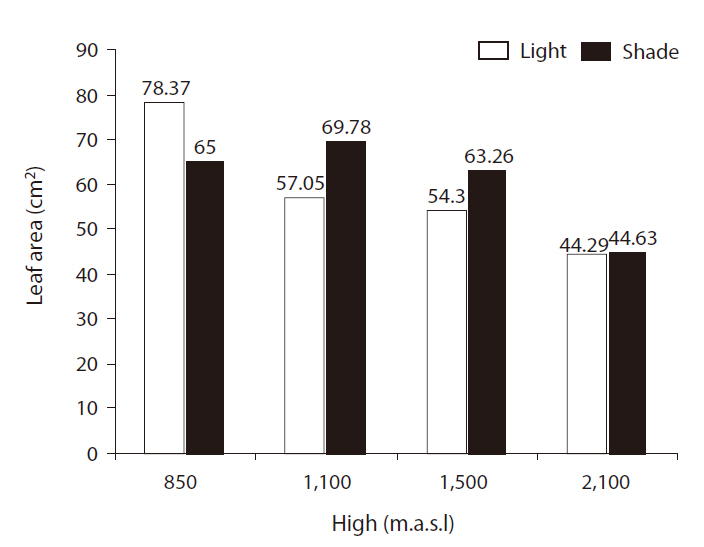
There were significant differences in leaf areas between light treatments (
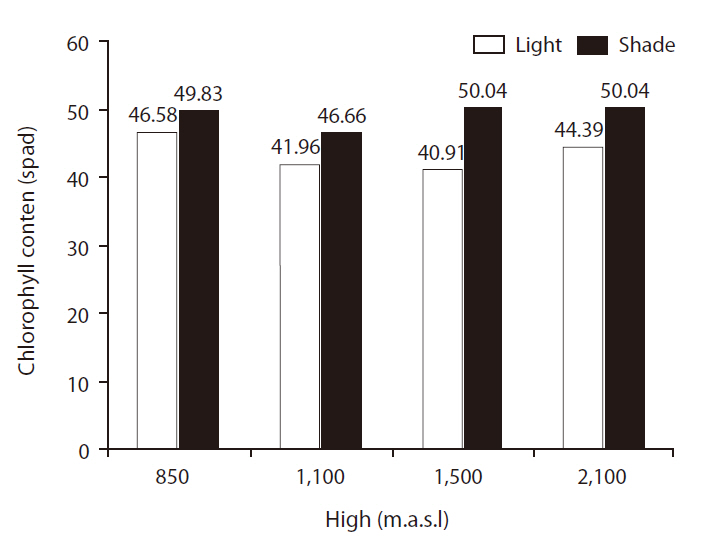
CC differed significantly between light treatments (
Source variation tests is one of method to screen the naturally available genetic variation to select the best planting material for higher productivity (Bhat and Chauhan 2002). The seed may respond to changes in environment (Egli 1998). This study has revealed that the present provenance is a factor controlling the performance of
Forest production differs from in open areas and under forest canopy (Chen et al. 1995). In this experiment, shade treatment had an influence on acorn germination, seedling emergence, and seedling growth. E