Hydrogels are crosslinked hydrophilic matrices suitable for a variety of biomedical applications, including tissue engineering matrix, contact lenses, drug delivery carriers and cell therapy (Kweon
Among the various physical and chemical methods for hydrogel crosslinking, we are particularly interested in visible light induced gelation (Hao and Lin, 2014; Hao
Gelation of SF depends on a number of parameters including concentration, temperature, mechanical strength, chemical treatment, ultrasound sonication, and composition (Kim
The authors reported the modulating properties of SF-PEG hydrogels were reported (Bragg
SF was purified from
>
Preparation of SF aqueous solution
SF was dissolved in ethanolic calcium chloride solution (CaCl2 : H2O : ethanol = 1 : 8 : 2 in molar ratio). The solution was heated to 90℃ for 4 h, cool down to room temperature, and subsequently dialyzed against ddH2O using dialysis membrane with MWCO 6-8 kDa (Fisher) for 2 d to remove the salts. After dialysis, the SF solution contained in the dialysis membrane was placed in a bath of dry PEG (10kDa) to concentrate the SF solution. The final concentration (wt./vol.%) of SF aqueous solution was determined from lyophilization of a small portion of the dialyzed SF solution (5 – 6 wt%). Sonication of SF solution was achieved using a Branson 450 Sonifier with a converter, an externally threaded disruptor horn, and 1/8” diameter-tapered microtip. To avoid forming, SF solution was sonicated for 1 s at 10% amplitude and then sonicated at 20% amplitude for 20 s (Wang
>
Hydrogel formation initiated by visible light
Hydrolytically degradable PEG hydrogels were formed by visible light initiated thiol-acrylate photopolymerization using PEGDA and dithiol liker. A typical pre-polymer solution was prepared in pH 7.4 PBS and contained macromer PEGDA (10 wt%), photosensitizer eosin-Y (0.1 mM), bi-functional co-initiator DTT (7.5 mM), and co-monomer NVP (0.1 vol%). macromers (Hao and Lin, 2014; Hao
Surface morphology of SF hydrogel was observed using scanning electron microscope (S-3500N, Hitachi, Japan). The hydrogels were coated with platinum and before observation. To investigate the molecular structure of the hydrogels, X-ray diffraction (XRD) was measured on X’Pert-Pro (PANalytical, Netherlands) using Cu-Kα radiation. The X-ray source was operated at 40 kV and 40 mA. Differential scanning calorimetry was analyzed using Differential Scanning Calorimeter (Mettler Toledo, Australia) at a scanning rate of 10℃/min over 50-350℃. Themrogravimetric analysis was done using Mettler Toledo ThermoGravimetric Analyzer (DSC 823e, Australia) from 35℃ to 800℃ at the heating rate 10℃/min with a nitrogen flow rate of 50 mL/min.
In this study, SF-PEG hydrogel crosslinked from PEGDA, co-monomer NVP, di-thiol containing linker DTT, and photosensitizer eosion-Y were evaluated by instrumental analysis on morphological, structural, and thermal properties.
>
Morphology of SF-PEG hydrogel
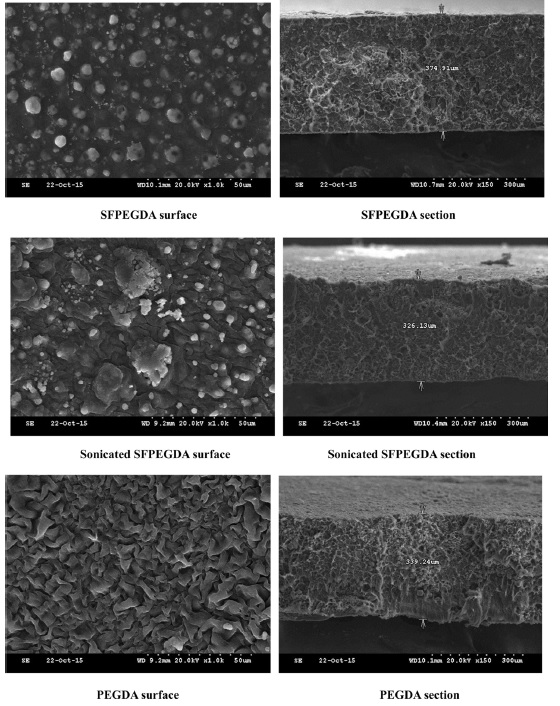
To observe the surface and fractured section of SF hydrogel, SEM is a powerful method. As Fig. 1, Surface of SFPEGDA showed dot and uneven surface morphology with network cross-section structure. Sonicated SFPEGDA shows some big and small islets with network cross section. However, PEGDA showed that relatively even surface morphology with network structure. This results means that SF and PEGDA were phase-separated and results in that SF was distributed in the PEG matrix.
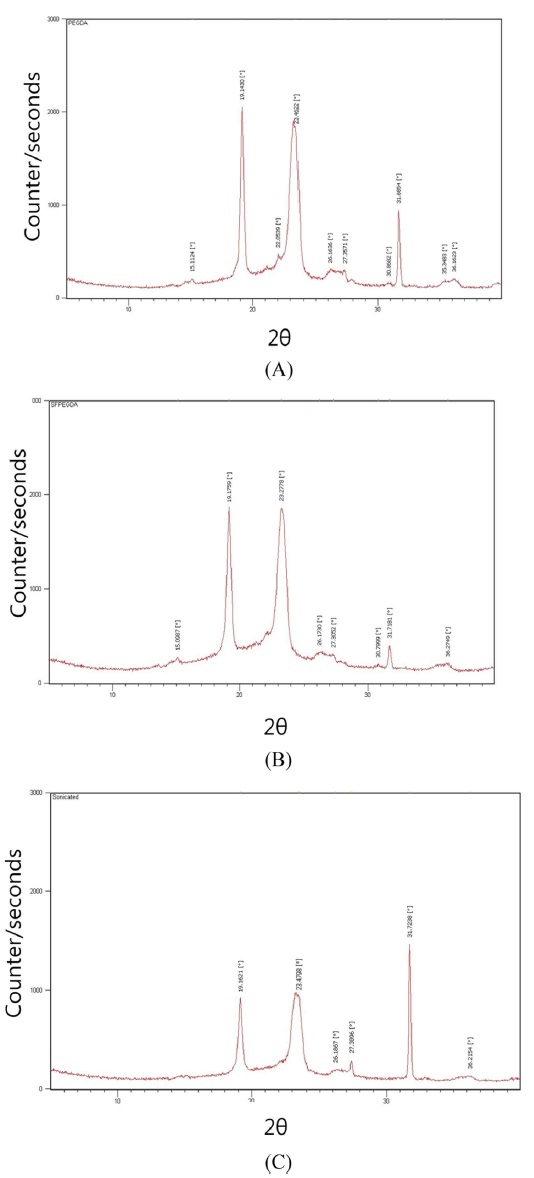
XRD curves of SF-PEG hydrogels are shown in Fig. 2 SF showed a typical amorphous diffraction pattern (data not shown), whereas PEGDA hydrogel showed sharp diffraction peak at 2θ=19.1, 23.5, and 31.7° (Kweon
SF was entrapped in crosslinked PEG networks. XRD curves show that entrapped SF does not affect the crystal structure of PEG. Kweon
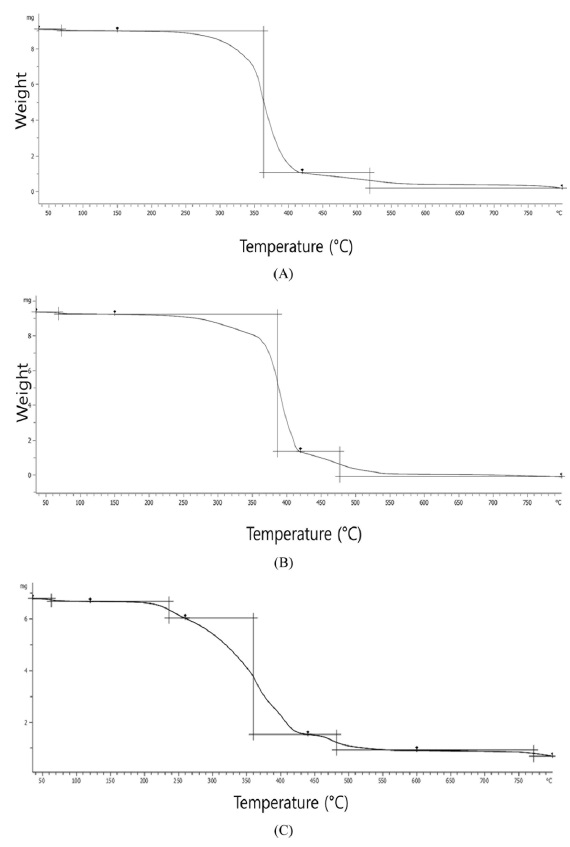
Fig. 3 showed the thermogravimetric curves of PEGDA, SFPEGDA and sonicated SFPEGDA. PEGDA showed slightly two step mass loss around 230-350℃ and 350-390℃. SFPEGDA showed two step weight losses around 220-360℃ and 360℃ and 390℃. On the other hand, sonicated one showed more complex degradation behavior from 180-420℃
Decomposition of pure PEG were occurred in one step around 270-370℃ (Massoumi
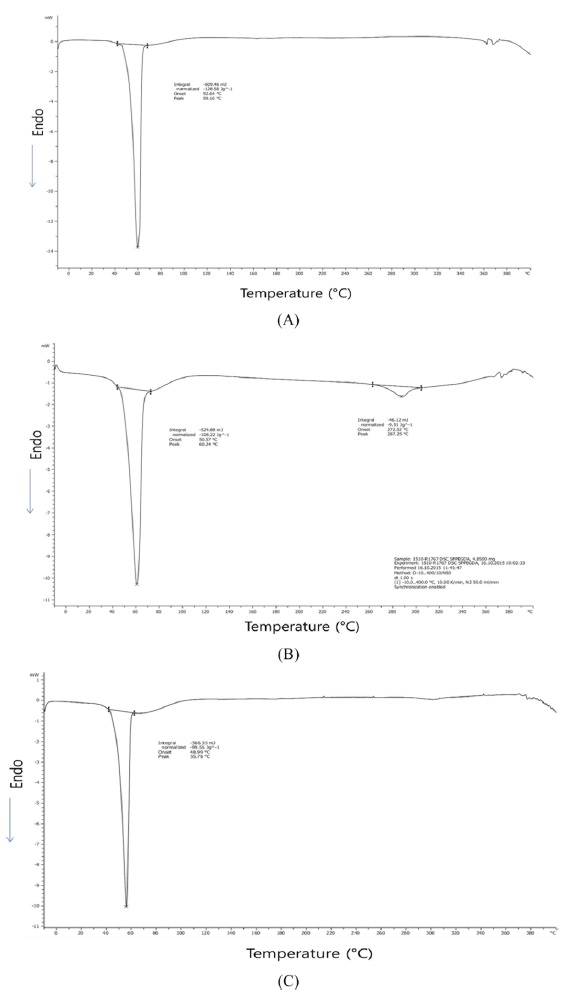
Fig. 4 showed the DSC thermograms of PEGDA, SFPEGDA, and sonicated SFPEGDA. PEGDA shows a sharp endothermic peak at 59.2℃ with ΔHm 128.6 J/g and SFPEGDA shows two endothermic peaks at 60.2℃ with 108.2 J/g and at 287℃ with 9.5 J/g. Sonicated SF hydrogel (Fig. 4(c)) also shows two endothermic peaks at 55.8℃ with 99.6 J/g and weak peaks around 290℃.
The melting temperature of PEG crystalline observed at 50- 60℃ (Icart