Over 90% of commercial algae biomass production is currently with large-scale open-pond systems. Such systems usually suffer from low production rates during the cold seasons (Sheehan et al. 1998, Benemann 2008). Hence, cyanobacteria isolated from the polar regions offer interesting potential for the production of biomass and biofuel due to their psychotrophic characteristics as Tang et al. (1997) previously reported that many high-latitude cyanobacterial strains remain active at low temperatures.
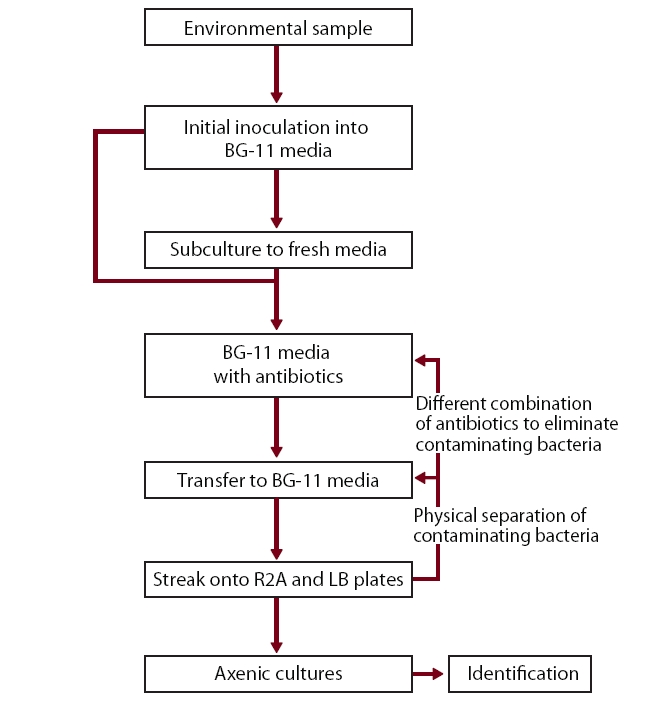
However, obtaining axenic cultures is essential for genetic, physiological and taxonomic research. Although numerous methods to produce axenic cultures of cyanobacteria have been suggested (Rippka 1988, Ferris and Hirsch 1991, Bolch and Blackburn 1996, Choi et al. 2008), it is still very difficult to eliminate all contaminating bacteria from environmental samples. Since cyanobacteria are a very diverse group, exhibiting enormous variations in growth, morphology and metabolic capabilities, any particular approach cannot guarantee success of purification.
The objectives of the current study were to axenically obtain cyanobacteria from an Arctic freshwater bloom sample by using a combination of antibiotics and physical separation techniques and to test the isolate’s cold-tolerance capability. Our eventual goal is to discover the mechanisms of cold tolerance in Arctic cyanobacteria, identify and isolate genes with high activity. These genes will then be incorporated into indigenous cyanobacteria for consistent biofuels production during the low-temperature seasons.
>
Sample collection and cyanobacterial isolation
Arctic cyanobacterial bloom samples were taken from the temporal water runoff region, located approximately 10 km east of Dasan Station in Ny-Alesund, Svalbard Islands in August 2009 (Table 1, Fig. 1). Samples were then taken to the laboratory and 1 mL of bloom samples were inoculated into 100 mL BG-110 medium (Rippka et al. 1979) with cycloheximide (Sigma, St. Louis, MO, USA) at a concentration of 250 μg mL-1. The flasks were incubated on an orbital shaker (Vision Scientific Co. Ltd., Bucheon, Korea) at 160 rpm and 15°C until cyanobacterial growth was apparent.
Well-grown cyanobacterial cultures (1.5 mL) were centrifuged at × 3,000 g for 15 min. Resulting pellets were streaked onto BG-11 agar supplemented with imipenem (100 μg mL-1) (Choongwae Pharma Corporation, Seoul, Korea) and cycloheximide (20 μg mL-1) and incubated in the dark for 24 hours to eliminate bacterial contamination. Plates were then incubated in a light : dark cycle (16 : 8 hours) at 15°C and filamentous growth was observed daily. When visible to the naked eye, emerging cyanobacterial filaments were aseptically transferred to fresh BG-11 plates to separate cyanobacteria from contaminating bacteria. Cyanobacterial filaments were then streaked onto R2A and LB agar plates (Becton, Dickinson and Company, Sparks, MD, USA) and incubated in the dark to check the axenic status of the culture for 7 to 14 days. Contaminating bacteria that survived the imipenem treatment were identified by 16S rRNA gene sequencing (Lane 1991). Non-axenic (bacterially contaminated) cyanobacterial culture was further incubated on BG-11 agar with kanamycin (100 μg mL-1) (Duchefa Biochemie, Haarlem, The Netherlands) for another 24 hours in the dark and the rest of the purification steps were repeated until a pure culture of the cyanobacterium was obtained (Fig. 2).
[Table 1.] Description of sampling point

Description of sampling point
>
Morphological identification
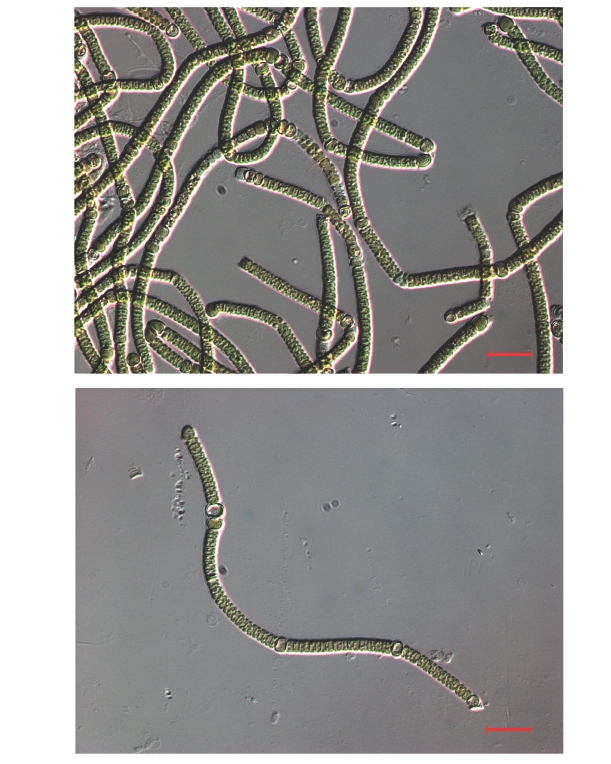
The strain was grown in BG-110 medium (without nitrogen) for 10 days. Live cells were harvested and suspended in sterile dH2O and inspected at × 400 magnification on a Zeiss Axioskop 2 light microscope (Carl Zeiss, Korea Co. Ltd., Seoul, Korea) equipped with differential interference contrast optics.
PCR conditions and the primer sets CYA106F, CYA781R(a), and CYA781R(b) were used for 16S rRNA sequence analysis as described by Nubel et al. (1997). The phycocyanin encoding operon intergenic spacer (PC-IGS) region was amplified using the primer pair, PCβF and PCαR specific for cyanobacteria (Neilan et al. 1995). Region ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)
A seed culture of Nodularia spumigena KNUA005 was inoculated into both BG-110 and BG-11(+) media in triplicate and incubated in a light : dark cycle (16 : 8 hours) on an orbital shaker at 160 rpm and 10, 15, 20 and 25°C for 21 days. Cyanobacterial density was determined by measuring the optical density of a culture at 750 nm on an Optimizer 2120UV spectrophotometer (Mecasys Co. Ltd., Daejeon, Korea). Then, growth curves were drawn to reveal the effect of temperature on
>
Axenic production of the culture
After imipenem treatment and physical separation by streaking onto BG-11 agar plates, two strains of contaminating bacteria still survived and co-existed with the cyanobacterial culture. These bacteria, CB1 and CB2, were identified as
>
Identification of the axenic Arctic cyanobacterium
Molecular characterization showed that the Arctic cyanobacterium was
>
Cold-tolerance of Nodularia spumigena KNUA005
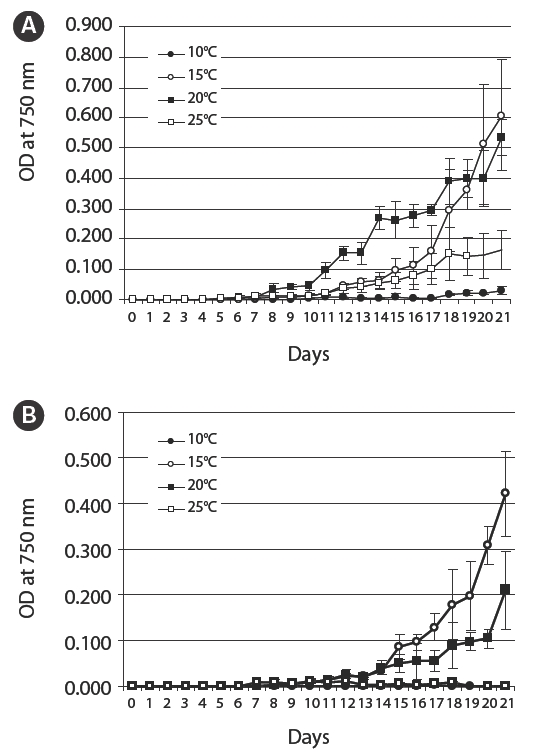
As shown in Fig. 4A, N. spumigena KNUA005’s optimal growth temperature in BG-110 was 20°C, but it was also able to grow well at 15°C. However, this heterocystous organism showed a tendency of growing slower in BG-11(+) medium than in nitrogen-free BG-110 medium (Fig. 4B). There was little or no cyanobacterial growth in either BG-110 or BG-11(+) media at 10°C, but the cyanobacterium remained alive and subsequently grew well when placed under favourable conditions (data not shown).
[Table 2.] Identification of contaminating bacteria isolated from the cyanobacterial culture

Identification of contaminating bacteria isolated from the cyanobacterial culture
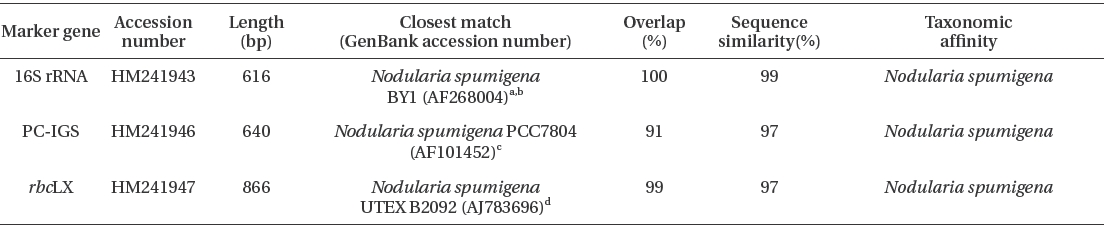
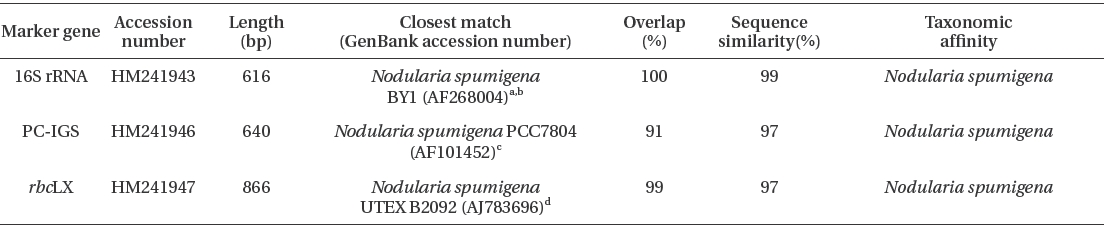
Results from BLAST searches using 16S rRNA, PC-IGS, rbcLX sequences of Nodularia spumigena KNUA005
Many research groups have successfully isolated cyanobacterial cultures from a variety of environmental samples. Nevertheless, cultures maintained in laboratories are mostly unialgal, not axenic cultures, which are not suitable for understanding genetic, biochemical or physiological properties of a particular taxon. In this study, we have developed a solid medium-based isolation method for effective axenic culture production of filamentous cyanobacteria.
Imipenem is a β-lactam antibiotic derived from
This method uses a combination of two antibiotics with different mechanisms of action in an attempt to prevent any contaminating bacterial growth. This approach may provide an effective way of axenic culture production for filamentous cyanobacteria from heavily contaminated environmental samples.
Our research group has already obtained four potential candidates for biofuel production that were isolated from cyanobacterial bloom samples in Lake Daecheong, Korea in late summer 2009 (unpublished data). It is hoped that new genetic strains may have potential for sustainable biofuel production under unfavourable weather conditions such as those experienced in the autumn and winter seasons.
However, it should be noted that