Activated carbons (ACs) are widely used in numerous fields, such as electric double layer capacitors, fuel gas storage systems, adsorbents, catalysts, and catalyst supports, because of their extended surface area, high adsorption amount and rate, and specific surface reactivity [1-5]. The adsorption capacity, which is one of the most important properties of ACs, depends on raw materials, the pore structure induced by the activation process, and surface functionality [6,7].
Depending on the nature of the raw materials, ACs are generally activated by physical activation or chemical activation. The advantages of chemical activation include a low activation temperature, a reduced activation time, a high surface area, a high yield, and an important reduction of the mineral content [8-10]. The activation agents reportedly used in the chemical activation process include potassium hydroxide (KOH), H3PO4, and ZnCl2, though KOH is the most widely used agent [11].
ACs can be manufactured from many kinds of raw materials, including nutshells, peat, wood, coir, lignite, coal, and petroleum pitch [8,12,13]. Pitch has long been known as an excellent raw material in the carbon manufacturing industry because of its high carbon content,low price, and ability to produce carbon with different characteristic structures and properties [14]. Furthermore, the crystallite structures of carbon materials are made up of many aromatic layers, and the carbon atoms on the layers are hard to activate because of the stronger covalence bonds; however, the edge carbon atoms (the noncrystallite carbon atoms) with unsaturated bonds have more reactive capabilities [15,16]. Pitch is difficult to activate because of its stable micrographitic structure and lack of initial pores. Furthermore, the fact that pitch usually needs a large amount of KOH to produce ACs makes it costly and environmentally unfriendly [17,18]. For these reasons, it would be of great value for further research if the technical conditions could be optimized to produce a lower KOH/pitch ratio; to date, however, very few studies have examined how chemical activation affects the developed porosity of petroleum pitch.
This work relies on Fourier transform infrared spectroscopy (FT-IR) to investigate the use of potassium permanganate (KMnO4)-pretreated petroleum pitch as an AC precursor; it also discusses the effect of KMnO4 pretreatment on the textural properties of petroleum pitch-based ACs.
2.1 Pretreatment of petroleum pitch
The petroleum pitch (Carbonix Co., Korea) was used as a precursor to prepare the ACs. A 2 g sample of pitch was added to 50 mL of concentrated sulfuric acid (H2SO4). The mixture was stirred for 12 h at room temperature, and then KMnO4 was added with constant stirring while the temperature of the mixture was maintained at 20℃. The pitch was treated with different amounts of KMnO4 (0 g, 0.5 g, 1.0 g, and 2.0 g). The KMnO4-treated pitch (K-pitch) was filtered, washed, and then dried at 100℃ overnight. The prepared samples of K-pitch were named 0K-pitch, 0.5K-pitch, 1.0K-pitch, and 2.0K-pitch.
The KOH impregnation process was initiated by adding 1 g of K-pitch to a 100 mL ethanol solution that contained 3 g of KOH. The mixture was stirred for 3 h at 100℃ and then dried for 24 h at 110℃. The K-pitch activation was completed by pyrolysis at 800℃ for 2 h at a heating rate of 5℃/min under N2. The activated product was washed with 1 M HCl and distilled water, filtered, and then dried at 110℃ overnight. The K-pitch-based ACs were named 0 K-ACs, 0.5K-ACs, 1.0K-ACs, and 2.0 K-ACs.
The structures of 0K-pitch, 0.5K-pitch, 1.0K-pitch, and 2.0K-pitch were characterized by using an X-ray diffractometer (XRD, D/MAX 2200V, Rigaku, Japan) with CuKα radiation. The XRD patterns were obtained in a 2 θ range between 10° and 60° at a scanning rate of 5°/min.
The different types of functional groups were identified by using a Fourier transform infrared spectroscope (JASCO FT/IR 4200) in the range of 1000 cm-1 to 4000 cm-1.
Nitrogen adsorption-desorption isotherms were measured at 77 K with BELSORP measuring instruments (BEL Japan Inc., Japan). The specific surface area was calculated with the Brunauer-Emmett-Teller (BET) equation. The total pore volume was obtained from the N2 adsorption at P/P0 = 0.990. The total micropore and mesopore volumes were calculated from the application of the Dubinin-Radushkevich (DR) equation. The average pore diameter (Dp) was calculated as follows:
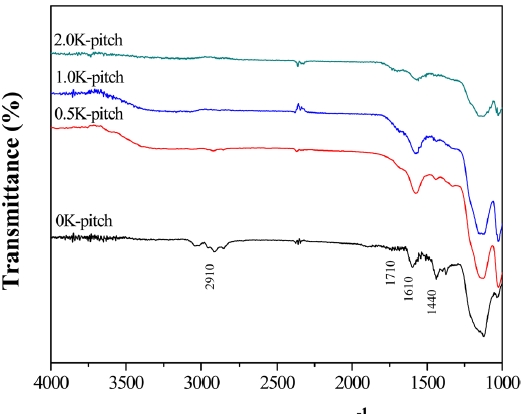
Fig. 1 shows FT-IR spectra of the K-pitch. The spectra can be assigned to asymmetric and symmetric ?CH stretching vibrations in aliphatic compounds such as ?CH3, =CH3, and ?CH2CH3. The most important features of K-pitch are the reduction of intensity in the IR bands at around 2910 cm-1 (which are assigned to the alkyl group) and the appearance of an IR band at around 1710 cm-1 (which is assigned to the C=O stretching vibration). Compared with the 0K-pitch, the 2.0K-pitch
[Table 1.] Microcrystalline structural parameters of the pitch and pretreated pitch

Microcrystalline structural parameters of the pitch and pretreated pitch
has a stronger C=O intensity at 1710 cm-1. The FT-IR results show that the pretreatment promotes the formation of surface oxygen groups and simultaneously decreases the alkyl groups of the pitch. The conversion process from alkyl groups to oxygen groups may occur during the KMnO4 pretreatment. The surface oxygen groups of the precursor may serve as an active site; it plays an important role in activation.
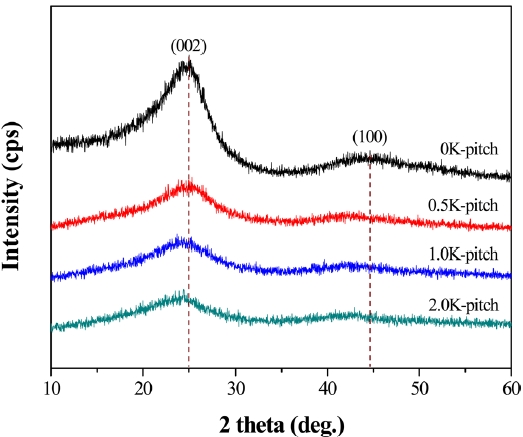
Fig. 2 shows XRD patterns of K-pitch. Two broad diffraction peaks occur at 25° and 43° in each spectrum; they correspond to the diffraction of (002) and (100), respectively. It can be seen that the 0K-pitch displays larger crystallites and good tropism; these traits are due to the narrower and sharper (002) peaks. On the other hand, K-pitch has more microstructural defects with the broader (002) peaks, which are indicative of the formation of noncrystallite carbons [15].
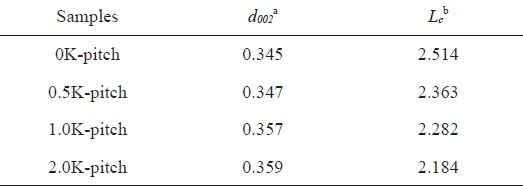
Table 1 lists the microcrystalline structural parameters of all the samples. The interplanar distance (d002) and the dimension (Lc) perpendicular to the basal plane of the graphitic microcrystallines in the samples were calculated with Bragg and Scherer equations [16]. The XRD results show that the crystalline thickness decreased from 2.514 nm for 0K-pitch to 2.184 nm for 2.0K-pitch. They also show that the interplanar distance, d002, for 2.0K-pitch (0.359 nm) is slightly larger than the interplanar distance for 0 K-pitch (0.345 nm). The difference in the interplanar distances is due to a disorder that occurs after the KMnO4 pretreatment. Thus, the microstructural defects of KMnO4-treated pitch increase the value of d002 and decrease the value of Lc.
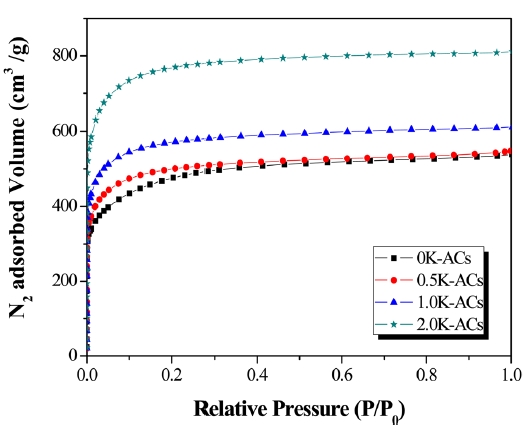
Fig. 3 shows N2/77 K adsorption isotherms of ACs prepared from K-pitch by chemical activation. In all cases, the obtained results correspond to Type-Ι isotherms according to the International Union of Pure and Applied Chemistry (IUPAC) classification;the results are similar to the results of previous research on different kinds of ACs [19]. The adsorption isotherms of all samples are typical microporous carbons with a very steep initial uptake at a low relative pressure. In accordance with their higher position on the isotherm curve, K-ACs have a greater adsorption ability than 0K-ACs.
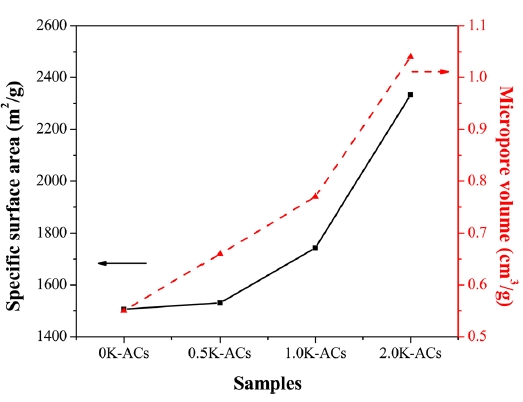
Fig. 4 shows the specific surface area and micropore volumes of all the samples calculated by the BET equation and the DR equation, respectively. The DR equation is as follows [20]:
where W is the amount of the liquid adsorbate at the relative pressure (p/p0), W0 is the total amount of adsorbate in the micropores, B is the adsorbent constant, and β is the affinity coef-
ficient (0.34 for nitrogen).
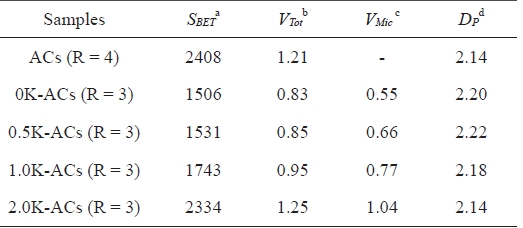
Table 2 lists the porous structural parameters of the ACs. The results suggest that the KMnO4 pretreatment prior to activation increases the specific surface area and total pore volume (Vt) of the resulting ACs. The specific surface area gradually increases as the amount of KMnO4 is increased. Jiang et al. [17] studied microporous carbon obtained from ACs prepared by chemical activation with KOH at a constant KOH/petroleum coke ratio of 4/1. The results reveal a specific surface area of 2408 m2/g and a total pore volume of 1.21 cm3/g. The porosity of the 2.0 K-ACs was obtained as a specific surface area of 2334 m2/g and 1.25 cm3/g. These results suggest that ACs prepared from KMnO4-treated petroleum pitch can be a way of producing ACs with a high surface area at a relatively low KOH/C ratio. This capability is attributed to the fact that the formation of oxygen groups leads to an increase in active sites on the pitch. The active sites make it easier for KOH to react with the carbon of the precursor. This process ultimately facilitates the development of the porous structure. Thus, pretreating the pitch with KMnO4 is a useful way of preparing the ACs with a low KOH/pitch ratio.
[Table 2.] Porous structure parameters of the pitch-based ACs
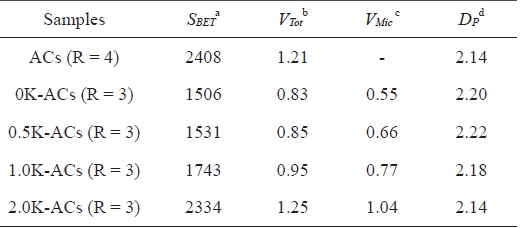
Porous structure parameters of the pitch-based ACs
In this work, ACs were prepared from KMnO4-pretreated petroleum pitch by chemical activation with KOH. And an evaluation was made of the effects of the KMnO4 pretreatment of petroleum pitch on the structural and textural properties of the resulting ACs. FT-IR analysis was used to identify the presence of oxygen groups on the surface of the KMnO4-pretreated petroleum pitch. The surface oxygen groups serve as active sites in the chemical activation with KOH. XRD reveals that the d002 value of the pitch can be increased by a modification process. Microstructural defects in the KMnO4-treated pitch increase the value of d002. Furthermore, the specific surface area and micropore volume are increased as the amount of KMnO4 pretreatment is increased. The of the pitch combines the oxygencontaining groups into graphitic microcrystalline. In short, the KMnO4 pretreatment, which involves chemical activation at a relatively low KOH/pitch ratio, is a useful way of producing ACs with a high surface area.